Abstract
Research has documented neurophysiological indicators of anticipation (Stimulus Preceding Negativity [SPN]) and perception (Late Positive Potential [LPP]) of threat, yet little is known as to how self-focused attention manipulations influence emotion processing within the context of cued picture viewing. With self-referent attention moderating attention to external stimuli, it is necessary to document how self-focused attention impacts attention and the ability to emotionally process external threat. The goal of the present study was to evaluate the impact of self-focused attention on the anticipation and perceptual processing of unpleasant pictures within a cued-picture viewing paradigm among 33 participants. Overall, the results suggest that the self-focused attention manipulations disrupted anticipation but not processing of pictures, as indexed by the SPN and LPP respectively. Self-focused attention appears to disrupt the preparatory attention for upcoming unpleasant stimuli, potentially through loading cognitive resources or activation of associative defensive responding. Collectively, these findings demonstrate the impact of self-focused attention within the context of emotional picture processing and suggest further areas of investigation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Decades of literature on emotional information processing provides insight into the attentional, motivational, and physiological systems engaged when anticipating and perceiving unpleasant and threatening information, as well as how these systems are modulated by psychopathology (Bradley et al., 2001; Lang et al., 1997; Lang et al., 2016). Multiple contextual factors impact the processing of emotional information (Bradley et al., 2001; Wangelin et al., 2011). Two such factors are anticipation and attentional demands (Judah et al., 2016; Mills et al., 2014; Sege et al., 2015; Sege et al., 2017; White & Grant, 2017), yet little is known how manipulation of attention impacts anticipation and perception of external threat. The purpose of this study was to document how manipulation of attention focus (i.e., self-focus) impacts electrocortical markers of emotional processing for unpleasant pictures during anticipation and perception.
Research on emotional processing using picture viewing tasks suggests that corresponding behavior, physiology, and attention largely represent appetitive or defensive motivations (Lang & Bradley, 2013). Unpleasant pictures activate defensive motivation systems, which regulate threat perception (e.g., arousal, attention) to support behavioral responding, such as avoidance, escape, or fight (Bradley et al., 2001). Consistent with this perspective, empirical data have demonstrated that unpleasant pictures elicit greater allocation of attention (Calvo & Lang, 2004). Utilizing the late positive potential (LPP), a positive-going event-related potential (ERP) occurring at posterior electrode sites between 400-1,000 ms after the onset of a stimulus (Hajcak & Olvet, 2008; Schupp et al., 2006), which indexes motivated attention, researchers have found potentiation resulting from viewing unpleasant and threatening pictures compared with neutral pictures (Cuthbert et al., 2000; Hajcak et al., 2009; White & Grant, 2017). Collectively, these studies, as well as others (Bradley et al., 2001), indicate that threatening and unpleasant pictures activate defensive motivation systems, including increased allocation of attentional resources during perception.
One factor that impacts this emotional processing system is cueing (Grant et al., 2015; Judah et al., 2013; Sege et al., 2015, 2018; White & Grant, 2017). Cued-picture viewing has been shown to modulate these systems during anticipation, such that defensive activation during anticipation of unpleasant pictures is enhanced (Sabatinelli et al., 2001). Using the stimulus-preceding negativity (SPN), a negative slow-going ERP measured at frontocentral electrode sites occurring just before the onset of an emotionally or motivationally significant stimulus, which indexes anticipatory preparation to upcoming stimuli (Van Boxtel & Böcker, 2004), studies demonstrate enhanced neural attentional recruitment during anticipation of threatening and unpleasant pictures relative to neutral pictures (Amrhein et al., 2005; Poli et al., 2007). Predictable cueing not only influences attentional anticipation but also defensive responses during perception of pictures. For example, startle responses are shown to be attenuated during the perception of cued threatening pictures (Sege et al., 2014, 2015), representing a blunting of defensive responding during perception of a more predictable unpleasant event compared with unpredictable. Thus, cueing of aversive stimuli activates defensive motivational systems during anticipation, which results in attenuation of defensive responding during emotional processing of the stimuli.
