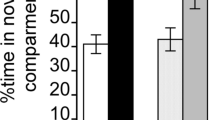
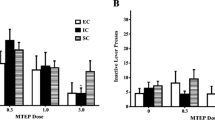
Abstract
The nucleus accumbens and its related neural circuitry are known to be involved in mediating drug reward. The present brief review provides evidence that manipulation of the stimulus environment may alter the behavioral effects of drugs of abuse by altering accumbal circuitry. In experiments conducted in our laboratory, rats were raised in either an enriched condition (EC) or an impoverished condition (IC) and then were challenged with acute intravenous amphetamine. EC rats were more sensitive than IC rats to the locomotor stimulant effect of amphetamine, as well as to the accumbal dopamine release measured by in vivo microdialysis. No environment-induced difference in [3H]-amphetamine pharmacokinetics was observed. In vitro neurochemical experiments also indicated that accumbal tissue slices isolated from EC and IC rats did not differ in dopamine levels, electrically evoked dopamine release, or dopamine autoreceptor sensitivity. Although the kinetic parameters for [3H]-dopamine uptake into striatal synaptosomes were not different between EC and IC rats, the transport inhibitor GBR 12935 was more potent in inhibiting uptake in EC rats than in IC rats. The latter results suggest that dopamine transporter proteins in the nucleus accumbens may be an important target for future studies aimed at identifying the cellular mechanism responsible for the environmentinduced difference in the behavioral response to amphetamine.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahmed, S. H., Stinus, L., Le Moal, M., & Cador, M. (1995). Social deprivation enhances the vulnerability of male Wistar rats to stressorand amphetamine-induced behavioral sensitization. Psychopharmacology,117, 116–124.
Bardo, M. T., Bowling, S. L., Rowlett, J. K., Manderscheid, P., Buxton, S. T., & Dwoskin, L. P. (1995). Environmental enrichment attenuates locomotor sensitization, but not in vitro dopamine release, induced by amphetamine. Pharmacology, Biochemistry & Behavior, 51, 397–405.
Bardo, M. T., & Hammer, R. P. (1991). Autoradiographic localization of dopamine D1 and D2 receptors in rat nucleus accumbens: Resistance to differential rearing conditions. Neuroscience, 45, 281–290.
Bennett, E. L., Diamond, M. C., Krech, D., & Rosenzweig, M. R. (1964). Chemical and anatomical plasticity of brain. Science, 164, 610–619.
Bennett, E. L., Rosenzweig, M. R., & Diamond, M. C. (1970). Time courses of effects of differential experience on brain measures and behavior of rats. In W. L. Byrne (Ed.), Molecular approaches to learning and memory (pp. 55–89). New York: Academic Press.
Black, J. E., Sirevaag, A. M., Wallace, C. S., Savin, M. H., & Greenough, W. T. (1989). Effects of complex experience on somatic growth and organ development in rats. Developmental Psychobiology, 22,727–752.
Bowling, S. L., & Bardo, M. T. (1994). Locomotor and rewarding effects of amphetamine in enriched, social, and isolate reared rats. Pharmacology, Biochemistry & Behavior, 48,459–464.
Bowling, S. L., Rowlett, J. K., & Bardo, M. T. (1993). The effect of environmental enrichment on amphetamine-stimulated locomotor activity, dopamine synthesis and dopamine release. Neuropharmacology, 32, 885–893.
Comery, T. A., Stamoudis, C.X., Irwin, S. A., & Greenough, W. T. (1996). Increased density of multiple-head dendritic spines on medium-sized spiny neurons of the striatum in rats reared in a complex environment. Neurobiology of Learning & Memory, 66, 93–96.
Crabbe, J. C., Belknap, J. K., & Buck, K. J. (1994, June). Genetic animal models of alcohol and drug abuse. Science, 264, 1715–1723.
Diamond, M. C., Law, F., Rhodes, H., Linder, B., Rosenzweig, M. R., Krech, D., & Bennett, E. L. (1966). Increases in cortical depth and glia numbers in rats subjected to enriched environments. Journal of Comparative Neurology, 128, 117–126.
Elmer, G. I., Brockington, A., Gorelick, D. A., Carrol, F. L., Rice, K. C., Matecka, D., Goldberg, S. R., & Rothman, R. B. (1996). Cocaine cross-sensitization to dopamine uptake inhibitors: Unique effects of GBR12909. Pharmacology, Biochemistry & Behavior, 53, 911–918.
