Abstract
To evaluate the association of multidrug resistance 1 (MDR1) polymorphism and the expression of P-glycoprotein (Pgp) in Chinese refractory lupus nephritis (LN) patients. Polymerase chain reaction-direct sequencing was used to analyze MDR1 polymorphism. The genotype distribution of MDR1 polymorphism in 132 SLE (systemic lupus erythematosus) patients was evaluated. ELISA was used to measure the expression of Pgp. Relationship among Pgp expression, MDR1 polymorphism, SLEDAI (SLE disease activity index), and kidney pathological score was analyzed by using One-way ANOVA and Pearson linear correlation. The frequency distribution of the MDR1 gene was consistent with the Hardy-Weinberg equilibrium. Compared with CT and CC, patients with T/T homozygote in MDR1 C3435T had significantly increased Pgp expression in the refractory group (p < 0.05). Additionally, SLEDAI score was positively correlated with Pgp expression (r = 0.481, p < 0.05). Also, Pgp expression was positively correlated with renal pathological activity index (r = 0.76, p < 0.05). MDR1 C3435T polymorphism is significantly associated with Pgp expression in patients with refractory LN. Pgp expression is closely related to SLEDAI and renal pathological score. Thus, Pgp may be useful in evaluation of the prognosis of patients with refractory LN.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Systemic lupus erythematosus (SLE) is a diffuse, systemic autoimmune disease. When renal involvement occurs, it is known as lupus nephritis (LN). About 50% of patients with SLE gradually develop LN (Lanata et al. 2018). Renal pathological biopsy results show almost 100% of SLE patients have kidney disease, and about 20% of SLE patients eventually progress to end-stage renal disease (Shao 2017). Hormones and immunosuppressants are the main drugs for the treatment of LN, and the reactivity to drugs is an important indicator to measure the prognosis of LN (Bawazier 2017). In recent years, the survival rate of SLE patients has been significantly improved due to early diagnosis and comprehensive treatment (Timlin et al. 2017). However, some patients with refractory SLE still have poor or no response to traditional immunosuppressive therapy. Non-response or low response to hormones and immunosuppressive agents, with appearance of multidrug resistance (MDR), are important factors affecting the treatment of SLE (Chen et al. 2016).
Multidrug resistance 1 (MDR1) and the metabolic enzyme CYP3A are two major factors affecting pharmacokinetic metabolism. The protein product encoded by MDR1, P-glycoprotein (Pgp), is an ATP-dependent export-oriented transport pump composed of 1280 amino acids. The main function of Pgp is to actively pump out various chemicals and drugs, which play an important role in the metabolism of drugs in the body (Zhao et al. 2015). Studies have shown that MDR1 gene polymorphism affects the expression and function of Pgp (Ayaz et al. 2013; Wincewicz et al. 2016). There are more than 50 single nucleotide polymorphisms in MDR1, and the two common gene polymorphisms affecting MDR1 gene function are in exon 26 (C3435T) and exon 12 (C1236T) (Subhani et al. 2015; Uludag et al. 2014). The C1236T is a synonymous mutation that does not cause amino acid change (Kozhakhmetov et al. 2013). While, C3435T is the only silent polymorphism currently identified, which may affect the expression of Pgp in different organs/tissues and even in different races (Aydos et al. 2015).
Pgp is one of the most important causes of drug resistance (Hansen et al. 2015; Zoghbi et al. 2017). Wang et al. (2017) found that Pgp overexpression in hormone-resistant SLE patients may be the cause of steroid resistance. Many drugs commonly used to treat SLE, such as methotrexate, glucocorticoids, etc., are Pgp substrates, which could be pumped out of the cell by Pgp, thereby reducing their intracellular concentrations (Feng et al. 2017; García-Carrasco et al. 2015). Some drugs can inhibit the function of Pgp, such as cyclosporine (Gohla 2018; Llaudó et al. 2013). As a transport substrate of Pgp, cyclosporine exerts competitive inhibition and reverses steroid resistance (Yigitaslan et al. 2016). Also, mycophenolic acid has shown inhibitory effect on Pgp activity of lymphocyte in vitro, but its effect is significantly weaker than Pgp-specific blocking agents and cyclophosphamide (Wang et al. 2017). Based on the mentioned study, hormones and some immunosuppressive agents are substrates of Pgp, which may be involved in the formation of MDR in SLE patients. Therefore, it is important to explore the role of MDR1 genotype and Pgp in patients with refractory SLE.
