Abstract
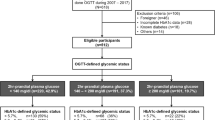
Background: Glycated hemoglobin (HbA1c) 6.5% has recently been recommended by the World Health Organization (WHO) and the American Diabetes Association (ADA) as an alternative diagnostic criterion for diabetes mellitus (DM). Aim: To evaluate HbA1c as an alternative to oral glucose tolerance test (OGTT) for diagnosis of DM and pre-diabetes and to find the optimal HbA1c cut-off points for DM and pre-diabetes in our population. Subjects and methods: The subjects were recruited from the Tromsø Study, performed for the 6th time in 2007–2008 with 12,984 participants. All subjects with HbA1c in the range 5.8–6.9% and a random sample of subjects with levels 5.3–5.7% were invited to an OGTT. Results: Among 3476 subjects who completed the OGTT, 199 were diagnosed with DM. The best sensitivity (69.8%) and specificity (81.8%) were found at HbA1c 6.2%. For HbA1c 6.5% we found a sensitivity of 34.7% and specificity 97.1%. The best cut-off points for impaired fasting glucose (no.=314) and impaired glucose tolerance (no.=404) were found at HbA1c 5.9% and 6.0%, respectively. Pre-diabetes detected only by OGTT was associated with worse metabolic characteristics than pre-diabetes detected only by HbA1c. Conclusions: The optimum HbA1c cutoff point for DM in our population was lower than that proposed by WHO and ADA. To establish more precisely the HbA1c levels predictive of micro- and macro-vascular complications, long-term prospective studies are needed. Population-specific optimum cut-off points may be necessary.
Similar content being viewed by others
References
Sacks DB. Carbohydrates. In: Burtis CA, Ashwood ER, Bruns DE, eds. Tietz Textbook of Clinical Chemistry and Molecular Diagnostics. St. Louis: Elsevier Saunders, 2006, 837–902.
Little RR, Sacks DB. HbA1c: how do we measure it and what does it mean? Curr Opin Endocrinol Diabetes Obes 2009, 16: 113–8.
Rahbar S, Blumenfeld O, Ranney HM. Studies of an unusual hemoglobin in patients with diabetes mellitus. Biochem Biophys Res Commun 1969, 36: 838–43.
Nathan DM, Turgeon H, Regan S. Relationship between glycated haemoglobin levels and mean glucose levels overtime. Diabetologia 2007, 50: 2239–44.
World Health Organization and International Diabetes Federation. Definition and diagnosis of diabetes mellitus and intermediate hyperglycemia: report of a WHO/IDF consultation. Geneva: World Health Organization, 2006.
Diagnosis and classification of diabetes mellitus. Diabetes Care 2010, 33(Suppl 1): S62–9.
International Expert Committee. International Expert Committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care 2009, 32: 1327–34.
World Health Organization. Use of glycated haemoglobin (HbA1c) in the diagnosis of diabetes mellitus: abbreviated report of a WHO consultation. World Health Organization: Geneva, 2011.
Sacks DB. A1c versus glucose testing: a comparison. Diabetes Care 2011, 34: 518–23.
Nitin S. HbA1c and factors other than diabetes mellitus affecting it. Singapore Med J 2010, 51: 616–22.
Roberts WL, De BK, Brown D et al. Effects of hemoglobin C and S traits on eight glycohemoglobin methods. Clin Chem 2002, 48: 383–5.
Sacks DB, Bruns DE, Goldstein DE, Maclaren NK, McDonald JM, Parrott M. Guidelines and recommendations for laboratory analysis in the diagnosis and management of diabetes mellitus. Clin Chem 2002, 48: 436–72.
Little RR, Rohlfing CL, Sacks DB; National Glycohemoglobin Standardization Program (NGSP) Steering Committee. Status of hemoglobin A1c measurement and goals for improvement: from chaos to order for improving diabetes care. Clin Chem 2011, 57: 205–14.
Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 1997, 20: 1183–97.
McCance DR, Hanson RL, Charles MA, et al. Comparison of tests for glycated haemoglobin and fasting and two hour plasma glucose concentrations as diagnostic methods for diabetes. BMJ 1994, 308: 1323–8.
Engelgau MM, Thompson TJ, Herman, et al. Comparison of fasting and 2-hour glucose and HbA1c levels for diagnosing diabetes. Diagnostic criteria and performance revisited. Diabetes Care 1997, 20: 785–91.
Colagiuri S, Lee CM, Wong TY, Balkau B, Shaw JE, Borch-Johnsen K; DETECT-2 Collaboration Writing Group. Glycemic thresholds for diabetes-specific retinopathy: implications for diagnostic criteria for diabetes. Diabetes Care 2011, 34: 145–50.
Jacobsen BK, Eggen AE, Mathiesen EB, Wilsgaard T, Njølstad I. Cohort profile: The Tromso Study. Int J Epidemiol 2012, 41: 961–7.
Kamycheva E, Jorde R, Figenschau Y, Haug E. Insulin sensitivity in subjects with secondary hyperparathyroidism and the effect of a low serum 25-hydroxyvitamin D level on insulin sensitivity. J Endocrinol Invest 2007, 30: 126–32.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28: 412–9.
Katz A, Nambi SS, Mather K, et al. Quantitative insulin sensitivity check index: a simple, accurate method for assessing insulin sensitivity in humans. J Clin Endocrinol Metab 2000, 85: 2402–10.
Colagiuri S. Epidemiology of prediabetes. Med Clin North Am 2011, 95: 299–307, vii.