Clearly, predictive cueing of unpleasant pictures modulates various indicators of anticipation and perception. However, many studies employing a cued picture paradigm have specifically utilized an instructed cuing approach, wherein participants are informed of the S1 cue association with a subsequent S2 picture. These studies have not evaluated 1) whether this contingency immediately leads to enhanced anticipatory processing; and 2) whether uninstructed cueing could still lead to anticipatory processing via associative learning. Accordingly, the current study utilized an uninstructed cued paradigm that reflects a more naturalistic method for investigating anticipatory processing and associative learning processes.
In addition to picture valance and external contexts (i.e., cueing), internal states are exceptionally important in emotional processing, because they provide interoceptive cues of arousal and help to regulate attentional resources (García-Cordero et al., 2017). In line with prior work suggesting that interoceptive cues enhance self-focused attention, empirical investigations have manipulated cues that are effective and reliable for inducing self-focused attention (Mansell et al., 2003; Papageorgiou & Wells, 2002; Wells & Papageorgiou, 2001) and increasing anxiety across both anxious and control participants (Wild et al., 2008). Moreover, self-focused attention using a false cue of elevated heart rate is implemented to determine how heightened interoceptive awareness impacts neural markers of inhibition and shifting (Judah et al., 2013), error monitoring and feedback anticipation (Judah et al., 2016), and attention to heartbeat (Judah et al., 2018). Specifically, enhanced self-focused attention reduces preparatory activity in basic cognitive control tasks (Judah et al., 2013), as well as error-related feedback processing, but not error monitoring directly (Judah et al., 2016), potentially representing the deleterious impacts of self-focus on action preparation and feedback processing. Moreover, attentional processing generally is thought to have a limited capacity and is influenced by competing demands. Interestingly in emotional modulations of attention, affective pictures have been shown to modulate event-related potential anticipation and attention deployment (Cuthbert et al., 2000; Grant et al., 2015; Poli et al., 2007; White & Grant, 2017). However, it is unclear whether the blunting influence of self-focused attention on anticipation and attentional responding reported previously will impact anticipation and perception of emotional stimuli.
Collectively, research has documented neurophysiological phenomenon of anticipation and perception of unpleasant pictures, yet little is known as to how self-focused attention manipulations influence emotion processing within the context of cued picture viewing. With self-referent attention moderating attention to external stimuli (Judah et al., 2016), it is necessary to document how self-focused attention impacts attention and the ability to anticipate and process external threat information. The current study employed a cued-picture viewing paradigm (i.e., S1-S2 task) to evaluate how cue a manipulation of self-focused attention impacted motivated attention (i.e., the anticipation and perception of unpleasant pictures). To accomplish this goal, a “+” cue (S1; yellow or blue and 0° or 45°) indicated the valence of the upcoming picture (S2; unpleasant or neutral) via cue color and indicated elevations in basal heart rate via cue rotation. The technique of instructed cue rotation regarding heart rate has previously been shown to effectively induce interoceptive self-focus (Judah et al., 2016).
It was hypothesized that the self-focused attention manipulation will result in decreased ability to effectively prepare for upcoming pictures and/or process the pictures themselves. Based on previous research, it was expected that self-focused attention would increase defensive activations and/or decrease attentional resources to the task, thereby resulting in decreased emotional processing of unpleasant pictures. More specifically, it was expected that the SPN would be potentiated in preparation of unpleasant pictures, but that this would be attenuated during trials with self-focused attention prompts as self-referent information may consume attentional resources away from external threat. This effect was expected to be present in the second half of the task, after the cue-picture relationship was established. It was hypothesized that the LPP would be potentiated for unpleasant pictures, but this effect also would be attenuated by the self-focused cue.
Methodology
Participants
Participants were recruited from a large Midwestern university (N = 27) and from the community (N = 6). The sample was slightly more male (54.5%) with an average age of 21.79 years (SD = 5.95; range = 18-49). Participants identified as white (63.6%), Latinx (12.1%), Asian (9.1%), or Native American (3.0%), whereas 12.1% identified multiple groups. A power analysis using effect sizes found in similar studies (Judah et al., 2016) suggested that 30 participants were required for adequate power for the hypothesized within-subject’s effects (α = 0.05, power = 0.80). For this reason, we tried to include at least 30 total participants.
