Abstract
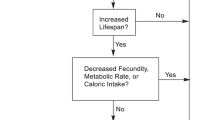
Although the pathophysiology of neurodegenerative diseases is distinct for each disease, considerable evidence suggests that a single manipulation, dietary restriction, is strikingly protective against a wide range of such diseases. Thus pharmacological mimetics of dietary restrictions could prove widely protective across a range of neurodegenerative diseases. The PPAR transcription complex functions to re-program gene expression in response to nutritional deprivation as well as in response to a wide variety of lipophilic compounds. In mammals there are three PPAR homologs, which dimerize with RXR homologs and recruit coactivators Pgc1-alpha and Creb-binding protein (Cbp). PPARs are currently of clinical interest mainly because PPAR activators are approved for use in humans to reduce lipidemia and to improve glucose control in Type 2 diabetic patients. However, pharmacological enhancement of the activity of the PPAR complex is neuroprotective across a wide variety of models for neuropathological processes, including stroke, Alzheimer’s disease, Parkinson’s disease, and Huntington’s disease. Conversely activity of the PPAR transcriptional complex is reduced in a variety of neuropathological processes. The main mechanisms mediating the neuroprotective effects of the PPAR transcription complex appear to be re-routing metabolism away from glucose metabolism and toward alternative subtrates, and reduction in inflammatory processes. Recent evidence suggests that the PPAR transcriptional complex may also mediate protective effects of dietary restriction on neuropathological processes. Thus this complex represents one of the most promising for the development of pharmacological treatment of neurodegenerative diseases.
Similar content being viewed by others
References
Morrison J.H., Hof P.R., Life and death of neurons in the aging brain, Science, 1997, 278, 412–419
Olshansky S.J., Carnes B.A., Ever since Gompertz, Demography, 1997, 34, 1–15
Greer E.L., Brunet A., Different dietary restriction regimens extend lifespan by both independent and overlapping genetic pathways in C. elegans, Aging Cell, 2009, 8, 113–127
Spindler S.R., Rapid and reversible induction of the longevity, anticancer and genomic effects of caloric restriction, Mech. Ageing Dev., 2005
Anson R.M., Guo Z., de Cabo R., Iyun T., Rios M., Hagepanos A., et al., Intermittent fasting dissociates beneficial effects of dietary restriction on glucose metabolism and neuronal resistance to injury from calorie intake, Proc. Natl. Acad. Sci. USA, 2003, 100, 6216–6220
Fontana L., Partridge L., Longo V.D., Extending healthy life span—from yeast to humans, Science, 2010, 328, 321–326
Schroeder J.E., Richardson J.C., Virley D.J., Dietary manipulation and caloric restriction in the development of mouse models relevant to neurological diseases, Biochim. Biophys. Acta, 2010, 1802, 840–846
Wang J., Ho L., Qin W., Rocher A.B., Seror I., Humala N., et al., Caloric restriction attenuates beta-amyloid neuropathology in a mouse model of Alzheimer’s disease, Faseb J., 2005, 19, 659–661
Patel N.V., Gordon M.N., Connor K.E., Good R.A., Engelman R.W., Mason J., et al., Caloric restriction attenuates Abeta-deposition in Alzheimer transgenic models, Neurobiol. Aging, 2005, 26, 995–1000
Halagappa V.K., Guo Z., Pearson M., Matsuoka Y., Cutler R.G., Laferla F.M., et al., Intermittent fasting and caloric restriction ameliorate agerelated behavioral deficits in the triple-transgenic mouse model of Alzheimer’s disease, Neurobiol. Dis., 2007, 26, 212–220
