Abstract
Aims
Insulin resistance has been implicated as a risk factor for metabolic disorders. Due to the clinical interest in insulin resistance, it is of great importance to develop a simple test that can be used in routine clinical settings for identifying insulin-resistant individuals in advance. Glycated hemoglobin (HbA1c) has been considered as a potentially good indicator of overall glycemic exposure and likely risk for long-term complications. Therefore, this study is designed to investigate the importance of HbA1c in predicting insulin resistance among apparently healthy at-risk German populations.
Methods
The association between HbA1c and several surrogate markers of insulin resistance/sensitivity based on plasma glucose and insulin concentrations was analyzed by bivariate correlation along with multivariate linear regression and receiver-operating characteristic curve analysis among normal (NGT) and impaired glucose tolerance (IGT) individuals (n = 3578 aged >18 years) in Germany.
Results
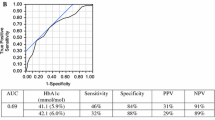
Spearman’s correlation coefficients showed that HbA1c had a significant association with insulin resistance/sensitivity markers in both NGT and IGT individuals and that this was stronger in the younger age group (<50 years) (rho = 0.410 with HOMA-IR, rho = −0.379 with the Matsuda index). Moreover, the ROC curve also showed that the HbA1c occupied a significant area under the curve (0.731 with 95% CI 0.661–0.801) and that the cutoff point for estimating insulin resistance corresponded to 5.8% (40 mmol/mol) with 34% sensitivity and 80% specificity.
Conclusion
From this study, it could be concluded that HbA1c may be a clinically useful and simple index for predicting the concomitant presence of insulin resistance and dysglycemia among apparently healthy, young (<50 years) German populations.

Similar content being viewed by others
References
American Diabetes Association (2007) Diagnosis and classification of diabetes mellitus. Diabetes Care 30(suppl 1):S42–S47
American Diabetes Association (2010) Diagnosis and classification of diabetes mellitus. Diabetes Care 33(suppl 1):S62–S69
Bergman RN, Phillips LS, Cobelli C (1981) Physiologic evaluation of factors controlling glucose tolerance in man: measurement of insulin sensitivity and beta-cell glucose sensitivity from the response to intravenous glucose. J Clin Invest 68:1456–1467
Bonora E, Calcaterra F, Lombardi S, Bonfante N, Formentini G, Bonadonna RC, Muggeo M (2001) Plasma glucose levels throughout the day and HbA(1c) interrelationships in type 2 diabetes: implications for treatment and monitoring of metabolic control. Diabetes Care 24:2023–2029
Brehm A, Pfeiler G, Pacini G, Vierhapper H, Roden M (2004) Relationship between serumlipoprotein ratios and insulin resistance in obesity. Clin Chem 50:2316–2322
Carson AP, Reynolds K, Fonseca VA, Muntner P (2010) Comparison of A1C and fasting glucose criteria to diagnose diabetes among US adults. Diabetes Care 33:95–97
Cavaghan MK, Ehrmann DA, Polonsky KS (2000) Interactions between insulin resistance and insulin secretion in the development of glucose intolerance. J Clin Invest 106:329–333
DeFronzo RA (1988) (1987) Lilly lecture. The triumvirate: beta-cell, muscle, liver. A collusion responsible for NIDDM. Diabetes 37:667–687
DeFronzo RA, Tobin JD, Andres R (1979) Glucose clamp technique: a method for quantifying insulin secretion and resistance. Am J Physiol 237:E214–E223
Ferrannini E, Buzzigoli G, Bonadonna R, Giorico MA, Oleggini M, Graziadei L, Pedrinelli R, Brandi L, Bevilacqua S (1987) Insulin resistance in essential hypertension. N Engl J Med 317:350–357
Ferrara CM, Goldberg AP (2001) Limited value of the homeostasis model assessment to predict insulin resistance in older men with impaired glucose tolerance. Diabetes Care 24:245–249