Materials
Visual stimuli
Negative and neutral pictures were taken from the International Affective Picture Systems (IAPS: Lang et al., 2005). The IAPS represents a collection of emotional pictures that have been standardized for valence (positive, negative, neutral) and arousal level and often are used for empirical investigation of attention and emotion processing. Fifty threat pictures were chosen that depicted violence, extreme poverty, carnage, and other distressing content (mean valance = 3.04, SD = 1.08); mean arousal = 6.03, SD = 0.78). Fifty neutral pictures were chosen that depicted plants, people engaged in everyday activities, and other relatively nonemotional content (mean valance = 5.50, SD = 0.73; mean arousal = 3.42, SD = 0.82).Footnote 1
Procedure
All procedures fell in line with the declaration of Helsinki and were approved by the Institutional Review Board at the university where the study was conducted. Participants were provided informed consent before completing all study demographics and questionnaires. Participants were seated approximately 70 cm from the stimulus presentation monitor and EEG electrodes were attached. Participants then completed a cued picture viewing task. After completion of the study’s procedures, participants were compensated ($20) for their time.
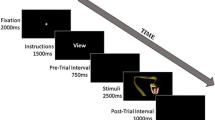
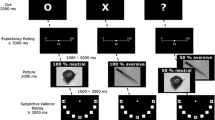
Cued picture viewing task
A modified version of the S1-S2 tasks used in previous research was used for cued picture viewing (Grant et al., 2015; Fig. 1). Participants were instructed that they would see a series of pictures and that, prior to each picture (S2), they would see a colored (either yellow or blue) “+” cue (S1) that indicated a picture was about to be presented. The color of the S1, which was counterbalanced across participants, was paired with the valence of S2, although participants were uninstructed regarding the color-valence pairing (e.g., they were not informed that a yellow cue indicated a subsequent neutrally valanced picture). In addition to the color indicating the valence of the upcoming picture, the S1 stimuli also were manipulated to enhance self-focused attention. The S1 cue was presented in two orientations (0° or 45° rotation) in a 50:50 ratio. Participants were told that one of the orientations would appear when their heart rate increased (Judah et al., 2016). As in prior protocols (Judah et al., 2016), to increase believability, participants were told that this was caused by a software glitch and that they could ignore it as it was not relevant to the task. The orientation was randomized across the task, and the 0°/45° rotation-HR pairing was counterbalanced across participants. The order of stimuli presentation within subject was also randomized. Participants viewed the S1 stimulus for 500 ms, followed by a 3,500-ms interstimulus interval, and the S2 stimulus for 2,000 ms.
Electrophysiological measures
Electroencephalographic (EEG) data were recorded using a 32-channel (Fp1/2, AF3/4, F7/8, F3/4, FZ, FC5/6, FC1/2, T7/8, C3/4, CZ, CP5/6, CP1/2, P7/8, P3/4, PZ, PO3/4, O1/2, and OZ) BioSemi Active II system (BioSemi, Amsterdam, The Netherlands) and were sampled at 256 Hz. Two electrodes (Common Mode Sensor active and Driven Right Leg passive) were incorporated into the electrode cap and used as ground electrodes. Electrodes were placed on the left and right mastoids and were used for references (Luck, 2014). Vertical and horizontal eye movement (EOG) data were collected using electrodes placed 1 cm below the left eye and 1 cm to the left and right of the outer canthus of each eye.