Steinkraus K.A., Smith E.D., Davis C., Carr D., Pendergrass W.R., Sutphin G.L., et al., Dietary restriction suppresses proteotoxicity and enhances longevity by an hsf-1-dependent mechanism in Caenorhabditis elegans, Aging Cell, 2008, 7, 394–404
Zhang M., Poplawski M., Yen K., Cheng H., Bloss E., Zhu X., et al., Role of CBP and SATB-1 in aging, dietary restriction, and insulin-like signaling, PLoS Biol., 2009, 7, e1000245
Qin W., Chachich M., Lane M., Roth G., Bryant M., de Cabo R., et al., Calorie restriction attenuates Alzheimer’s disease type brain amyloidosis in Squirrel monkeys (Saimiri sciureus), J. Alzheimers Dis., 2006, 10, 417–422
Bruce-Keller A.J., Umberger G., McFall R., Mattson M.P., Food restriction reduces brain damage and improves behavioral outcome following excitotoxic and metabolic insults, Ann. Neurol., 1999, 45, 8–15
Duan W., Mattson M.P., Dietary restriction and 2-deoxyglucose administration improve behavioral outcome and reduce degeneration of dopaminergic neurons in models of Parkinson’s disease, J. Neurosci. Res., 1999, 57, 195–206
Holmer H.K., Keyghobadi M., Moore C., Menashe R.A., Meshul C.K., Dietary restriction affects striatal glutamate in the MPTP-induced mouse model of nigrostriatal degeneration, Synapse, 2005, 57, 100–112
Maswood N., Young J., Tilmont E., Zhang Z., Gash D.M., Gerhardt G.A., et al., Caloric restriction increases neurotrophic factor levels and attenuates neurochemical and behavioral deficits in a primate model of Parkinson’s disease, Proc. Natl. Acad. Sci. USA, 2004, 101, 18171–18176
Duan W., Guo Z., Jiang H., Ware M., Li X.J., Mattson M.P., Dietary restriction normalizes glucose metabolism and BDNF levels, slows disease progression, and increases survival in huntingtin mutant mice, Proc. Natl. Acad. Sci. USA, 2003, 100, 2911–2916
Yu Z.F., Mattson M.P., Dietary restriction and 2-deoxyglucose administration reduce focal ischemic brain damage and improve behavioral outcome: evidence for a preconditioning mechanism, J. Neurosci. Res., 1999, 57, 830–839
Manzanero S., Gelderblom M., Magnus T., Arumugam T.V., Calorie restriction and stroke, Exp. Transl. Stroke Med., 2011, 3, 8
Plunet W.T., Streijger F., Lam C.K., Lee J.H., Liu J., Tetzlaff W., Dietary restriction started after spinal cord injury improves functional recovery, Exp. Neurol., 2008, 213, 28–35
Ingram D.K., Weindruch R., Spangler E.L., Freeman J.R., Walford R.L., Dietary restriction benefits learning and motor performance of aged mice, J. Gerontol., 1987, 42, 78–81
Markowska A.L., Savonenko A., Retardation of cognitive aging by lifelong diet restriction: implications for genetic variance, Neurobiol. Aging, 2002, 23, 75–86
Adams M.M., Shi L., Linville M.C., Forbes M.E., Long A.B., Bennett C., et al., Caloric restriction and age affect synaptic proteins in hippocampal CA3 and spatial learning ability, Exp. Neurol., 2008, 211, 141–149
Carter C.S., Leeuwenburgh C., Daniels M., Foster T.C., Influence of calorie restriction on measures of age-related cognitive decline: role of increased physical activity, J. Gerontol. A Biol. Sci. Med. Sci., 2009, 64, 850–859
Johnstone A.M., Fasting — the ultimate diet?, Obes. Rev., 2007, 8, 211–222
Mobbs C.V., Hof P.R., Mechanisms of dietary restriction in aging and disease,Karger, Basel, 2007
Caccamo A., Maldonado M.A., Bokov A.F., Majumder S., Oddo S., CBP gene transfer increases BDNF levels and ameliorates learning and memory deficits in a mouse model of Alzheimer’s disease, Proc. Natl. Acad. Sci. USA, 2010, 107, 22687–22692