Grundy SM (2012) Pre-diabetes, metabolic syndrome, and cardiovascular risk. J Am Coll Cardiol 59:635–643
Gulli G, Ferrannini E, Stern M, Haffner S, DeFronzo RA (1992) The metabolic profile of NIDDM is fully established in glucose-tolerant offspring of two Mexican-American NIDDM parents. Diabetes 41:1575–1586
Herman WH, Ma Y, Uwaifo G, Haffner S, Kahn SE, Horton ES, Lachin JM, Montez MG, Brenneman T, Barrett-Connor E, Diabetes Prevention Program Research G (2007) Differences in A1C by race and ethnicity among patients with impaired glucose tolerance in the diabetes prevention program. Diabetes Care 30:2453–2457
Hosker JP, Matthews DR, Rudenski AS, Burnett MA, Darling P, Bown EG, Turner RC (1985) Continuous infusion of glucose with model assessment: measurement of insulin resistance and beta-cell function in man. Diabetologia 28:401–411
International Expert Committee (2009) International Expert committee report on the role of the A1C assay in the diagnosis of diabetes. Diabetes Care 32:1327–1334
Jeon JY, Ko SH, Kwon HS, Kim NH, Kim JH, Kim CS, Song KH, Won JC, Lim S, Choi SH, Jang MJ, Kim Y, Oh K, Kim DJ, Cha BY, Taskforce Team of Diabetes Fact Sheet of the Korean Diabetes A (2013) Prevalence of diabetes and prediabetes according to fasting plasma glucose and HbA1c. Diabetes & metabolism journal 37:349–357
Karhapaa P, Voutilainen E, Kovanen PT, Laakso M (1993) Insulin resistance in familial and nonfamilial hypercholesterolemia. Arterioscleresis and Thrombosis 13:41–47
Klein R, Klein BE, Moss SE, Davis MD, DeMets DL (1988) Glycosylated hemoglobin predicts the incidence and progression of diabetic retinopathy. J Am Med Assoc 260:2864–2871
Laakso M, Pyorala K, Voutilainen E, Marniemi J (1987) Plasma insulin and serum lipids and lipoproteins in middle-aged non- insulin dependent diabetic and non-diabetic subjects. Am J Epidemiol 125:611–621
Lee JM, Okumura MJ, Davis MM, Herman WH, Gurney JG (2006) Prevalence and determinants of insulin resistance among US adolescents: a population based study. Diabetes Care 29:2427–2432
Li C, Ford ES, Meng YX, Mokdad AH, Reaven GM (2008) Does the association of the triglyceride to high-density lipoprotein cholesterol ratio with fasting serum insulin differ by race/ethnicity? Cardiovasc Diabetol 7:1–9
Marcovina S, Bowsher RR, Miller WG, Staten M, Myers G, Caudill SP, Campbell SE, Steffes MW (2007) Standardization of insulin immunoassays: report of the American Diabetes Association workgroup. Clin Chem 53:711–716
Matsuda M, DeFronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing. Diabetes Care 22:1462–1470
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC (1985) Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28:412–419
McLaughlin T, Abbasi F, Cheal K, Chu J, Lamendola C, Reaven GM (2003) Use of metabolic markers to identify overweight individuals who are insulin resistant. Ann Intern Med 139:802–809
McLaughlin T, Reaven GM, Abbasi F, Lamendola C, Saad M, Waters D, Simon J, Krauss RM (2005) Is there a simple way to identify insulin resistant individuals at increased risk of cardiovascular disease? Am J Cardiol 96:399–404
Moran A, Jacobs DR, Steinberger J, Hong CP, Prineas R, Luepker R, Sinaiko AR (1999) Insulin resistance during puberty: results from clamp studies in 357 children. Diabetes 48:2039–2044
Moro E, Gallina P, Pais M, Cazzolato G, Alessandrini P, Bittolo-Bon Govindarajalu S (2003) Hypertriglyceridemia is associated with increased insulin resistance in subjects with normal glucose tolerance: evaluation in a large cohort of subjects assessed with the 1999 World Health Organization criteria for the classification of diabetes. Metabolism 52:616–619