EEGLAB version 2021.0 (Delorme & Makeig, 2004) and ERPLAB version 8.10 (Lopez-Calderon & Luck, 2014) were used to process the data. Offline, data were re-referenced to the average of the left and right mastoids (Luck, 2014) and filtered using a band-pass filter (0.01–30 Hz) with a half-amplitude cutoff and a 12 dB/oct roll-off and a 60-Hz notch filter. Ocular artifacts (e.g., blinks, saccades) were isolated and corrected by using independent components analysis (ICA; Luck, 2014) with manual identification of components with spatial and temporal characteristic of ocular artifacts consistent with those reported in previous research (Mognon et al., 2011). Automated routines in ERPLAB were used to detect and reject large (200 μV) voltage changes at any electrode in a 200-ms window moving at 50-ms steps. Previous research informed a priori measurement sites and time windows for ERPs and was confirmed with visual inspection. The SPN was measured from the FZ electrode in the 200 ms before the onset of S2 (Van Boxtel & Böcker, 2004) and baselined using the 200 ms before the onset of S1. The LPP was measured at PZ 400-1,000 ms after S2 onset (Cuthbert et al., 2000) and baselined 200 ms prior to S2 onset. Less than 25% of trials were rejected for any subject. The average number of rejected trials across all conditions was 42.93 (SD = 36.46; % of total trials = 18.63). The number of trials rejected did not differ per condition (all p’s > 0.152). Previous research (Grant et al., 2015) suggested that the first and second halves of the task should be evaluated separately as encoding of the cue/picture pairing is ongoing in the first half, whereas the pairing has been encoded in the second. Thus, the first and second half of the task are separated for analyses.
Analytic Approach
To evaluate the impact of cuing and self-focused attention on anticipation and subsequent perception of pictures, two separate 2 (Cue: Self-Focus Cue vs. Standard Cue) × 2 (Stimuli Type: Negative vs. Neutral) × 2 (Task Half: H1 vs. H2) repeated-measures ANOVAs were used to evaluate SPN and LPP mean amplitude, respectively. Sphericity was assessed using Mauchly’s Test of Sphericity. The results section indicates where there are violations and in these instances the adjustments were made using the Greenhouse-Geisser correction. When examining pairwise comparisons, the Bonferroni adjustment was used when necessary to follow up for significant interactions. For brevity, only significant (p < 0.05) effects are presented in the results section; however, all effects are presented in Table 1. Bayesian factors (BF) for each effect were calculated to determine the strength of the effect relative to the null hypothesis, such that BF values greater than 1.00 suggest that data are more likely to occur under the research hypothesis, whereas BF values less than 1.00 suggest that data are more likely to occur under the null hypothesis. Evaluation of the early posterior negativity (EPN) was also conducted and can be found in the supplement.
Results
All analyses presented within this section were additionally computed when including worry, as indexed by scores on the Penn State Worry Questionnaire (Meyer et al., 1990), as a covariate based on research suggesting the impact of state anxiety on cued picture viewing. No significant interactions or main effects were observed when including this covariate.
SPN
Results showed a main effect of Cue (F(1,32) = 6.45, p = 0.016, ηp2 = 0.17) such that the SPN was attenuated following Self-Focus Cues (M = 0.43, SE = 1.24) compared with Standard Cues (M = 2.86, SE = 1.03). This was qualified by an interaction of Cue, Stimuli Type, and Task Half (F(1,32) = 6.85, p = 0.013, ηp2 = 0.18). Follow-up pairwise comparisons were used to investigate this interaction, which revealed that the SPN amplitudes were reduced for Self-Focus Cues (M = 5.45, SE = 2.07) relative to Standard Cues (M = −4.78, SE = 1.72) for an upcoming unpleasant picture specifically during the latter half of the task. No other pairwise comparison was significant. Grand-average SPN waveforms are presented in Fig. 2.
Grand averaged SPN waveforms and mean SPN amplitudes. Note. a SPN waveform from first half of trials. b SPN waveform from second half of trials. c and d SPN means and standard errors for first and second halves, respectively. H1 = first half. H2 = second half. Standard = Standard Cue. Self-Focus = Self-Focus Cue. Waveform scale is in microvolts (μV) and milliseconds (ms). SPN was measured from electrode Fz in the 200 ms before the onset of S2 and baselined using the 200 ms before the onset of S1. Yellow highlight on a and b represents measurement window
LPP
Results revealed a significant main effect of Stimuli Type, such that there was an enhanced LPP amplitude for unpleasant (M = 9.67, SE = 1.16) compared with neutral pictures (M = 4.26, SE = 0.89), F(1,32) = 47.34, p < 0.001, ηp2 = 0.60. Grand-average LPP waveforms are presented in Fig. 3.