Steffan J.S., Kazantsev A., Spasic-Boskovic O., Greenwald M., Zhu Y.Z., Gohler H., et al., The Huntington’s disease protein interacts with p53 and CREB-binding protein and represses transcription, Proc. Natl. Acad. Sci. USA, 2000, 97, 6763–6768
Cong S.Y., Pepers B.A., Evert B.O., Rubinsztein D.C., Roos R.A., van Ommen G.J., et al., Mutant huntingtin represses CBP, but not p300, by binding and protein degradation, Mol. Cell. Neurosci., 2005, 30, 560–571
Jiang H., Poirier M.A., Liang Y., Pei Z., Weiskittel C.E., Smith W.W., et al., Depletion of CBP is directly linked with cellular toxicity caused by mutant huntingtin, Neurobiol. Dis., 2006, 23, 543–551
Kilgore M., Miller C.A., Fass D.M., Hennig K.M., Haggarty S.J., Sweatt J.D., et al., Inhibitors of class 1 histone deacetylases reverse contextual memory deficits in a mouse model of Alzheimer’s disease, Neuropsychopharmacology, 2010, 35, 870–880
Govindarajan N., Agis-Balboa R.C., Walter J., Sananbenesi F., Fischer A., Sodium butyrate improves memory function in an Alzheimer’s disease mouse model when administered at an advanced stage of disease progression, J. Alzheimers Dis., 2011, 26, 187–197
Steffan J.S., Bodai L., Pallos J., Poelman M., McCampbell A., Apostol B.L., et al., Histone deacetylase inhibitors arrest polyglutaminedependent neurodegeneration in Drosophila, Nature, 2001, 413, 739–743
Bates E.A., Victor M., Jones A.K., Shi Y., Hart A.C., Differential contributions of Caenorhabditis elegans histone deacetylases to huntingtin polyglutamine toxicity, J. Neurosci., 2006, 26, 2830–2838
Jia H., Pallos J., Jacques V., Lau A., Tang B., Cooper A., et al., Histone deacetylase (HDAC) inhibitors targeting HDAC3 and HDAC1 ameliorate polyglutamine-elicited phenotypes in model systems of Huntington’s disease, Neurobiol. Dis., 2012, 46, 351–361
Kidd S.K., Schneider J.S., Protective effects of valproic acid on the nigrostriatal dopamine system in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson’s disease, Neuroscience, 2011, 194, 189–194
Langley B., Gensert J.M., Beal M.F., Ratan R.R., Remodeling chromatin and stress resistance in the central nervous system: histone deacetylase inhibitors as novel and broadly effective neuroprotective agents, Curr. Drug Targets CNS Neurol. Disord., 2005, 4, 41–50
Mobbs C.V., Mastaitis J.W., Zhang M., Isoda F., Cheng H., Yen K., Secrets of the lac operon. Glucose hysteresis as a mechanism in dietary restriction, aging and disease, Interdiscip. Top. Gerontol., 2007, 35, 39–68
Mobbs C.V., Yen K., Mastaitis J., Nguyen H., Watson E., Wurmbach E., et al., Mining microarrays for metabolic meaning: nutritional regulation of hypothalamic gene expression, Neurochem. Res., 2004, 29, 1093–1103
Poplawski M.M., Mastaitis J.W., Yang X.J., Mobbs C.V., Hypothalamic responses to fasting indicate metabolic reprogramming away from glycolysis toward lipid oxidation, Endocrinology, 2010, 151, 5206–5217
Poplawski M.M., Mastaitis J.W., Mobbs C.V., Naloxone, but not valsartan, preserves responses to hypoglycemia after antecedent hypoglycemia: role of metabolic reprogramming in counterregulatory failure, Diabetes, 2011, 60, 39–46
Diano S., Liu Z.W., Jeong J.K., Dietrich M.O., Ruan H.B., Kim E., et al., Peroxisome proliferation-associated control of reactive oxygen species sets melanocortin tone and feeding in diet-induced obesity, Nat. Med., 2011, 17, 1121–1127