Here, we designed this experiment to investigate different MDR1 genotype at C3435T and C1236T site as well as its corresponding Pgp expression. Understanding the role of MDR1 resistance gene and Pgp in refractory LN may provide a new therapeutic target for refractory LN.
Materials and methods
Patients
A total of 132 patients with SLE were enrolled from Haikou People’s Hospital between January 2012 and December 2015. They were all Han patients. All cases were diagnosed in accordance with the SLE classification standard revised by the American College of Rheumatology in 1997. SLE patients involved in this research had at least four positive items of the 11 criteria. Besides, all participants were in SLE active period. The disease activity was evaluated by SLE disease activity index (SLEDAI) (Ward et al. 2000). Patients with other connective tissue diseases, tumors, severe trauma, serious infections and other diseases were excluded. Besides the diagnosis of SLE, patients with persistent proteinuria (> 0.5 g/day) or multiple urinary protein (+++), and/or cellular casts (including red cell, hemoglobin, granular, tubular or mixed) were diagnosed as LN. LN patients who were without remission after treatment with at least one immunosuppressive agent for more than 6 months, or with LN accompanied by pathological transformation, or with continued positive immunological markers were considered to have refractory LN (Bonilla-Abadía et al. 2013). Renal biopsy puncture was required for refractory LN, and the pathological type was determined according to the 2003 ISN/RPS classification criteria. Age and gender matched patients were enrolled as the control group. The inclusion criteria were as follows: i. SLE patients without renal involvement; ii. SLE patients with normal serum creatinine, negative proteinuria, and negative hematuria; iii. SLE patients without other connective tissue diseases, tumors, severe trauma, or serious infections, etc.
All subjects volunteered to participate in this study and signed informed consent. This study was approved by the ethics committee of Haikou People’s Hospital (Approval no. 2017-082).
Laboratory test
Hemoglobin (Hb), Usea nitrogen (BUN), Serum creatinine (Scr), Albumin (ALB), Immunoglobulin (IgG), C3, C4, antinuclear antibodies (ANA), anti-double-stranded DNA (dsDNA), anti-nuclear extract antibody (anti-ENA antibody), anti-cardiolipin antibody and Pgp were tested in blood of patients with SLE. In addition, 24-h urine protein was also tested.
Polymerase chain reaction-restriction fragment length polymorphism assay
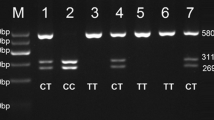
The leukocytes were isolated from peripheral blood by hypotonic hemolysis, and DNA was further extracted by phenol chloroform extraction. The MDR1 gene at C3435T and C1236T site was detected by polymerase chain reaction-restriction fragment length polymorphism assay, respectively. The primer for C3435T and C1236T sites were listed as follows: C3435T Forward primer 5′-TGTTTTCAGCTGCTTGATGG-3′ and Reverse primer5′-AAGGATGTATGTT-GGCCTC-3′; and, C1236T Forward primer 5′- TTGAATGAAGAGTTTCTGATGTTTTC-3′ and Reverse primer: 5′-CCTGACTCACCACACCAATG-3′. Then, the PCR products were digested and then DNA electrophoresis (2.5% agarose gel) was performed to analyze the results.
Statistical analysis
Data analysis was performed using the SPSS 16.0 software package. The measurement data were expressed as mean ± SD. The data was compared by t-test or one-way ANOVA. Hardy-Weinberg equilibrium and rate comparison were performed by chi-square test. The comparison of different genotype parameters also used one-way ANOVA. The correlation between Pgp expression and SLEDAI score or Activity Index (AI) was analyzed by using Pearson linear correlation analysis. P < 0.05 was considered statistically significant.
