Abstract
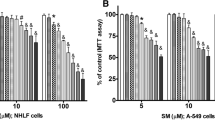
To investigate the mutation inducibility of surfactin C, we performed the chromosome aberration assay with Chinese hamster lung cells in vitro. The colorimetric MTT screening assay was carried out to determine the cytotoxicity index (IC50) of surfactin C. The IC50 value was 125 μg/ml. For the chromosome aberration test of surfactin C, the maximum concentration was employed as 125 μg/ml, followed by 62.5 and 31.25 μg/ml for the lower concentrations, with or without metabolic activation (S9). Cyclophosphamide and mitomycin C were used as positive controls in the presence and absence of S9 metabolic activation, respectively. These results showed that surfactin C was not capable of inducing chromosome aberration, as measured by the chromosome aberration test using Chinese hamster lung cell line. There is no evidence for surfactin C to have a genotoxic potential.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Arima, K., Kakinuma, A. and Tamura, G. (1968). Surfactin, a crystalline peptidolipid surfactant produced by Bacillus subtilis: isolation, characterization and its inhibition of fibrin clot formation. Biochem. Res. Commun., 31, 488–494.
Banat, I.M., Makkar, R.S. and Cameotra, S.S. (2000). Potential commercial applications of microbial surfactants. Appl. Microbiol. Biotechnol., 53, 495–508.
Dearfield, K.L., Cimino, M.C., McCarroll, N.E., Mauer, I. and Valcovic, L.R. (2002). Genotoxicity risk assessment: a proposed classification strategy. Mutat. Res., 521, 121–135.
Desai, J.D. and Banat, I.M. (1997). Microbial production of surfactants and their commercial potential. Microbiol. Mol. Biol. Rev., 61, 47–64.
Galloway, S., Aardema, M., Ishidate, M., Ivett, J., Kirkland D., Morita, T., Mosesso, P. and Sofuni, T. (1994). Report from the working group on in vitro tests for chromosomal aberrations. Mutat. Res., 312, 241–262
Georgiou, G., Liu, S.C. and Sharma, M.M. (1990). Surface active compounds from microorganisms. Biotechnol., 10, 60–65.
Hwang, M.H., Lim, J.H., Kim, K.S., Rhee, M.H., Kim, N.W., Kim, J.C. and Park, S.C. (2005a). Antibacterial activity in vitro and primary dermal irritationtest in rabbits of surfactin produced in bacillus subtilus complex BC2121. J. Toxicol. Pub. Health, 20, 39–43.
Hwang, M.H., Lim, J.H., Yun, H.I., Rhee, M.H., Cho, J.Y., Hsu, W.H. and Park, S.C. (2005b). Surfactin C inhibits the lipopolysaccharide-induced transcription of interleukin-1beta and inducible nitric oxide synthase and nitric oxide production in murine RAW 264.7 cells. Biotechnol Lett., 27, 1605–1608.
Hwang, Y.H., Park, B.K., Lim, J.H., Kim, M.S., Song, I.B., Park, S.C. and Yun, H.I. (2008). Evaluation of genetic and developmental toxicity of surfactin C from Bacillus subtilis BC1212. J. Health Sci., 54, 101–106.
Kanatomo, S., Nagai, S., Ohki, K. and Yasuda, Y. (1995). Study on surfactin, a cyclic depsipeptide. I. Isolation and structure of eight surfuctin analogs produced by Bacillus natto KMD 2311. Yakugaku. Zasshi., 115, 756–764.
Kikuchi, T. and Hasumi, K. (2002). Enhancement of plasminogen activation surfactin C: augmentation of fibrinolysis in vitro and in vivo. Biochem. Biophys. Acta., 1596, 234–245.
Lim, J.H., Park, B.K., Kim, M.S., Hwang, M.H., Rhee, M.H., Park, S.C. and Yun, H.I. (2005). The anti-thrombotic activity of surfactins. J. Vet. Sci., 6, 353–355.
Mulligan, C.N. (2005). Environmental applications for biosurfactants. Environ Pollut., 133, 183–198.
O’Brien, P.J., Hales, B.F., Josephy, P.D., Castonguay, A., Yamazoe, Y. and Guengerich, F.P. (1996). Chemical carcinogenesis, mutagenesis and teratogenesis. Can. J. Physiol. Pharmacol., 74, 565–571.
Organization for Economic Cooperation and Development (OECD, 1997). Test Guideline 473, In vitro Mammalian chromosome aberration test. In: OECD Guidelines for the Testing of Chemicals. OECD Paris.
Park, B.K., Lim, J.H., Hwang, Y.H., Kim, M.S., Song, I.B., Lee, H.G., Han, S.J., Hwang, M.H., Kim, J.W., Park, S.C., Rhee, M.H. and Yun, H.I. (2006). Acute oral toxicity of surfactin C in mice. J. Toxicol. Pub. Health, 22, 453–458.
Rodrigues, L., Banat, I.M., Teixeira, J. and Oliveira, R. (2006). Biosurfactants: potential applications in medicine. J. Antimicrob. Chemother., 57, 609–618.
Singh, P. and Cameotra, S.S. (2004). Potential applications of microbial surfactants in biomedical sciences. Trends Biotechnol., 22, 142–146.
Scott, D., Danford, N., Dean, B., Kirkland, D. and Richardson C. (1983). In vitro chromosome aberration assays, Report of the UKEMS Sub-Committee on Guidelines for Mutagenicity Testing, Part 1, Cambridge University Press, Cambridge, UK, pp. 41–64.
Takahashi, T., Ohno, O., Ikeda, Y., Sawa, R., Homma, Y., Igarashi, M. and Umezawa, K. (2006). Inhibition of lipopolysaccharide activity by a bacterial cyclic lipopeptide surfactin. J. Antibiot (Tokyo), 59, 35–43.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lim, J.H., Song, I.B., Park, B.K. et al. Genotoxic Evaluation of Surfactin C in Chinese Hamster Lung Cell Line. Toxicol Res. 25, 47–50 (2009). https://doi.org/10.5487/TR.2009.25.1.047
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.5487/TR.2009.25.1.047