Abstract
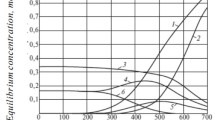
The equilibrium composition of the Fe2O3-H2-CO system depending on temperature and pressure was calculated by the methods of chemical thermodynamics at different molar ratios between the system components. It was found that the equilibrium composition of the mixtures formed in this system mainly depends on ratios between the system components. The effect of the partial pressures of H2 and CO on the equilibrium composition of products in the Fischer-Tropsch synthesis was determined.
Similar content being viewed by others
References
Cairns, E.J. and Tevebaugh, A.D., J. Chem. Eng. Data, 1964, vol. 9, no. 3, p. 453.
Dry, M.E., Chemtech., 1982, p. 744.
Shroff, M.D., Kalakkad, D.S., Coulter, K.E., et al., J. Catal., 1993, vol. 156, p. 185.
Davis, B.H., Catal. Today, 2009, vol. 141, p. 25.
Stepanov, N.F., Erlykina, M.E., and Filippov, G.G., Metody lineinoi algebry v fizicheskoi khimii (Methods of Linear Algebra in Physical Chemistry), Moscow: Mosk. Gos. Univ., 1976.
Oh-Shim Joo and Kwang-Deog Jung, Bull. Korean Chem. Soc., 2003, vol. 24,no. 1, p. 45.
Ho Joon Seo and Eui Yeon Yu, J. End. Eng. Chem., 1997, vol. 3, no. 2, p. 85.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.M. Gyul’maliev, A.Yu. Krylova, A.S. Lyadov, 2012, published in Khimiya Tverdogo Topliva, 2012, No. 1, pp. 31–38.
About this article
Cite this article
Gyul’maliev, A.M., Krylova, A.Y. & Lyadov, A.S. A thermodynamic study of the Fe2O3-H2-CO system. Solid Fuel Chem. 46, 28–36 (2012). https://doi.org/10.3103/S0361521912010053
Received:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0361521912010053