Results
LN patients have obvious abnormalities in Scr, SLEDAI, Hb and ALB
A total of 132 SLE patients were enrolled, including 17 males and 115 females. This may be caused by the fact that SLE mainly affects female, with the ratio of female to male more than 8:1 (Kotzin 1996). The average age of SLE patients was 35.06 ± 13.45 years old. Among them, 88 patients were with LN, 46 patients were with refractory LN, and 44 patients were non-LN (control). With comparison to the non-LN group, Scr and SLEDAI were significantly increased in the LN and refractory LN group (p < 0.05); however, Hb and ALB were significantly decreased (p < 0.05) (Table 1).
Allele and genotype frequencies
The MDR1 allele and gene frequency was analyzed. The MDR1 C1236T gene frequency was consistent with the Hardy-Weinberg equilibrium (wild homozygous CC = 9.8%, heterozygous CT = 36.8%, and mutant homozygous TT = 51.9%; p = 0.999). Also, MDR1 C3435T gene frequency was consistent with Hardy-Weinberg Equilibrium (wild homozygous CC = 33.1%, heterozygous CT = 52.6% and mutant homozygous TT = 12.8%; p = 0.660). There were no statistical differences between MDR1 C1236T genotype (p = 0.878) and C3435T (p = 0.285) genotype in skin, mucosa, arthritis, serous effusion, renal system, blood system and central nervous system, which indicates no significant difference in genotype of extrarenal involvement.
Compared with CT and CC, MDR1 C3435T significantly increases Pgp expression in refractory group
As shown in Table 2, there was no significant difference in gene frequency between patients with refractory LN and non-refractory LN. Compared with CT and CC, MDR1 C3435T in the refractory LN group had a significant increase in Pgp expression (17.51 ± 29.08 vs3.6 ± 3.09 vs 6.42 ± 9.97; p = 0.047), and the Pgp expression level in different genotypes as follows: TT > CC > CT (Table 3), indicating that the MDR1 C3435TT genotype can increase Pgp expression level or its activity. Other biochemical items showed no significant differences. In addition, immunological parameters such as ANA, dsDNA, anti-SM antibody, anti-RNP antibody and anti-CL antibody among different (MDR1 C1236T or C3435T) genotypes (p > 0.05) also showed no statistical significance (Table 4).
The distribution of genotypes in the different pathological types of refractory LN
As shown in Table 5, in the refractory LN group, there were 5 cases of type III LN, 3 cases of type III + IV LN, 25 cases of type IV LN, 8 cases of type IV + V LN, and 5 cases of type V LN. There were no significant differences in gender, age, biochemical indexes, immunological factors, SLEDAI, and Pgp expression levels among different pathological types. Also, no significant difference was found between MDR1 C1236T (P = 0.184) and C3435T (P = 0.578) genotypes in the distribution of five different pathological types, indicating no correlation was found between different MDR1 genotype distribution and the pathological type.
SLEDAI score and AI are positively correlated to Pgp expression level
Pearson linear correlation analysis was used to determine the relationship of Pgp level with SLEDAI and AI. The results showed that SLEDAI score was closely related to the Pgp expression. Patients with higher SLEDAI score showed a stronger Pgp expression (r = 0.481, p = 0.000, Fig. 1). Moreover, there was a positive correlation between SLEDAI score and Pgp expression in the LN group (r = 0.608, p = 0.000) (Fig. 2) and refractory LN group (r = 0.744, p = 0.000) (Fig. 3), suggesting that lupus activity was positively correlated with Pgp expression (p < 0.01). In addition, Pgp expression was also positively correlated with AI. This indicates that Pgp expression is positively correlated with SLE activity and AI.