Grand averaged LPP waveforms and mean LPP amplitudes. Note. a LPP waveform from first half of trials. b LPP waveform from second half of trials. c and d LPP means and standard errors for first and second halves, respectively. H1 = first half. H2 = second half. Standard = Standard Cue. Self-Focus = Self-Focus Cue. Waveform scale is in microvolts (μV) and milliseconds (ms). LPP was measured at Pz 400-1,000 ms after S2 onset, and baselined 200 ms before S2 onset. Yellow highlight on a and b represents measurement window
Discussion
The goal of the present study was to evaluate the impact of self-focused attention on the anticipation and perceptual processing of unpleasant pictures within a cued-picture viewing paradigm. Overall, the results provided some support for the hypotheses, such that the self-focused attention manipulation disrupted anticipation but not processing of pictures. Specifically, the SPN was attenuated as a result of the self-focus cue before unpleasant pictures, although the cue did not subsequently modulate the LPP, suggesting processing of the picture was unhindered despite decreased anticipatory preparation of attentional resources. These findings provide significant insight into how self-focused attention disrupts preparation of attention resources during cued unpleasant picture viewing. These results also document that self-focused attention manipulations during anticipation did not affect motivated attention during perception, indicating that increased attentional resources during perception of threat may be robust to self-focused attention during anticipation.
Consistent with hypothesized effects, the self-focused attention cue resulted in an attenuated SPN to unpleasant pictures compared with a standard fixation cue on the second but not first half of trials. These results suggest that self-focused attention attenuates anticipation of upcoming unpleasant stimuli and that this associative learning occurs relatively quickly. SPN blunting potentially represents decreased anticipation or preparatory responding to the upcoming threat after participants learn the meaning of the predictable cues. Whereas potentiated anticipatory activity has been observed for upcoming threat stimuli (Poli et al., 2007; Sabatinelli et al., 2001), these results indicate that self-focused attention blunts this effect. Notably, self-focused attention, like that which was initiated by the self-focus cue used in this study, has been shown to increase anxiety, increase avoidance, and distract from task-focused attention (Norton & Abbott, 2016). Given these data were evaluated within the context of a relatively healthy sample, these results also could be suggestive of potentially adaptive responding to cues of internal threat (e.g., anxious arousal).
Based on defensive motivation literature, it is possible that the self-focused attention cue, which is a manipulation of increased physiological arousal, resulted in activation of the defensive motivation system (Lang et al., 1997). The activation of this system may have blunted responding to additional anticipated threat, through the early application of coping strategies (i.e., distraction; Sege et al., 2015). Research often documents that activating arousal before anxious stimuli is presented results in the application of coping strategies before the presentation of anxious stimuli (Monat, 1976). This blunting of anticipation suggest that self-focused attention may be used as a distraction strategy or may activate other coping strategies, which hinder anticipatory processing of threat. For example, in healthy participants, internal cues of anxious arousal may deploy attentional resources in order to manage or cope with the cause of the anxiety. Whereas this decreases preparation for an upcoming potential threat (i.e., the unpleasant picture in this case), deployment of this attention does not impair subsequent processing and motivated attention to the threat. Alternatively, this mechanism is not yet fully supported through these data, so self-focused attention also could suppress anticipatory attentional preparation for unpleasant pictures, potentially through avoidance. If this explanation is, in fact, true, then it would be expected that our effects could be exacerbated by high levels of trait anxiety or high levels of social anxiety compared with low levels of anxiety similar to the results of Sege et al. (2017; in press).
It also is possible that, through the framework of limited attention resources (Derakshan & Eysenck, 2009), self-focused attention draws attention resources towards the self, resulting in decreased attention resources to process the upcoming threatening stimuli. This is consistent with literature showing self-focused attention results in decreased attention resource allocation to processing task-relevant stimuli (Judah et al., 2016). Similarly, researchers have found that the LPP amplitude to emotional stimuli is reduced under cognitive load, suggesting that allocation of attentional resources may modulate the processing of emotional stimuli (MacNamara et al., 2011). This also was true in a picture viewing paradigm (White & Grant, 2017). It is plausible that the self-focused attention acted as a cognitive load, which resulted in disrupted anticipatory processing of emotional stimuli. Regardless, these data suggest that self-focused attention may disrupt or distract from attentional preparation for upcoming threat pictures, although the mechanism for which this occurs is not yet clear. Future studies could explore these explanations by testing these effects across varying levels of anxiety and/or cognitive load/executive control (Grant et al., 2022).