Fiala, B., Snow, F. M., & Greenough, W. T. (1977). “Impoverished” rats weigh more than “enriched” rats because they eat more. Developmental Psychobiology, 10, 537–541.
Fowler, S. C., Johnson, J. S., Kallman, M. J., Liou, J. R., Wilson, M. C., & Hikal, A. H. (1993). In a drug discrimination procedure isolation-reared rats generalized to lower doses of cocaine and amphetamine than rats reared in an enriched environment. Psychopharmacology, 110, 115–118.
Freeman, B. J., & Ray, O. S. (1972). Strain, sex, and environmental effects on appetitively and aversively motivated learning tasks. Developmental Psychobiology, 5, 101–109.
Fuchs, J. L., Montemayor, M., & Greenough, W. T. (1990). Effect of environmental complexity on size of the superior colliculus. Behavioral & Neural Biology, 54, 198–203.
Gerhardt, G. A., Dwoskin, L. P., & Zahniser, N. R. (1989). Outflow and overflow of picogram level of endogenous dopamine and DOPAC from rat striatal slices: Improved methodology for studies of stimulus-evoked release and metabolism. Journal of Neuroscience Methods, 26, 217–227.
Gonzalez-Lima, F., Ferchmin, P. A., Eterovic, V. A., & Gonzalez-Lima, E. M. (1994). Metabolic activation of the brain of young rats after exposure to environmental complexity. Developmental Psychobiology, 27, 343–351.
Greenough, W. T., Madden, T. C., & Fleischman, T. B. (1972). Effects of isolation, daily handling, and enriched rearing on maze learning. Psychonomic Science, 27, 279–280.
Hall, F. S., Wilkinson, L. S., Humby, T., Inglis, W., Kendall, D. A., Marsden, C. A., & Robbins, T. W. (1998). Isolation rearing in rats: Pre- and postsynaptic changes in striatal dopaminergic systems. Pharmacology, Biochemistry & Behavior, 59, 859–872.
Hill, S. Y., & Powell, B. J. (1976). Cocaine and morphine self-administration: Effects of differential rearing. Pharmacology, Biochemistry & Behavior, 5, 701–704.
Jones, G. H., Marsden, C. A., & Robbins, T. A. (1990). Increased sensitivity to amphetamine and reward-related stimuli following social isolation in rats: Possible disruption of dopamine-dependent mechanisms of the nucleus accumbens. Psychopharmacology, 102, 364–372.
Kuhn, C. M., Schanberg, S. M., & Breese, G. R. (1978). Metabolism of amphetamine by rat brain tissue. Biochemical Pharmacology, 27, 343–351.
Liang, N. Y., & Rutledge, C. O. (1982). Comparison of the release of [3H]dopamine from isolated corpus striatum by amphetamine, fenfluramine and unlabeled dopamine. Biochemical Pharmacology, 31, 983–992.
Lore, R. K., & Levowitz, A. (1966). Differential rearing and free versus forced exploration. Psychonomic Science, 5, 421–422.
Masserano, J. M., Venable, D., & Wyatt, R. J. (1994). Effects of chronic cocaine administration on [3H]dopamine uptake in the nucleus accumbens, striatum and frontal cortex of rats. Journal of Pharmacology & Experimental Therapeutics, 270, 133–141.
Melega, W. P., Williams, A. E., Schmitz, D. A., DiStefano, E. W., & Cho, A. K. (1995). Pharmacokinetic and pharmacodynamic analysis of the actions of d-amphetamine and d-methamphetamine on the dopamine terminal. Journal of Pharmacology & Experimental Therapeutics, 272, 90–96.
Olsson, T., Mohammed, A. H., Donaldson, L. F., Henriksson, B. G., & Seckl, J. R. (1994). Glucorticoid receptor and NGFI-A gene expression are induced in the hippocampus after environmental enrichment in adult rats. Molecular Brain Research, 23, 349–353.
Rebec, G. V., Christensen, J. R. C., Guerra, C., & Bardo, M. T. (1997). Regional and temporal differences in real-time dopamine efflux in the nucleus accumbens during free-choice novelty. Brain Research, 776,61–67.