Discussion
LN is a common complication of SLE, hormone insensitivity or hormone resistance is still a tricky issue for LN treatment (Kalloo et al. 2013). Excessive expression of Pgp in peripheral lymphocytes is thought to be a reason of hormone resistance (Laberge et al. 2014). Kansal A et al. (Kansal et al. 2016) found that the expression of Pgp in peripheral monocytes of SLE patients was positively correlated with SLEDAI score, and its expression could lead to a decrease in hormone content in lymphocytes, which was considered to be related to the poor efficacy of SLE treatment. Ragab et al. (Ragab and Soliman 2013) found that Pgp expression was positively correlated with SLE disease activity in CD5 and CD7 lymphocyte of SLE children. Compared with hormone alone therapy, hormone combined with cyclophosphamide treatment could significantly reduce Pgp expression (Xuan et al. 2014). The expression level of Pgp in peripheral blood lymphocytes of patients with SLE is closely related to disease activity or the drug treatment efficacy. The imbalance of T cell subsets is not only an important part of the development of SLE, but also closely related to Pgp-mediated drug resistance (Zhang et al. 2018). Studies (Mu et al. 2014) have found that regulatory T cells (Tregs) selectively increased the sensitivity to drugs due to the lack of Pgp expression. Henmi et al. (Henmi et al. 2008) found that Pgp was overexpressed in peripheral blood CD4+ T cells in patients with poor SLE response. These studies demonstrate that down-regulation of Pgp expression is beneficial in improving the efficacy of patients with high-activity SLE, and that Pgp can be used as an important predictor of drug resistance to help patients with SLE to choose appropriate initial treatment strategies.
Many factors may affect Pgp expression, such as drugs, cytokines and gene polymorphisms, etc. (Cuppen et al. 2015). Here, in this study, we found no significant difference in the expression of Pgp among MDR1 C1236T genotypes. The possible mechanism is that MDR1 C1236T is a synonymous mutation and does not affect the function of Pgp. Compared with CT and CC, the TT genotype of MDR1 C3435T in refractory LN group showed a significant increase in Pgp expression (TT > CC > CT), suggesting TT genotype can significantly increase the expression level or activity of Pgp. Although MDR1 C3435T is a synonymous mutation, it can affect the protein folding process by changing the choice and use of codons, and finally lead to the local structural change of Pgp, which affects its affinity for each substrate (Brambila-Tapia 2013). This study also found that MDR1 CC and CT genotype had low Pgp expression level. MDR1 C3435T polymorphism is an extensively studied polymorphism. It is shown that MDR1 TT genotype is associated with low Pgp expression in different tissues and cells (such as enterocytes and peripheral blood mononucler cells) whereas MDR1 CC genotype is associated with increased Pgp expression (Owen et al. 2004). This is not consistent with our results, which may be because of different study populations.
At present, the evaluation system commonly used in the international evaluation of lupus activity (Wang et al. 2016). The SLEDAI score currently has certain limitations including poor correlation in subjective assessment of patients, low score sensitivity and inability to reflect the severity of some activities (Zhou and Jiang 2012). Renal pathology is the gold standard that reflects the activity of LN. However, renal biopsy is an invasive test. Thus, there is an urgent need for a better system to evaluate the activity of SLE disease. This study found that the higher the SLEDAI score, the stronger the Pgp expression, suggesting that the activity of lupus is positively correlated with Pgp expression. The study also found that the expression of Pgp was positively correlated with the AI of kidney disease. The higher the AI, the stronger the expression of Pgp, suggesting that the detection of Pgp levels may predict the disease activity of LN patients to some extent. In this study, MDR1 TT genotype had higher Pgp expression level whereas MDR1 CC and CT genotype had low Pgp level in refractory LN group. The MDR1 C3435T may also predict the disease activity of refractory LN patients to some extent. MDR1 TT genotype may predict greater lupus activity while MDR1 CC and CT genotype may predict weak lupus activity.
This study has some limitations. First, besides Pgp, other factors may also affect SLE hormone resistance. Second, there is a lack of placebo control group. Third, the sample size is small. To better explain the mechanism of MDR1 gene polymorphism on Pgp expression at the gene and cellular levels, we will further detect Pgp mRNA and protein expression in kidney tissue in the following work.
Conclusion
MDR1 C3435T polymorphism is significantly associated with Pgp expression in patients with refractory LN in Chinese Han SLE patients. Pgp expression is closely related to SLEDAI and renal pathological score. Thus, Pgp may be useful in evaluation of the prognosis of patients with refractory LN. Additionally, it is suggested that a more accurate drug regimen should be applied in accordance with different MDR1 C3435T genotype and Pgp expression level. Also, hormone therapy should be individualized. Moreover, blockage of Pgp may become an ideal intervention for refractory LN treatment.
