Abstract
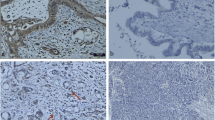
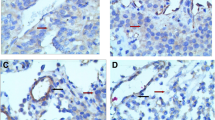
FSH receptor (FSHR) expression is restricted to gonads, where it drives FSH-dependent cell differentiation; in addition, FSHR plays an important role in the regulation of ovarian angiogenesis. Recently, FHSR expression has been shown in blood vessels of various tumors. However, pancreatic neuroendocrine tumors (p-NET), which have high-degree blood supply, were not included in that study. The aim of this study was to evaluate FSHR expression in p-NET. FSHR expression was evaluated in tumor samples from 30 patients with p-NET by immunohistochemistry and Western blot; fluorescence microscopy was used to localize FSHR in specific cells from tissue samples. von Willebrand factor (vWF) and chromograninA (chrA) was used as blood vessel and NET cells marker, respectively, to co-localize FSHR. FSHR expression was detected in all p-NET by immunohistochemistry. Western blot confirmed FSHR expression on p-NET although different FSHR isoforms, ranging from 240 kD to 55 kD were found in the samples studied. Surprisingly, FSHR co-localized with chrA but not with vWF, suggesting that neoplastic cells of neuroendocrine origin rather than blood vessels expressed FSHR. No relationship was found between degree of FSHR expression and histology of p-NET. FSHR may be aberrantly expressed in neoplastic cells from p-NET and not in tumor blood vessels; however, its biological significance as well as its clinical relevance remains to be elucidated.
Similar content being viewed by others
References
Camp TA, Rahal JO, Mayo KE. Cellular localization and hormonal regulation of follicle-stimulating hormone and luteininzing hormone receptor messenger RNAs in the rat ovary. Mol Endocrinol 1991, 5: 1405–17.
Sprengel R, Braun T, Nikolics K, Segaloff DL, Seeburg PH. The testicular receptor for follicle stimulating hormone: structure and functional expression of cloned cDNA. Mol Endocrinol 1990, 4: 525–30.
Kelton CA, Cheng SV, Nugent NP, et al. The cloning of the human follicle stimulating hormone receptor and its expression in COS-7, CHO, and Y-1 cells. Mol Cell Endocrinol 1992, 89: 141–51.
Reisinger K, Baal N, McKinnon T, Münstedt K, Zygmunt. The gonadotropins: tissue-specific angiogenic factors? Mol Cell Endocrinol 2007, 269: 65–80.
Kuo SW, Ke FC, Chang GD, Lee MT, Hwang JJ. Potential role of follicle-stimulating hormone (FSH) and transforming growth factor (TGFβ1) in the regulation of ovarian angiogenesis. J Cell Physiol 2011, 226: 1608–19.
Vu Hai MT, Lescop P, Loosfelt H, Ghinea N. Receptor-mediated transcytosis of follicle stimulating hormone through the rat testicular microvasculature. Biol Cell 2004, 96: 133–44.
Sun L, Peng Y, Sharrow AC, et al. FSH directly regulates bone mass. Cell 2006, 125: 247–60.
Robinson LJ, Tourkova I, Wanga Y, et al. FSH-receptor isoforms and FHS-dependent gene transcription in human monocytes and osteoclasts. Biochem Biophys Res Comm 2010, 394: 12–7.
Radu A, Pichon C, Camparo P, et al. Expression of follicle-stimulating hormone receptor in tumour blood vessels. N Engl J Med 2010, 363: 1621–30.
Klöppel G, Couvelard A, Perren A, et al. ENETS consensus guidelines for the standards care in neuroendocrine tumours: towards a standardized approach to the diagnosis of gastroenteropancreatic neuroendocrine tumours and their prognostic stratification. Neuroendocrinology 2009, 90: 162–6.
