Abstract
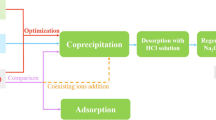
The removal of As, Sb, and Bi impurities from copper electrolyte is a primary objective of copper electrorefineries. The present experimental work demonstrates that the presence of Sb2O3 facilitates efficient and fast removal these impurities (with removal rates of 38.50, 98.50, and 99.00% for As, Sb, and Bi) through the formation of antimonate (AsSbO4/Sb2O4/BiSbO4), which plays a critical role in the self-purification of copper electrolyte. However, the antimonate which is a valuable metallurgical by-product contained high contents of As and Sb. The thermal decomposition of the antimonate was characterized by TG/DTA, a new method was proposed for recovering the target components, As, Sb, Bi, and to regenerate Sb2O3 with a two-stage roasting process under argon atmosphere. According to the results of XRD, SEM-EDS and ICP-MS, AsSbO4 decomposed during the first stage roasting at 800°C over 2 h, affording As with a recovery rate of 98.80%. During the second stage, decomposition of BiSbO4 and Sb2O4 at 1200°C over 2 h resulted in 99.01, 95.14% recovery rates for Sb, Bi.
Similar content being viewed by others
References
Wang, X.W., Study on the mechanism of the formation and action of arsenato and antimonic acid in copper electrorefining (Doctor’s Dissertation), Changsha: Central South Univ., 2003, pp. 17–18 (in Chinese).
Xiao, F.X., Cao, D., Mao, J.W., Shen, X.N., and Ren, F.Z., Role of Sb(V) in removal of As, Sb and Bi impurities from copper electrolyte, Trans. Nonferrous Met. Soc. China. 2014, vol. 24, no. 1, pp. 271–278.
Xiao, F.X., Cao, D., Mao, J.W., Shen, X.N., and Ren, F.Z., Role of Sb(III) in removal of As, Sb and Bi impurities from copper electrolyte, Int. J. Miner. Metal. Mater., 2013, vol. 20, no. 1, pp. 9–16.
Zhai, X.J., Heavy Metal Metallurgy, Beijing: Metallurgical Industry. 2013, p. 73 (in Chinese).
Krayuhin, S.A., Maltsev, G.I., Timofeev, K.L., and Naboychenko, S.S., Thermodynamic prediction of melting of copper-electrolyte slime, Rus. J. Non-Ferrous Met., 2016, vol. 57, no. 3, pp. 175–179.
Navarro, P., and Alguacil, F.J., Adsorption of antimony and arsenic from a copper electrorefining solution onto activated carbon, Hydrometallurgy. 2002, vol. 66, no. 1, pp. 101–105.
Wang, X.W., Chen, Q.Y., Yin, Z.P., Zhang, P.M., Long, Z.P., and Su, Z.F., Removal of impurities from copper electrolyte with adsorbent containing antimony, Hydrometallurgy. 2003, vol. 69, nos. 1–3, pp. 39–44.
Wang, X.W., Xiao, B.R. and Zhang, F., New technology of Bismuth carbonate from barium carbonate, Trans. Nonferrous Met. Soc. China. 2006, vol. 16, no. 7, pp. 1295–1299 (in Chinese).
Tong, C.R., Wu, J.P., and Li, J.C., Synergistic extraction of As, Sb and Bi in copper electrolyte with TBP and N1923, Nonferrous Met., 2012, no. 5, pp. 17–20 (in Chinese).
Xiao, F. X., Zheng, Y. J., Yong, W., Jian, H.S., Huang, X.Y., and Ma, Y.T., Purification mechanism of copper electrolyte by As (III), Trans. Nonferrous Met. Soc. China. 2008, vol. 18, no. 5, pp. 1275–1279.
Wang, X.W., Wang, X.M., and Wang, M.Y., Characteristics of BiAsO4 precipitate formation in copper electrolyte, Hydrometallurgy. 2017, vol. 171, pp. 95–98.
Wang, X.W., Chen, Q.Y., Yin, Z.L., Wang, M.Y., and Tang, F., The role of arsenic in the homogeneous precipitation of As, Sb and Bi impurities in copper electrolyte, Hydrometallurgy. 2011, vol. 108, no. 3, pp. 199–204.
Peng, Y.L., Zheng, Y.J., and Chen, W.M., The oxidation of arsenic from As(III) to As(V) during copper electrorefining, Hydrometallurgy. 2012, vols. 129–130, pp. 156–160.
Xiao, F.X., Mao, J.W., Cao, D., Shen, X.N., and Yang, D.X., Research progress of self-purification of copper electrolyte, China Nonferrous Met., 2011, vol. B, no. 6, pp. 73–79 (in Chinese)
Wang, X.W., Wang, M.Y., and Wang, X.M., CN Patent 104060295A, 2014.
Binz, F., and Friedrich, B., Recovery of antimony trioxide flame retardants from lead refining residues by slag conditioning and fuming, Chem. Ingen. Technik- CIT. 2015, vol. 87, no. 11, pp. 1569–1579.
Yin, L. and Sridhar, S., Effects of residual elements arsenic, antimony, and tin on surface hot shortness, Metall. Mater. Trans. B., 2011, vol. 42, no. 5, pp. 1031–1043.
Morales, A., Cruells, M., Roca, A., and Bergo, R., Treatment of copper flash smelter flue dusts for copper and zinc extraction and arsenic stabilization, Hydrometallurgy. 2010, vol. 105, nos. 1–2, pp. 148–154.
Chen, Y., Liao, T., Li, G.B., Chen, B.Z., and Si, X.C., Recovery of bismuth and arsenic from copper smelter flue dusts after copper and zinc extraction, Miner. Eng., 2012, vol. 39, no. 12, pp. 23–28.
Aguilera, E.M., Vera, M., Viñals, J, et al., Characterization of raw and decopperized anode slimes from a Chilean fefinery, Metall. Mater. Trans. B., 2016, vol. 47, no. 2, pp. 1315–1324.
Mirazimi, S.M.J., Rashchi, F., Vahidi, E., and Mostoufi, N., Optimization and dissolution kinetics of vanadium recovery from LDconverter slag in alkaline media, Rus. J. Non-Ferrous Met., 2016, vol. 57, no. 5, pp. 395–404.
Li, Y.H., Liu, Z.H., Li, Q.H., Zhao, Z.W., Liu, Z.Y., Li, Z., and Li, L., Removal of arsenic from arsenate complex contained in secondary zinc oxide, Hydrometallurgy. 2011, vol. 109, no. 3, pp. 237–244.
Yi, Y., Shi, J., Tian, Q.H, and Guo, X.Y., Novel technology for preparation of sodium pyroantimonate from alkali leaching residue of high arsenic dust, Trans. Nonferrous Met. Soc. China. 2015, no. 1, pp. 241–249 (in Chinese).
Liang, Y.X., Chen, S.L., and Guo, X.Y., The status quo of purification and impurities removal of As, Sb, Bi in copper electrolyte, China Nonferrous Metal., 2009, vol. B, no. 4, pp. 69–73, (in Chinese).
Wang, L.M., Study on purification of copper electrolyte by chemical method (Master’s Dissertation), Changsha: Central South University. 2010, pp. 23–27 (in Chinese).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
About this article
Cite this article
Wang, JL., Hu, HZ. The Use of Antimony Trioxide in Copper Electrolyte Purification and Its Subsequent Regeneration: An Experimental and Mechanistic Study. Russ. J. Non-ferrous Metals 59, 237–249 (2018). https://doi.org/10.3103/S1067821218030161
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S1067821218030161