Perkovic V, Verdon C, Ninomiya T, Barzi F, Cass A, Patel A, Jardine M, Gallagher M, Turnbull F, Chalmers J, Craig J, Huxley R (2008) The relationship between proteinuria and coronary risk: a systematic review and meta-analysis. PLoS Med 5:e207
Reaven GM (1988) Role of insulin resistance in human disease. Diabetes Care 37:1595–1607
Salazar MR, Carbajal HA, Espeche WG, Leiva Sisnieguez CE, Balbín E, Dulbecco CA, Aizpurúa M, Marillet AG, Reaven GM (2012) Relation among the plasma triglyceride/high-density lipoprotein cholesterol concentration ratio, insulin resistance, and associated cardio-metabolic risk factors in men and women. Am J Cardiol 109:1749–1753
Satchell SC, Tooke JE (2008) What is the mechanism of microalbuminuria in diabetes: a role for the glomerular endothelium? Diabetologia 51:714–725
Simental-Mendia LE, Rodriguez-Moran M, Guerrero-Romero F (2008) The product of fasting glucose and triglycerides as surrogate for identifying insulin resistance in apparently healthy subjects. Metab Syndr Relat Disord 6:299–304
Tapp RJ, Shaw JE, Zimmet PZ, Balkau B, Chadban SJ, Tonkin AM, Welborn TA, Atkins RC (2004) Albuminuria is evident in the early stages of diabetes onset: results from the Australian diabetes, obesity, and lifestyle study (AusDiab). American Journal of Kidney Diseases. The official journal of the National Kidney Foundation 44:792–798
Tuomilehto J, Lindstrom J, Eriksson JG, Valle TT, Hamalainen H, Ilanne-Parikka P, Keinanen-Kiukaanniemi S, Laakso M, Louheranta A, Rastas M, Salminen V, Uusitupa M, Finnish Diabetes Prevention Study G: (2001) Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. The New England Journal of Medicine; 344:1343–1350
Yeni-Komshian H, Carantoni M, Abbasi F, Reaven GM (2000) Relationship between several surrogate estimates of insulin resistance and quantification of insulin-mediated glucose disposal in 490 healthy nondiabetic volunteers. Diabetes Care 23:171–175
Ziemer DC, Kolm P, Weintraub WS, Vaccarino V, Rhee MK, Twombly JG, Narayan KM, Koch DD, Phillips LS (2010) Glucose-independent, black-white differences in hemoglobin A1c levels: a cross-sectional analysis of 2 studies. Ann Intern Med 152:770–777
Acknowledgments
The authors would like to thank Patrick Timpel for his careful reading of the manuscript.
Author information
Authors and Affiliations
Contributions
S.S. analyzed the data and wrote the manuscript. P.S. reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest statement
The authors state that there is no conflict of interest and they have nothing to declare.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institution, the Technical University of Dresden.
Informed consent
Written informed consent was obtained from all individuals according to the guidelines of the Institutional Review Board for Human Studies at the Technical University of Dresden.
Additional information
Main messages of the article
1. The aim of this study was to investigate the role of HbA1c in predicting insulin resistance among apparently healthy individuals.
2. The study shows that HbA1c has a significant association with insulin resistance markers among young IGT individuals.
3. The ROC curve also demonstrates that HbA1c occupied a significant area under the curve (0.731 with 95% CI 0.661–0.801). Hence, HbA1c may be considered as a clinically useful and simple index for predicting the concomitant presence of insulin resistance and dysglycemia among apparently healthy young German populations (<50 years).
Rights and permissions
About this article
Cite this article
Saha, S., Schwarz, P.E.H. Impact of glycated hemoglobin (HbA1c) on identifying insulin resistance among apparently healthy individuals. J Public Health 25, 505–512 (2017). https://doi.org/10.1007/s10389-017-0805-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10389-017-0805-4