Abbreviations
- MDR1:
-
Multidrug resistance 1
- Pgp:
-
P-glycoprotein
- SLE:
-
Systemic lupus erythematosus
- LN:
-
Lupus nephritis
- SLEDAI:
-
SLE disease activity index
- Hb:
-
Hemoglobin
- BUN:
-
Usea nitrogen
- Scr:
-
Serum creatinine
- ALB:
-
Albumin
- IgG:
-
Immunoglobulin
- ANA:
-
Antinuclear antibodies
- dsDNA:
-
Anti-double-stranded DNA
- anti-ENA antibody:
-
Anti-nuclear extract antibody
- anti-CL antibody:
-
Anti-cardiolipin antibody
- AI:
-
Activity Index
References
Ayaz G, Batar B, Kanigur G, Guven M, Onaran I, Karadag B, Ilerigelen B, Dirican A, Ulutin T (2013) The association of MDR1 C3435T and G2677T/A polymorphisms with plasma platelet-activating factor levels and coronary artery disease risk in Turkish population. Gene 527:301–305. https://doi.org/10.1016/j.gene.2013.06.046
Aydos SE, Karadag A, Ozkan T, Altinok B, Bunsuz M, Heidargholizadeh S, Aydos K, Sunguroglu A (2015) Association of MDR1 C3435T and C1236T single nucleotide polymorphisms with male factor infertility. Genet Mol Res 14:6330–6339. https://doi.org/10.4238/2015.June.11.8
Bawazier LA (2017) Current and emerging therapy on lupus nephritis. Acta Med Indones 49:369–377
Bonilla-Abadía F, Coronel RN, Tobón GJ, Echeverri AF, Muñoz-Buitrón E, Castro AM, Bejarano MA, Cañas CA (2013) Rituximab for remission induction and maintenance in refractory systemic lupus erythematosus. Autoimmune Diseases 2014:731806. https://doi.org/10.1155/2014/731806
Brambila-Tapia AJ (2013) MDR1 (ABCB1) polymorphisms: functional effects and clinical implications. Rev Investig Clin 65:445–454
Chen Z, Shi T, Zhang L, Zhu P, Deng M, Huang C, Hu T, Jiang L, Li J (2016) Mammalian drug efflux transporters of the ATP binding cassette (ABC) family in multidrug resistance: a review of the past decade. Cancer Lett 370:153–164. https://doi.org/10.1016/j.canlet.2015.10.010
Cuppen BV, Welsing PM, Sprengers JJ, Bijlsma JW, Marijnissen AC, van Laar JM, Lafeber FP, Nair SC (2015) Personalized biological treatment for rheumatoid arthritis: a systematic review with a focus on clinical applicability. Rheumatology 74:208–212. https://doi.org/10.1093/rheumatology/kev421
Feng L, Liu L, Zhao Y, Zhao R (2017) Saikosaponins A, C and D enhance liver-targeting effects of anticancer drugs by modulating drug transporters. Oncotarget 8:110092–110102. https://doi.org/10.18632/oncotarget.22639
García-Carrasco M, Mendozapinto C, Macias SD, Verarecabarren M, Vázquez LDL, Méndez SM, Sotosantillán P, Gonzálezramírez R, Ruizarguelles A (2015) P-glycoprotein in autoimmune rheumatic diseases. Autoimmun Rev 14:594–600. https://doi.org/10.1016/j.autrev.2015.02.006
Gohla A (2018) Do metabolic HAD phosphatases moonlight as protein phosphatases? Biochim Biophys Acta 1866:153–166. https://doi.org/10.1016/j.bbamcr.2018.07.007
Hansen SN, Westergaard D, Thomsen MB, Vistesen M, Do KN, Fogh L, Belling KC, Wang J, Yang H, Gupta R, Ditzel HJ, Moreira J, Brunner N, Stenvang J, Schrohl AS (2015) Acquisition of docetaxel resistance in breast cancer cells reveals upregulation of ABCB1 expression as a key mediator of resistance accompanied by discrete upregulation of other specific genes and pathways. Tumour Biol 36:4327–4338. https://doi.org/10.1007/s13277-015-3072-4
Henmi K, Yoshida M, Yoshikawa N, Hirano T (2008) P-glycoprotein functions in peripheral-blood CD4+ cells of patients with systemic lupus erythematosus. Biol Pharm Bull 31:873–878. https://doi.org/10.1248/bpb.31.873