Puigserver P., Adelmant G., Wu Z., Fan M., Xu J., O’Malley B., et al., Activation of PPARgamma coactivator-1 through transcription factor docking, Science, 1999, 286, 1368–1371
Frier B.C., Jacobs R.L., Wright D.C., Interactions between the consumption of a high-fat diet and fasting in the regulation of fatty acid oxidation enzyme gene expression: an evaluation of potential mechanisms, Am. J. Physiol. Regul. Integr. Comp. Physiol., 2011, 300, R212–221
Rodriguez A.M., Sanchez J., Tobaruela A., Priego T., Pico C., Palou A., Time-course effects of increased fatty acid supply on the expression of genes involved in lipid/glucose metabolism in muscle cells, Cell. Physiol. Biochem., 2010, 25, 337–346
Wahli W., Michalik L., PPARs at the crossroads of lipid signaling and inflammation, Trends Endocrinol. Metab., 2012, 23, 351–363
Issemann I., Green S., Activation of a member of the steroid hormone receptor superfamily by peroxisome proliferators, Nature, 1990, 347, 645–650
Vega R.B., Huss J.M., Kelly D.P., The coactivator PGC-1 cooperates with peroxisome proliferator-activated receptor alpha in transcriptional control of nuclear genes encoding mitochondrial fatty acid oxidation enzymes, Mol. Cell. Biol., 2000, 20, 1868–1876
Song S., Attia R.R., Connaughton S., Niesen M.I., Ness G.C., Elam M.B., et al., Peroxisome proliferator activated receptor alpha (PPARalpha) and PPAR gamma coactivator (PGC-1alpha) induce carnitine palmitoyltransferase IA (CPT-1A) via independent gene elements, Mol. Cell. Endocrinol., 2010, 325, 54–63
Yang S.Y., He X.Y., Schulz H., Fatty acid oxidation in rat brain is limited by the low activity of 3-ketoacyl-coenzyme A thiolase, J. Biol. Chem., 1987, 262, 13027–13032
Speijer D., Oxygen radicals shaping evolution: why fatty acid catabolism leads to peroxisomes while neurons do without it: FADH/NADH flux ratios determining mitochondrial radical formation were crucial for the eukaryotic invention of peroxisomes and catabolic tissue differentiation, Bioessays, 2011, 33, 88–94
Auestad N., Korsak R.A., Morrow J.W., Edmond J., Fatty acid oxidation and ketogenesis by astrocytes in primary culture, J. Neurochem., 1991, 56, 1376–1386
Deplanque D., Gele P., Petrault O., Six I., Furman C., Bouly M., et al., Peroxisome proliferator-activated receptor-alpha activation as a mechanism of preventive neuroprotection induced by chronic fenofibrate treatment, J. Neurosci., 2003, 23, 6264–6271
Inoue H., Jiang X.F., Katayama T., Osada S., Umesono K., Namura S., Brain protection by resveratrol and fenofibrate against stroke requires peroxisome proliferator-activated receptor alpha in mice, Neurosci. Lett., 2003, 352, 203–206
Howitz K.T., Bitterman K.J., Cohen H.Y., Lamming D.W., Lavu S., Wood J.G., et al., Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan, Nature, 2003, 425, 191–196
Collino M., Aragno M., Mastrocola R., Benetti E., Gallicchio M., Dianzani C., et al., Oxidative stress and inflammatory response evoked by transient cerebral ischemia/reperfusion: effects of the PPAR-alpha agonist WY14643, Free Radic. Biol. Med., 2006, 41, 579–589
D’Agostino G., Russo R., Avagliano C., Cristiano C., Meli R., Calignano A., Palmitoylethanolamide protects against the amyloid-beta25-35-induced learning and memory impairment in mice, an experimental model of Alzheimer disease, Neuropsychopharmacology, 2012, 37, 1784–1792
