Abstract
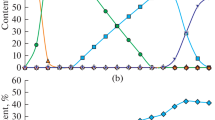
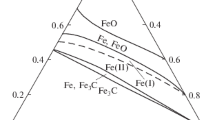
A computer simulation of the chemical process of direct iron reduction in aggregates is performed with the help of the MIDREX technology. Four variants of this technology, which differ by the type of raw material and working process temperature, are analyzed. It is determined that when the process temperature increases, the required consumption of natural gas decreases and the methane in the waste gas is completely removed. It is also noted that at a constant process temperature, the concentrations of all of the components of the system linearly depend on the flow rate of the reducing gas. This circumstance allowed us to record a single balance equation for all of the variants of the studied chemical process.
Similar content being viewed by others
REFERENCES
Ünal, H.I., Turgut, E., Atapek, Ş.H., and Alkan, A., Direct reduction of ferrous oxides to form an iron-rich alternative charge material, High Temp. Mater. Process., 2015, vol. 34, no. 8, pp. 751–756. https://doi.org/10.1515/htmp-2014-0125
Atsushi, M., Uemura, H., and Sakaguchi, T., MIDREX processes, Kobelco Technol. Rev., 2010, no. 29, pp. 50–57.
Mouer, A., Direct from MIDREX, 2nd Quarter, 2009, pp. 3–9.
MIDREX process: Production of sponge iron. https://uas.su/books/srmp/511/razdel511.php.
Trusov, B.G., Baza dannykh i programmnyi kompleks TERRA, redaktsiya 6.3 (Database and Program Complex TERRA, Version 6.3), Moscow: Mosk. Gos. Tekh. Univ. im. Baumana, 2013.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by S. Kuznetsov
About this article
Cite this article
Berdnikov, V.I., Gudim, Y.A. On Thermodynamics of Iron Reduction from Oxides. Steel Transl. 52, 199–200 (2022). https://doi.org/10.3103/S0967091222020024
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0967091222020024