Kalloo S, Aggarwal N, Mohan P, Radhakrishnan J (2013) Lupus nephritis: treatment of resistant disease. Clin J Am Soc Nephrol 8:154–161. https://doi.org/10.2215/CJN.05870612
Kansal A, Tripathi D, Rai MK, Agarwal V (2016) Persistent expression and function of P-glycoprotein on peripheral blood lymphocytes identifies corticosteroid resistance in patients with systemic lupus erythematosus. Clin Rheumatol 35:341–349. https://doi.org/10.1007/s10067-015-3079-7
Kotzin BL (1996) Systemic lupus erythematosus. Cell 85:303–306. https://doi.org/10.1016/S0092-8674(00)81108-3
Kozhakhmetov S, Supiyev A, Kushugulova A, Tynybayeva I, Kossumov A, Utepova L, Saduakhasova S, Shakhabayeva G, Nurgozhin T, Zhumadilov Z (2013) Genotype frequencies of polymorphic MDR1 variants in the Kazakhstani population. Cent Asian J Glob Health 2:118. https://doi.org/10.5195/cajgh.2013.118
Laberge RM, Ambadipudi R, Georges E (2014) P-glycoprotein mediates the collateral sensitivity of multidrug resistant cells to steroid hormones. Biochem Biophys Res Commun 447:574–579. https://doi.org/10.1016/j.bbrc.2014.04.045
Lanata CM, Nititham J, Taylor KE, Chung SA, Torgerson DG, Seldin MF, Pons-Estel BA, Tusie-Luna T, Tsao BP, Morand EF, Alarcon-Riquelme ME, Criswell LA (2018) Genetic contributions to lupus nephritis in a multi-ethnic cohort of systemic lupus erythematous patients. PLoS One 13:e0199003. https://doi.org/10.1371/journal.pone.0199003
Llaudó I, Colom H, Giménez-Bonafé P, Torras J, Caldés A, Sarrias M, Cruzado JM, Oppenheimer F, Sánchez-Plumed J, Gentil MÁ, Ekberg H, Grinyó JM, Lloberas N (2013) Do drug transporter (ABCB1) SNPs and P-glycoprotein function influence cyclosporine and macrolides exposure in renal transplant patients? Results of the pharmacogenomic substudy within the symphony study. Transpl Int 26:177–186. https://doi.org/10.1111/tri.12018
Mu Y, Wang C, Luo J, Zhao W, Zhao X, Li X (2014) Association between P-glycoprotein and lymphocyte subsets in systemic lupus erythematosus. Chinese Remedies & Clinics 101:49–52. https://doi.org/10.11655/zgywylc2014.05.004
Owen A, Chandler B, Bray PG, Ward SA, Hart CA, Back DJ, Khoo SH (2004) Functional correlation of P-glycoprotein expression and genotype with expression of the human immunodeficiency virus type 1 coreceptor CXCR4. J Virol 78(21):12022–12029. https://doi.org/10.1128/JVI.78.21.12022-12029.2004
Ragab SM, Soliman MA (2013) P-glycoprotein-1 functional activity in CD5+CD7+ and CD20+ lymphocytes in systemic lupus erythematosus children: relation to disease activity, complications and steroid response. Egypt J Immunol 20:101–115
Shao SJ (2017) Evolution in epidemiology of lupus nephritis. Chinese Journal of Nephrology, Dialysis & Transplantation 26:562–566. https://doi.org/10.3969/j.issn.1006-298X.2017.06.014
Subhani S, Jamil K, Nirni SS (2015) Association of MDR1 gene (C3435T) polymorphism and gene expression profiling in lung cancer patients treated with platinum-based chemotherapy. Mol Diagn Ther 19:289–297. https://doi.org/10.1007/s40291-015-0154-3
Timlin H, Magder L, Petri M (2017) Clinical outcomes observed among biopsy proven lupus nephritis patients treated with Mycophenolate Mofetil as first-line therapy. Cureus 9:e1907. https://doi.org/10.7759/cureus.1907