Self-focused attention did not have an impact on perceptual processing of pictures in general. These results also replicate well-established enhancements in LPP amplitude for unpleasant pictures compared to neutral (Cuthbert et al., 2000; Hajcak et al., 2009; White & Grant, 2017), suggesting that different levels of emotional processing of threatening versus neutral pictures appears to be robust against self-focused attention. Taken together, this pattern of findings indicates that the self-focused attention dampens attention preparation, which does not have downstream effects on attention deployment during perception. Self-focused attention did not uniquely impact attention deployment to unpleasant pictures, suggesting that if defensive systems were activated by the self-focused attention manipulation, they may not have had downstream effects. This pattern of effects may be more supportive of an attention resource allocation interpretation of the SPN data as opposed to a defensive activation, as a defensive activation interpretation may suggest downstream effects on the LPP as well.
There are limitations to this study. ERPs represent only one aspect of emotional processing. Incorporating a multimodal assessment of the impact of self-focused attention on cued picture viewing would provide a more complete picture of emotionally motivated response systems. Although these effects represent insight into a novel aspect of emotional processing, clinical utility of the findings depends upon examination of such effects in clinical samples or in relation to dimensional symptomology. Considering emotional processing disruptions occur across many forms of psychopathology, it would be important to extend these findings to clinical populations. Moreover, the current study did not include pleasant pictures, which would provide more data regarding appetitive versus defensive responding. Additionally, the SPN (and LPP) represent neural activity within the measurement window in addition to the preceding activity (Luck, 2014). The baselines used within this study are based on extensive previous research; however, it is important to recognize that neural activity before the measurement window may contribute to the effects.
Conclusions
Emotional processing during a picture viewing task requires several cognitive processes, two of which were investigated in this study (i.e., attentional preparation and attention deployment). The purpose of this study was to investigate the impact of self-focused attention manipulations on these attentional processes within the context of cued-picture viewing. Self-focused attention appears to disrupt the preparatory attention for upcoming unpleasant stimuli, potentially through loading cognitive resources or activation of associative defensive responding. Self-focused attention does not appear to have a downstream impact on the subsequent attention deployed during perception of pictures, although future evaluations should test these effects in high trait or clinical levels of anxiety and/or social phobia. Collectively, these findings demonstrate the impact of self-focused attention on anticipation within the context of emotional picture processing.
Data availability
Data are not openly available; however, material can be made available upon request. This study was not preregistered.
Code availability
Code is not openly available.
Notes
The specific IAPS pictures used for the unpleasant valence consisted of pictures portraying violence, negative, and threatening scenes (1019, 1022, 1026, 1040, 1050, 1090, 1122, 1200, 1202, 1230, 1274, 1303, 1304, 1321, 1560, 1617, 2690, 2694, 2770, 2811, 3051, 3080, 3102, 3110, 3261, 3530, 5040, 6021, 6022, 6230, 6241, 6242, 6313, 6350, 6360, 6370, 6510, 6540, 6560, 6570, 6571, 6821, 6940, 9050, 9252, 9403, 9409, 9414, 9921); and neutral pictures portrayed household objects and daily tasks (1333, 1350, 1670, 2020, 2032, 2036, 2038, 2101, 2107, 2190, 2191, 2210, 2211, 2383, 2880, 5471, 5660, 5731, 5800, 5811, 5870, 5875, 5900, 7000, 7001, 7002, 7010, 7014, 7034, 7035, 7036, 7038, 7043, 7044, 7045, 7100, 7224, 7233, 7490, 7500, 7710, 7830, 7950, 8220, 8260, 8312, 8325, 8510, 9469, 9260).
References
Amrhein, C., Pauli, P., Dengler, W., & Wiedemann, G. (2005). Covariation bias and its physiological correlates in panic disorder patients. Journal of Anxiety Disorders, 19(2), 177–191.
Bradley, M. M., Codispoti, M., Cuthbert, B. N., & Lang, P. J. (2001). Emotion and motivation I: Defensive and appetitive reactions in picture processing. Emotion, 1(3), 276.