Bhateja D.K., Dhull D.K., Gill A., Sidhu A., Sharma S., Reddy B.V., et al., Peroxisome proliferator-activated receptor-alpha activation attenuates 3-nitropropionic acid induced behavioral and biochemical alterations in rats: possible neuroprotective mechanisms, Eur. J. Pharmacol., 2012, 674, 33–43
Lovett-Racke A.E., Hussain R.Z., Northrop S., Choy J., Rocchini A., Matthes L., et al., Peroxisome proliferator-activated receptor alpha agonists as therapy for autoimmune disease, J. Immunol., 2004, 172, 5790–5798
Dasgupta S., Roy A., Jana M., Hartley D.M., Pahan K., Gemfibrozil ameliorates relapsing-remitting experimental autoimmune encephalomyelitis independent of peroxisome proliferator-activated receptor-alpha, Mol. Pharmacol., 2007, 72, 934–946
Gocke A.R., Hussain R.Z., Yang Y., Peng H., Weiner J., Ben L.H., et al., Transcriptional modulation of the immune response by peroxisome proliferator-activated receptor-{alpha} agonists in autoimmune disease, J. Immunol., 2009, 182, 4479–4487
Gray E., Ginty M., Kemp K., Scolding N., Wilkins A., Peroxisome proliferator-activated receptor-alpha agonists protect cortical neurons from inflammatory mediators and improve peroxisomal function, Eur. J. Neurosci., 2011, 33, 1421–1432
Zhu Y., Alvares K., Huang Q., Rao M.S., Reddy J.K., Cloning of a new member of the peroxisome proliferator-activated receptor gene family from mouse liver, J. Biol. Chem., 1993, 268, 26817–26820
Kliewer S.A., Forman B.M., Blumberg B., Ong E.S., Borgmeyer U., Mangelsdorf D.J., et al., Differential expression and activation of a family of murine peroxisome proliferator-activated receptors, Proc. Natl. Acad. Sci. USA, 1994, 91, 7355–7359
Sarruf D.A., Yu F., Nguyen H.T., Williams D.L., Printz R.L., Niswender K.D., et al., Expression of peroxisome proliferator-activated receptorgamma in key neuronal subsets regulating glucose metabolism and energy homeostasis, Endocrinology, 2009, 150, 707–712
Lehrke M., Lazar M.A., The many faces of PPARgamma, Cell, 2005, 123, 993–999
Gillespie W., Tyagi N., Tyagi S.C., Role of PPARgamma, a nuclear hormone receptor in neuroprotection, Indian J. Biochem. Biophys., 2011, 48, 73–81
Chen Y.C., Wu J.S., Tsai H.D., Huang C.Y., Chen J.J., Sun G.Y., et al., Peroxisome Proliferator-Activated Receptor gamma (PPAR-gamma) and neurodegenerative disorders, Mol. Neurobiol., 2012, [Epub ahead of print]
Breidert T., Callebert J., Heneka M.T., Landreth G., Launay J.M., Hirsch E.C., Protective action of the peroxisome proliferator-activated receptor-gamma agonist pioglitazone in a mouse model of Parkinson’s disease, J. Neurochem., 2002, 82, 615–624
Shimazu T., Inoue I., Araki N., Asano Y., Sawada M., Furuya D., et al., A peroxisome proliferator-activated receptor-gamma agonist reduces infarct size in transient but not in permanent ischemia, Stroke, 2005, 36, 353–359
Sundararajan S., Gamboa J.L., Victor N.A., Wanderi E.W., Lust W.D., Landreth G.E., Peroxisome proliferator-activated receptorgamma ligands reduce inflammation and infarction size in transient focal ischemia, Neuroscience, 2005, 130, 685–696
Zhao Y., Patzer A., Gohlke P., Herdegen T., Culman J., The intracerebral application of the PPARgamma-ligand pioglitazone confers neuroprotection against focal ischaemia in the rat brain, Eur. J. Neurosci., 2005, 22, 278–282
Collino M., Aragno M., Mastrocola R., Gallicchio M., Rosa A.C., Dianzani C., et al., Modulation of the oxidative stress and inflammatory response by PPAR-gamma agonists in the hippocampus of rats exposed to cerebral ischemia/reperfusion, Eur. J. Pharmacol., 2006, 530, 70–80