Uludag A, Silan C, Atik S, Akurut C, Uludag A, Silan F, Ozdemir O (2014) Relationship between response to colchicine treatment and MDR1 polymorphism in familial Mediterranean fever patients. Genetic Testing & Molecular Biomarkers 18:73. https://doi.org/10.1089/gtmb.2013.0293
Wang M, Han DD, Wang LN, Zhou H, Fan QL, Liu LL, Yang S, Wang J, Liu N (2016) Comparative evaluation of systemic lupus erythematosus two scoring system for lupus nephritis activity. Chinese Journal of Rheumatology 20:391–395. https://doi.org/10.3760/cma.j.issn.1007-7480.2016.06.008
Wang MY, Zeng PY, Zhou YM, Lv JY, Zhu YS, Chen C, Wang QW, Guo CS (2017) Expression of multidrug resistance gene 1 and its expression products on lymphocytes in patients with steroid resistant systemic lupus erythematosus. Chinese. J Immunol 12:1053–1059. https://doi.org/10.13431/j.cnki.immunol.j.20170183
Ward MM, Marx AS, Barry NN (2000) Comparison of the validity and sensitivity to change of 5 activity indices in systemic lupus erythematosus. J Rheumatol 27:664–670
Wincewicz A, Kowalik A, Zięba S, Kopczyński J, Góźdź S, Sulkowski S (2016) Review of prognostic and predictive aspects of mutated TP53 in Wilms' tumor biology with morphological report and molecular analysis of 37-year-old man's nephroblastoma. Polish Journal of Pathology Official Journal of the Polish Society of Pathologists 67:307. https://doi.org/10.5114/pjp.2016.65860
Xuan M, Li H, Fu R, Yang Y, Zhang D, Zhang X, Yang R (2014) Association of ABCB1 gene polymorphisms and haplotypes with therapeutic efficacy of glucocorticoids in Chinese patients with immune thrombocytopenia. Hum Immunol 75:317–321. https://doi.org/10.1016/j.humimm.2014.01.013
Yigitaslan S, Erol K, Cengelli C (2016) The effect of P-glycoprotein inhibition and activation on the absorption and serum levels of cyclosporine and Tacrolimus in rats. Adv Clin Exp Med 25:237–242 https://doi.org/10.17219/acem/35254
Zhang XY, Zhou L, Guo Z, Kong Y, Zhang W, Lu LM, Li X (2018) Regulatory T cells may play a protection role in postoperative pulmonary dysfunction in rheumatic heart disease. J Thorac Dis 10:3196–3205 https://doi.org/10.21037/jtd.2018.05.71
Zhao JJ, Wang D, Yao H, Sun DW, Li HY (2015) CTLA-4 and MDR1 polymorphisms increase the risk for ulcerative colitis: a meta-analysis. World J Gastroenterol 21:10025–10040. https://doi.org/10.3748/wjg.v21.i34.10025
Zhou JQ, Jiang H (2012) Feasibility of SLEDAI-2000 and BILAG-2004 scoring systems for assessing renal disease activity in children with lupus nephritis. Chinese Journal of Contemporary Pediatrics 14:775–779 http://doi.org/www.zgddek.com/EN/abstract/abstract12924.shtml
Zoghbi ME, Mok L, Swartz DJ, Singh A, Fendley GA, Urbatsch IL, Altenberg GA (2017) Substrate-induced conformational changes in the nucleotide-binding domains of lipid bilayer-associated P-glycoprotein during ATP hydrolysis. J Biol Chem 292:20412–20424. https://doi.org/10.1074/jbc.M117.814186
Funding
This study was supported by the Haikou Key Science and Technology Project (Grant Nos. 2013-61, 2015-32).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the ethics committee of Haikou People’s Hospital (Approval no. 2017-082) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhou, J., Lin, H. & Chen, J. Relationship of MDR1 gene polymorphism and P-glycoprotein expression in Chinese refractory lupus nephritis. Biologia 76, 367–374 (2021). https://doi.org/10.2478/s11756-020-00577-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-020-00577-w