Calvo, M. G., & Lang, P. J. (2004). Gaze patterns when looking at emotional pictures: Motivationally biased attention. Motivation and Emotion, 28(3), 221–243.
Cuthbert, B. N., Schupp, H. T., Bradley, M. M., Birbaumer, N., & Lang, P. J. (2000). Brain potentials in affective picture processing: Covariation with autonomic arousal and affective report. Biological Psychology, 52(2), 95–111.
Delorme, A., & Makeig, S. (2004). EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. Journal of Neuroscience Methods, 134(1), 9–21.
Derakshan, N., & Eysenck, M. W. (2009). Anxiety, processing efficiency, and cognitive performance: New developments from attentional control theory. European Psychologist, 14(2), 168–176.
García-Cordero, I., Esteves, S., Mikulan, E. P., Hesse, E., Baglivo, F. H., Silva, W., et al. (2017). Attention, in and out: Scalp-level and intracranial EEG correlates of interoception and exteroception. Frontiers in Neuroscience, 11, 1–14.
Grant, D. M., Judah, M. R., White, E. J., & Mills, A. C. (2015). Worry and discrimination of threat and safety cues: An event-related potential investigation. Behavior Therapy, 46(5), 652–660.
Grant, D. M., Judah, M. R., White, E. J., & Mills, A. C. (2022). Electrocortical evidence of biased attention to safety cues and stimuli among worriers. Current Psychology, 1–10.
Hajcak, G., & Olvet, D. M. (2008). The persistence of attention to emotion: Brain potentials during and after picture presentation. Emotion, 8(2), 250–255.
Hajcak, G., Dunning, J. P., & Foti, D. (2009). Motivated and controlled attention to emotion: Time-course of the late positive potential. Clinical Neurophysiology, 120(3), 505–510.
Judah, M. R., Grant, D. M., Mills, A. C., & Lechner, W. V. (2013). The neural correlates of impaired attentional control in social anxiety: An ERP study of inhibition and shifting. Emotion, 13(6), 1096–1106.
Judah, M. R., Grant, D. M., & Carlisle, N. B. (2016). The effects of self-focus on attentional biases in social anxiety: An ERP study. Cognitive, Affective, & Behavioral Neuroscience, 16(3), 393–405.
Judah, M. R., Shurkova, E. Y., Hager, N. M., White, E. J., Taylor, D. L., & Grant, D. M. (2018). The relationship between social anxiety and heartbeat evoked potential amplitude. Biological Psychology, 139, 1–7.
Lang, P. J., & Bradley, M. M. (2013). Appetitive and defensive motivation: Goal-directed or goal-determined? Emotion Review, 5(3), 230–234.
Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (1997). Motivated attention: Affect, activation, and action. In P. J. Lang, R. F. Simons, & M. T. Balaban (Eds.), Attention and orienting: Sensory and motivational processes (pp. 97–136). Erlbaum.
Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (2005). International Affective Picture System (IAPS): Digitized photographs, instruction manual, and affective ratings (Tech. Rep. No. A-6). University of Florida, Center for Research in Psychophysiology.
Lang, P. J., McTeague, L. M., & Bradley, M. M. (2016). RDoC, DSM, and the reflex physiology of fear: A biodimensional analysis of the anxiety disorders spectrum. Psychophysiology, 53(3), 336–347.
Lopez-Calderon, J., & Luck, S. J. (2014). ERPLAB: An open-source toolbox for the analysis of event-related potentials. Frontiers in Human Neuroscience, 8(213), 1–14.
Luck, S. J. (2014). An introduction to the event-related potential technique. MIT Press.
MacNamara, A., Ferri, J., & Hajcak, G. (2011). Working memory load reduces the late positive potential and this effect is attenuated with increasing anxiety. Cognitive, Affective, & Behavioral Neuroscience, 11(3), 321–331.
Mansell, W., Clark, D. M., & Ehlers, A. (2003). Internal versus external attention in social anxiety: An investigation using a novel paradigm. Behaviour Research and Therapy, 41(5), 555–572.