Mann DM, Carson AP, Shimbo D, Fonseca V, Fox CS, Muntner P. Impact of A1c screening criterion on the diagnosis of pre-diabetes among U.S. adults. Diabetes Care 2010, 33: 2190–5.
Lorenzo C, Wagenknecht LE, Hanley AJ, Rewers MJ, Karter AJ, Haffner SM. A1c between 5.7 and 6.4% as a marker for identifying pre-diabetes, insulin sensitivity and secretion, and cardiovascular risk factors: the Insulin Resistance Atherosclerosis Study (IRAS). Diabetes Care 2010, 33: 2104–9.
Miyazaki M, Kubo M, Kiyohara Y, et al; Hisayama study. Comparison of diagnostic methods for diabetes mellitus based on prevalence of retinopathy in a Japanese population: the Hisayama Study. Diabetologia 2004, 47: 1411–5.
Tapp RJ, Zimmet PZ, Harper CA, et al; AusDiab Study Group. Diagnostic thresholds for diabetes: the association of retinopathy and albuminuria with glycaemia. Diabetes Res Clin Pract 2006, 73: 315–21.
Tay TL, Foo JP, Tan E, et al. HbA1c may not be a sensitive determinant of diabetic status in the elderly. Diabetes Res Clin Pract 2011, 92: e31–3.
van Leiden HA, Dekker JM, Moll AC, et al. Risk factors for incident retinopathy in a diabetic and nondiabetic population: the Hoorn study. Arch Ophthalmol 2003, 121: 245–51.
Massin P, Lange C, Tichet J, et al; DESIR (Data From an Epidemiological Study on the Insulin Resistance Syndrome) Study Group. Hemoglobin A1c and Fasting Plasma Glucose Levels as Predictors of Retinopathy at 10 Years: The French DESIR Study. Arch Ophthalmol 2011, 129: 188–95.
Bonora E, Kiechl S, Mayr A, et al. High-normal HbA1c is a strong predictor of type 2 diabetes in the general population. Diabetes Care 2011, 34: 1038–40.
Perry RC, Shankar RR, Fineberg N, McGill J, Baron AD; Early Diabetes Intervention Program (EDIP). HbA1c measurement improves the detection of type 2 diabetes in high-risk individuals with nondiagnostic levels of fasting plasma glucose: the Early Diabetes Intervention Program (EDIP). Diabetes Care 2001, 24: 465–71.
Inoue K, Matsumoto M, Akimoto K. Fasting plasma glucose and HbA1c as risk factors for Type 2 diabetes. Diabet Med 2008, 25: 1157–63.
Cosson E, Hamo-Tchatchouang E, Banu I, et al. A large proportion of prediabetes and diabetes goes undiagnosed when only fasting plasma glucose and/or HbA1c are measured in overweight or obese patients. Diabetes Metab 2010, 36: 312–8.
Olson DE, Rhee MK, Herrick K, Ziemer DC, Twombly JG, Phillips LS. Screening for diabetes and pre-diabetes with proposed A1C-based diagnostic criteria. Diabetes Care 2010, 33: 2184–9.
Cosson E, Nguyen MT, Hamo-Tchatchouang E, et al. What would be the outcome if the American Diabetes Association recommendations of 2010 had been followed in our practice in 1998–2006? Diabet Med 2011, 28: 567–74.
Pani LN, Korenda L, Meigs JB, et al. Effect of aging on A1c levels in individuals without diabetes: evidence from the Framingham Offspring Study and the National Health and Nutrition Examination Survey 2001–2004. Diabetes Care 2008, 31: 1991–6.
Rathmann W, Kowall B, Tamayo T, et al. Hemoglobin A1c and glucose criteria identify different subjects as having type 2 diabetes in middle-aged and older populations: The KORA S4/F4 Study. Ann Med 2012, 44: 170–7.
Hawkins R. Differences in HbA1c between Caucasians, Chinese, Indians, Malays and Eurasians. Clin Chim Acta 2011, 412: 1167.
Herman WH, Ma Y, Uwaifo G, et al; Diabetes Prevention Program Research Group. Differences in A1c by race and ethnicity among patients with impaired glucose tolerance in the Diabetes Prevention Program. Diabetes Care 2007, 30: 2453–7.
Davidson MB, Schriger DL. Effect of age and race/ethnicity on HbA1c levels in people without known diabetes mellitus: implications for the diagnosis of diabetes. Diabetes Res Clin Pract 2010, 87: 415–21.
Saltevo JT, Kautiainen H, Niskanen L, et al. Ageing and associations of fasting plasma glucose and 2h plasma glucose with HbA(1c) in apparently healthy population. “FIN-D2D” study. Diabetes Res Clin Pract 2011, 93: 344–9.
Gomyo M, Sakane N, Kamae I, et al. Effects of sex, age and BMI on screening tests for impaired glucose tolerance. Diabetes Res Clin Pract 2004, 64: 129–36.
Selvin E, Crainiceanu CM, Brancati FL, Coresh J. Short-term variability in measures of glycemia and implications for the classification of diabetes. Arch Intern Med 2007, 167: 1545–51.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hutchinson, M.S., Joakimsen, R.M., Njølstad, I. et al. Glycated hemoglobin in diagnosis of diabetes mellitus and pre-diabetes; validation by oral glucose tolerance test. The Tromsø OGTT Study. J Endocrinol Invest 35, 835–840 (2012). https://doi.org/10.3275/8191
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3275/8191