Tureyen K., Kapadia R., Bowen K.K., Satriotomo I., Liang J., Feinstein D.L., et al., Peroxisome proliferator-activated receptor-gamma agonists induce neuroprotection following transient focal ischemia in normotensive, normoglycemic as well as hypertensive and type-2 diabetic rodents, J. Neurochem., 2007, 101, 41–56
Schmerbach K., Schefe J.H., Krikov M., Muller S., Villringer A., Kintscher U., et al., Comparison between single and combined treatment with candesartan and pioglitazone following transient focal ischemia in rat brain, Brain Res., 2008, 1208, 225–233
Patzer A., Zhao Y., Stock I., Gohlke P., Herdegen T., Culman J., Peroxisome proliferator-activated receptorsgamma (PPARgamma) differently modulate the interleukin-6 expression in the peri-infarct cortical tissue in the acute and delayed phases of cerebral ischaemia, Eur. J. Neurosci., 2008, 28, 1786–1794
Lee S.R., Kim H.Y., Hong J.S., Baek W.K., Park J.W., PPARgamma agonist pioglitazone reduces matrix metalloproteinase-9 activity and neuronal damage after focal cerebral ischemia, Biochem. Biophys. Res. Commun., 2009, 380, 17–21
Ji S., Kronenberg G., Balkaya M., Farber K., Gertz K., Kettenmann H., et al., Acute neuroprotection by pioglitazone after mild brain ischemia without effect on long-term outcome, Exp. Neurol., 2009, 216, 321–328
Kaundal R.K., Iyer S., Kumar A., Sharma S.S., Protective effects of pioglitazone against global cerebral ischemic-reperfusion injury in gerbils, J. Pharmacol. Sci., 2009, 109, 361–367
Zhang H.L., Xu M., Wei C., Qin A.P., Liu C.F., Hong L.Z., et al., Neuroprotective effects of pioglitazone in a rat model of permanent focal cerebral ischemia are associated with peroxisome proliferators-activated receptor gamma-mediated suppression of nuclear factor-kappaB signaling pathway, Neuroscience, 2011, 176, 381–395
Feinstein D.L., Galea E., Gavrilyuk V., Brosnan C.F., Whitacre C.C., Dumitrescu-Ozimek L., et al., Peroxisome proliferator-activated receptor-gamma agonists prevent experimental autoimmune encephalomyelitis, Ann. Neurol., 2002, 51, 694–702
Schutz B., Reimann J., Dumitrescu-Ozimek L., Kappes-Horn K., Landreth G.E., Schurmann B., et al., The oral antidiabetic pioglitazone protects from neurodegeneration and amyotrophic lateral sclerosislike symptoms in superoxide dismutase-G93A transgenic mice, J. Neurosci., 2005, 25, 7805–7812
Kumar P., Kaundal R.K., More S., Sharma S.S., Beneficial effects of pioglitazone on cognitive impairment in MPTP model of Parkinson’s disease, Behav. Brain Res., 2009, 197, 398–403
Searcy J.L., Phelps J.T., Pancani T., Kadish I., Popovic J., Anderson K.L., et al., Long-term pioglitazone treatment improves learning and attenuates pathological markers in a mouse model of Alzheimer’s disease, J. Alzheimers Dis., 2012, 30, 943–961
Gupta R., Gupta L.K., Improvement in long term and visuo-spatial memory following chronic pioglitazone in mouse model of Alzheimer’s disease, Pharmacol. Biochem. Behav., 2012, 102, 184–190
Wiggin T.D., Kretzler M., Pennathur S., Sullivan K.A., Brosius F.C., Feldman E.L., Rosiglitazone treatment reduces diabetic neuropathy in streptozotocin-treated DBA/2J mice, Endocrinology, 2008, 149, 4928–4937
Yamagishi S., Ogasawara S., Mizukami H., Yajima N., Wada R., Sugawara A., et al., Correction of protein kinase C activity and macrophage migration in peripheral nerve by pioglitazone, peroxisome proliferator activated-gamma-ligand, in insulin-deficient diabetic rats, J. Neurochem., 2008, 104, 491–499