Meyer, T. J., Miller, M. L., Metzger, R. L., & Borkovec, T. D. (1990). Development and validation of the penn state worry questionnaire. Behaviour research and therapy, 28(6), 487–495.
Mills, A. C., Grant, D. M., Judah, M. R., & White, E. J. (2014). The influence of anticipatory processing on attentional biases in social anxiety. Behavior Therapy, 45(5), 720–729.
Mognon, A., Jovicich, J., Bruzzone, L., & Buiatti, M. (2011). ADJUST: An automatic EEG artifact detector based on the joint use of spatial and temporal features. Psychophysiology, 48(2), 229–240.
Monat, A. (1976). Temporal uncertainty, anticipation time, and cognitive coping under threat. Journal of Human Stress, 2(2), 32–43.
Norton, A. R., & Abbott, M. J. (2016). Self-focused cognition in social anxiety: A review of the theoretical and empirical literature. Behaviour Change, 33(1), 44–64.
Papageorgiou, C., & Wells, A. (2002). Effects of heart rate information on anxiety, perspective taking, and performance in high and low social-evaluative anxiety. Behavior Therapy, 33(2), 181–199.
Poli, S., Sarlo, M., Bortoletto, M., Buodo, G., & Palomba, D. (2007). Stimulus-preceding negativity and heart rate changes in anticipation of affective pictures. International Journal of Psychophysiology, 65(1), 32–39.
Sabatinelli, D., Bradley, M. M., & Lang, P. J. (2001). Affective startle modulation in anticipation and perception. Psychophysiology, 38(4), 719–722.
Schupp, H. T., Flaisch, T., Stockburger, J., & Junghöfer, M. (2006). Emotion and attention: Event-related brain potential studies. Progress in Brain Research, 156, 31–51.
Sege, C. T., Bradley, M. M., & Lang, P. J. (2014). Startle modulation during emotional anticipation and perception. Psychophysiology, 51, 977–981.
Sege, C. T., Bradley, M. M., & Lang, P. J. (2015). Prediction and perception: Defensive startle modulation. Psychophysiology, 52(12), 1664–1668.
Sege, C. T., Bradley, M. M., Weymar, M., & Lang, P. J. (2017). A direct comparison of appetitive and aversive anticipation: Overlapping and distinct neural activation. Behavioural Brain Research, 326, 96–102.
Sege, C. T., Bradley, M. M., & Lang, P. J. (2018). Avoidance and escape: Defensive reactivity and trait anxiety. Behaviour Research and Therapy, 104, 62–68.
Van Boxtel, G. J., & Böcker, K. B. (2004). Cortical measures of anticipation. Journal of Psychophysiology, 18(2/3), 61–76.
Wangelin, B. C., Löw, A., McTeague, L. M., Bradley, M. M., & Lang, P. J. (2011). Aversive picture processing: Effects of a concurrent task on sustained defensive system engagement. Psychophysiology, 48(1), 112–116.
Wells, A., & Papageorgiou, C. (2001). Social phobic interoception: Effects of bodily information on anxiety, beliefs and self-processing. Behaviour Research and Therapy, 39(1), 1–11.
White, E. J., & Grant, D. M. (2017). Electrocortical consequences of image processing: The influence of working memory load and worry. Psychiatry Research: Neuroimaging, 261, 1–8.
Wild, J., Clark, D. M., Ehlers, A., & McManus, F. (2008). Perception of arousal in social anxiety: Effects of false feedback during a social interaction. Journal of Behavior Therapy and Experimental Psychiatry, 39(2), 102–116.
Funding
The research results discussed in this publication were made possible in total or in part by funding through the award for project number HR16-023, from the Oklahoma Center for the Advancement of Science and Technology.
[Redacted for peer review] receives funding support from the National Institute on Minority Health and Health Disparities (NIMHD) under award number: [Redacted for peer review].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Consent to publish aggregate data was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 221 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kraft, J.D., Nagel, K.M., Deros, D.E. et al. The influence of self-focused attention on emotional picture processing: An ERP study. Cogn Affect Behav Neurosci 23, 162–170 (2023). https://doi.org/10.3758/s13415-022-01043-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/s13415-022-01043-7