Bogazzi F, Russo D, Raggi F, et al. Transgenic Mice Overexpression Growth Hormone (GH) Have Reduced or Increased Cardiac Apoptosis through Activation of Multiple GH-Dependent or -Independent Cell Death Pathways. Endocrinology 2008, 149: 5758–69.
Rosa P, Gerdes HH. The granin protein family: markers for neuroendocrine cells and tools for the diagnosis of neuroendocrine tumours. J Endocrinol Invest 1994, 17: 207–25.
Au-Yeung KW, Woo CWH, Sung FL, Yip JCW, Siow YL, Karmin O. Hyperhomocysteinemia Activates Nuclear Factor-κB in Endothelial Cells via Oxidative Stress. Circ Res 2004, 94: 28–36.
Goodpaster T, Legesse-Miller A, Hameed MR, Aisner SC, Randolph-Habecker J, Coller H. An Immunohistochemical Method for Identifying Fibroblast in formalin-fixed, paraffin-embedded Tissue. J Histochem Cytochem 2008, 56: 347–58.
Dattatreyamurty B, Zhang B-S, Reichert LE Jr. Purification of follitropin receptor from bovine calf testes. J Biol Chem 1990, 265: 5494–503.
George JW, Dille EA, Heckert LL. Current concepts of follicle-stimulating hormone receptor gene regulation. Biol Reproduct 2011, 84: 7–17.
Ben-Josef E, Yang SY, Ji TH, et al. Hormone-refractory prostate cancer cells express functional follicle-stimulating hormone receptor (FSHR). J Urol 1999, 161: 970–6.
Mariani S, Salvatori L, Basciani S, et al. Expression and cellular localization of follicle stimulating hormone receptor in normal human prostate, benign prostatic hyperplasia and prostate cancer. J Urol 2006, 175: 2072–7.
Zheng W, Magid MS, Kramer EE, Chen YT. Follicle stimulating hormone receptor is espresse in human ovarian suface epithelium and fallo pian tube. Am J Pathol 1996, 148: 47–53.
Grotzinger C. Tumor biology of gastroenteropancreatic neuroendocrine tumours. Neuroendocrinology 2004, 80 (Suppl 1): 8–11.
Vannier B, Loosfelt H, Meduri G, Pichon C, Milgrom E. Anti-human FSH receptor monoclonal antibodies: immunochemical and immunocytochemical characterization of the receptor. Biochemistry 1996, 35: 1358–66.
Rajapaksha WR, Robertson L, O’Shaughnessy PJ. Expression of follicle-stimulating hormone-receptor mRNA alternate transcripts in bovine granulosa cells during luteinization in vivo. Mol Cell Endocrinol 1996, 120: 25–30.
Rannikki AS, Zhang FP, Huhtaniemi IT. Ontogeny of follicle-stimulating hormone receptor gene expression in the rat testis and ovary. Mol Cell Endocrinol 1995, 107: 199–208.
Richards JS, Ireland JJ, Rao MC, Bernath GA, Midgley AR Jr, Reichert LE. Ovarian follicular development in the rat: hormone receptor regulation by estradiol, follicle-stimulating hormone and luteinizing hormone. Endocrinology 1976, 99: 1562–70.
Sokka T, Huhtaniemi I. Ontogeny of gonadotrophin receptors and gonadotropin-stimulated cyclic AMP production in the neonatal rat ovary. J Endocrinol 1990, 127: 297–303.
Powers S, Mu D. Genetic similarities between organogenesis and tumourigenesis on the lung. Cell Cycle 2008, 7: 200–4.
Arsic D, Beasley SW, Sullivan MJ. Switched-on Sonic hedgehog: a gene whose activity extends beyond fetal development-to oncogenesis. J Paediatr Child Health 2007, 43: 421–3.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sardella, C., Russo, D., Raggi, F. et al. Ectopic expression of FSH receptor isoforms in neoplastic but not in endothelial cells from pancreatic neuroendocrine tumors. J Endocrinol Invest 36, 174–179 (2013). https://doi.org/10.3275/8472
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3275/8472