Sato T., Hanyu H., Hirao K., Kanetaka H., Sakurai H., Iwamoto T., Efficacy of PPAR-gamma agonist pioglitazone in mild Alzheimer disease, Neurobiol. Aging, 2011, 32, 1626–1633
Geldmacher D.S., Fritsch T., McClendon M.J., Landreth G., A randomized pilot clinical trial of the safety of pioglitazone in treatment of patients with Alzheimer disease, Arch. Neurol., 2011, 68, 45–50
Harrington C., Sawchak S., Chiang C., Davies J., Donovan C., Saunders A.M., et al., Rosiglitazone does not improve cognition or global function when used as adjunctive therapy to AChE inhibitors in mild-to-moderate Alzheimer’s disease: two phase 3 studies, Curr. Alzheimer Res., 2011, 8, 592–606
Miller B.W., Willett K.C., Desilets A.R., Rosiglitazone and pioglitazone for the treatment of Alzheimer’s disease, Ann. Pharmacother., 2011, 45, 1416–1424
Roberts L.D., Murray A.J., Menassa D., Ashmore T., Nicholls A.W., Griffin J.L., The contrasting roles of PPARdelta and PPARgamma in regulating the metabolic switch between oxidation and storage of fats in white adipose tissue, Genome Biol., 2011, 12, R75
Woods J.W., Tanen M., Figueroa D.J., Biswas C., Zycband E., Moller D.E., et al., Localization of PPARdelta in murine central nervous system: expression in oligodendrocytes and neurons, Brain Res., 2003, 975, 10–21
Kalinin S., Richardson J.C., Feinstein D.L., A PPARdelta agonist reduces amyloid burden and brain inflammation in a transgenic mouse model of Alzheimer’s disease, Curr. Alzheimer Res., 2009, 6, 431–437
Iwashita A., Muramatsu Y., Yamazaki T., Muramoto M., Kita Y., Yamazaki S., et al., Neuroprotective efficacy of the peroxisome proliferatoractivated receptor delta-selective agonists in vitro and in vivo, J. Pharmacol. Exp. Ther., 2007, 320, 1087–1096
Yin K.J., Deng Z., Hamblin M., Xiang Y., Huang H., Zhang J., et al., Peroxisome proliferator-activated receptor delta regulation of miR-15a in ischemia-induced cerebral vascular endothelial injury, J. Neurosci., 2010, 30, 6398–6408
Cramer P.E., Cirrito J.R., Wesson D.W., Lee C.Y., Karlo J.C., Zinn A.E., et al., ApoE-directed therapeutics rapidly clear beta-amyloid and reverse deficits in AD mouse models, Science, 2012, 335, 1503–1506
Friling S., Bergsland M., Kjellander S., Activation of Retinoid X Receptor increases dopamine cell survival in models for Parkinson’s disease, BMC Neurosci., 2009, 10, 146
Tsunemi T., La Spada A.R., PGC-1alpha at the intersection of bioenergetics regulation and neuron function: from Huntington’s disease to Parkinson’s disease and beyond, Prog. Neurobiol., 2012, 97, 142–151
Lin J., Wu P.H., Tarr P.T., Lindenberg K.S., St-Pierre J., Zhang C.Y., et al., Defects in adaptive energy metabolism with CNS-linked hyperactivity in PGC-1alpha null mice, Cell, 2004, 119, 121–135
Leone T.C., Lehman J.J., Finck B.N., Schaeffer P.J., Wende A.R., Boudina S., et al., PGC-1alpha deficiency causes multi-system energy metabolic derangements: muscle dysfunction, abnormal weight control and hepatic steatosis, PLoS Biol., 2005, 3, e101
Cui L., Jeong H., Borovecki F., Parkhurst C.N., Tanese N., Krainc D., Transcriptional repression of PGC-1alpha by mutant huntingtin leads to mitochondrial dysfunction and neurodegeneration, Cell, 2006, 127, 59–69
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Mobbs, C.V., Moreno, C.L., Kim, E.S. et al. Neuroprotection by dietary restriction and the PPAR transcription complex. Translat.Neurosci. 3, 234–241 (2012). https://doi.org/10.2478/s13380-012-0035-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s13380-012-0035-8