Reige, W. H., & Morimoto, H. (1970). Effects of chronic stress and differential environments upon brain weights and biogenic amine levels in rats. Journal of Comparative & Physiological Psychology, 71, 396–404.
Reith, M. E., Coffey, L. L., Xu, C., & Chen, N. H. (1994). GBR 12909 and 12935 block dopamine uptake into brain synaptic vesicles as well as nerve endings. European Journal of Pharmacology, 253,175–178.
Renner, M. J., & Rosenzweig, M. R. (1986). Object interactions in juvenile rats (Rattus norvegicus): Effects of different experiential histories. Journal of Comparative Psychology, 100, 229–236.
Renner, M. J., & Rosenzweig, M. R. (1987). Enriched and impoverished environments: Effects on brain and behavior. New York: Springer-Verlag.
Rosenzweig, M. R., & Bennett, E. L. (1972). Cerebral changes in rats exposed individually to an enriched environment. Journal of Comparative & Physiological Psychology, 80, 304–313.
Rosenzweig, M. R., Krech, D., Bennett, E. L., & Diamond, M. C. (1962). Effects of environmental complexity and training on brain chemistry and anatomy: A replication and extension. Journal of Comparative & Physiological Psychology, 55, 429–437.
Sirevaag, A. M., & Greenough, W. T. (1988). A multivariate statistical summary of synaptic plasticity measures in rats exposed to complex, social and individual environments. Brain Research, 441, 386–392.
Smith, J. K., Neill, J. C., & Costall, B. (1997). Post-weaning housing conditions influence the behavioral effects of cocaine and d-amphetamine. Psychopharmacology, 131, 23–33.
Stahle, L., Collin, A. K., & Ungerstedt, U. (1990). Effects of halothane anesthesia on extracellular levels of dopamine, dihydroxy-phenylacetic acid, homovanillic acid and 5-hydroxyindoleacetic acid in rat striatum: A microdialysis study. Naunyn-Schmiedeberg’s Archives of Pharmacology, 342, 136–140.
Sulzer, D., Chen, T. K., Lau, Y. Y., Kristensen, H., Rayport, S., & Ewing, A. (1995). Amphetamine redistributes dopamine from synaptic vesicles to the cytosol and promotes reverse transport. Journal of Neuroscience, 15,4102–4108.
Takahashi, N., Miner, L. L., Sora, I., Ujike, H., Revay, R. S., Kostic, V., Jackson-Lewis, V., Przedborski, S., & Uhl, G. R. (1997). VMAT2 knockout mice: Heterozygotes display reduced amphetamine-conditioned reward, enhanced amphetamine locomotion, and enhanced MPTP toxicity. Proceedings of the National Academy of Sciences, 94, 9938–9943.
Wallace, C. S., Kilman, V. L., Withers, G. S., & Greenough, W. T. (1992). Increases in dendritic length in occipital cortex after 4 days of differential housing in weanling rats. Behavioral & Neural Biology, 58, 64–68.
Widman, D. R., & Rosellini, R. A. (1990). Restricted daily exposure to environmental enrichment increases the diversity of exploration. Physiology & Behavior, 47, 57–62.
Winterfeld, K. T., Teuchert-Noodt, G., & Dawirs, R. R. (1998). Social environment alters both ontogeny of dopamine innervation of the medial prefrontal cortex and maturation of working memory in gerbils (Meriones unguiculatus). Journal of Neuroscience Research, 52,201–209.
Zimmerberg, B., & Brett, M. B. (1992). Effects of early environmental experience on self-administration of amphetamine and barbital. Psychopharmacology, 106, 474–478.
Author information
Authors and Affiliations
Corresponding author
Additional information
We gratefully acknowledge the expert technical assistance of Susan T. Buxton, Susan Moore, Amy Williamson, and Melinda Marion in conducting the experiments. Helpful discussion with Peter Crooks on the pharmacokinetic experiment is acknowledged.
The research was supported by USPHS Grants DA05312 and DA06924. W.B.S. and L.P.D. are in the Division of Pharmaceutical Sciences in the College of Pharmacy.
Rights and permissions
About this article
Cite this article
Bardo, M.T., Valone, J.M., Robinet, P.M. et al. Environmental enrichment enhances the stimulant effect of intravenous amphetamine: Search for a cellular mechanism in the nucleus accumbens. Psychobiology 27, 292–299 (1999). https://doi.org/10.3758/BF03332123
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3758/BF03332123