Abstract
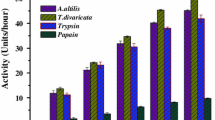
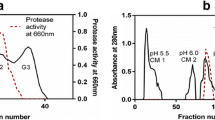
Latex of Maclura spinosa has been used by Kollamalayali tribal community of Western Ghats for curdling of milk. In our previous study we have reported the presence of multiple proteases in the latex, among which the serine protease possesses potent role in hemostasis. With a view to further characterize these proteases the current study was taken up. Maclura spinosa latex contains a serine protease which is resolved using molecular size exclusion column chromatography and an anion exchanger resin in a consecutive manner. The specific activity of the enzyme Maclura spinosa latex protease (MSLP) was found to be 56.15 units/mg and recovery to be 2.68% with a fold purity of 0.41. Being a typical serine protease, MSLP is significantly inhibited by PMSF up to 72.97%. The optimum temperature and pH for the enzyme were found to be 50 °C and 8 respectively. Excision wound healing assay in Swiss albino mice using MSLP, showed accelerated wound closure up to 89.35 ± 1.209% compared to 91.938 ± 1.649% shown by positive control. Further PMSF treated MSLP sample did not show considerable wound healing which confirms exclusive involvement of serine proteases. Examination of biochemical markers viz, hydroxy proline content in healing tissue and catalase activity of fatty tissue also ascertain the potential of MSLP in wound healing. Histopathological studies of healing tissue provide confirmatory evidence with dense collagenation of tissue and fibroblast proliferation rendered by MSLP in the respective treated subjects.










Similar content being viewed by others
Abbreviations
- MSL:
-
Maclura spinosa latex
- MSLP:
-
Maclura spinosa latex protease
- PMSF:
-
phenyl methyl sulphonyl fluoride
- IAA:
-
iodo acetic acid
- DEAE-sephadex:
-
diethyl-aminoethyl- sephadex
- p-DMAB:
-
para-dimethyl amino benzaldehyde
- TRIS:
-
trisaminomethane
- MM plot:
-
Michealis–Menten plot
- LB plot:
-
Lineweaver–Burk plot
- Vmax:
-
Maximal Velocity
- Km:
-
Michaelis Constant
- SDS-PAGE:
-
Sodium dodecyl sulphate polyacrylamide gel electrophoresis
References
Aderounmu A, Omonisi A, Akingbasote J, Makanjuola M, Bejide R, Orafidiya L, Adelusola K (2013) Wound-healing and potential anti-Keloidal properties of the latex of Calotropis Procera (Aiton) Asclepiadaceae in rabbits. Afr J Tradit Complement Altern Med 10:574–579. https://doi.org/10.4314/ajtcam.v10i3.28
Aebi H (1974) Catalase. Pp. 673–684. Methods of enzymatic analysis, Elsevier. https://doi.org/10.1016/B978-0-12-091302-2.50032-3
Angely CJ, Coppola DM (2010) How does long-term odor deprivation affect the olfactory capacity of adult mice? Behav Brain Funct BBF 6:26. https://doi.org/10.1186/1744-9081-6-26
Bonnans C, Chou J, Werb Z (2014) Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol 15:786–801. https://doi.org/10.1038/nrm3904
Bowden LG, Byrne HM, Maini PK, Moulton DE (2016) A morphoelastic model for dermal wound closure. Biomech Model Mechanobiol 15:663–681. https://doi.org/10.1007/s10237-015-0716-7
Bryan N., Ahswin H., Smart N., Bayon Y., Wohlert S. & Hunt J.A. 2012. Reactive oxygen species (ROS)--a family of fate deciding molecules pivotal in constructive inflammation and wound healing. Eur. Cell. Mater. 24: 249–265. Doi: https://doi.org/10.22203/eCM.v024a18
Chakraborti S, Dhalla NS (2017) Proteases in physiology and pathology. Springer, 619 pp. https://doi.org/10.1007/978-981-10-2513-6
De Wet H, Nciki S, van Vuuren SF (2013) Medicinal plants used for the treatment of various skin disorders by a rural community in northern Maputaland, South Africa. J Ethnobiol Ethnomedicine 9:51. https://doi.org/10.1186/1746-4269-9-51
Eagle H (1937) Studies in blood coagulation: V. the coagulation of blood by proteolytic enzymes (TRYPSIN, PAPAIN). J. Gen. Physiol. 20: 543–560. Doi: https://doi.org/10.1085/jgp.20.4.543. https://doi.org/10.1085/jgp.20.4.543
Eming S.A., Martin P. & Tomic-Canic M. 2014. Wound repair and regeneration: mechanisms, signaling, and translation. Sci. Transl. Med. 6: 265sr6. doi: https://doi.org/10.1126/scitranslmed.3009337
Frank S. & Kämpfer H. 2003. Excisional wound healing: an experimental approach. pp. 003–015. Wound healing, Humana Press, New Jersey. doi: https://doi.org/10.1385/1-59259-332-1:003
Geetha S, Lakshmi G, Ranjithakani P (1996) An ethnic method of milk curdling using plants. Anc Sci Life 16:60–61
Gillitzer R, Goebeler M (2001) Chemokines in cutaneous wound healing. J Leukoc Biol 69:513–521
Guo S, DiPietro LA (2010) Factors affecting wound healing. J Dent Res 89:219–229
Hartley BS (1960) Proteolytic enzymes. Annu Rev Biochem 29:45–72. https://doi.org/10.1146/annurev.bi.29.070160.000401
Henry RL, Steiman RH (1968) Mechanisms of hemostasis. Microvasc Res 1:68–82. https://doi.org/10.1016/0026-2862(68)90007-1
Hershko A, Ciechanover A (1992) The ubiquitin system for protein degradation. Annu Rev Biochem 61:761–807. https://doi.org/10.1146/annurev.bi.61.070192.003553
Khan AR, James MN (1998) Molecular mechanisms for the conversion of zymogens to active proteolytic enzymes. Protein Sci Publ Protein Soc 7:815–836. https://doi.org/10.1002/pro.5560070401
Konno K (2011) Plant latex and other exudates as plant defense systems: roles of various defense chemicals and proteins contained therein. Phytochemistry. 72:1510–1530. https://doi.org/10.1016/j.phytochem.2011.02.016
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 227:680–685. https://doi.org/10.1038/227680a0
Landén NX, Li D, Ståhle M (2016) Transition from inflammation to proliferation: a critical step during wound healing. Cell Mol Life Sci 73:3861–3885. https://doi.org/10.1007/s00018-016-2268-0
Lehninger AL (1950) Role of metal ions in enzyme systems. Physiol Rev 30:393–429. https://doi.org/10.1152/physrev.1950.30.3.393
Li J, Chen J, Kirsner R (2007) Pathophysiology of acute wound healing. Clin Dermatol 25:9–18. https://doi.org/10.1016/j.clindermatol.2006.09.007
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
McCarty SM, Cochrane CA, Clegg PD, Percival SL (2012) The role of endogenous and exogenous enzymes in chronic wounds: a focus on the implications of aberrant levels of both host and bacterial proteases in wound healing. Wound Repair Regen 20:125–136. https://doi.org/10.1111/j.1524-475X.2012.00763.x
McDonald JK (1985) An overview of protease specificity and catalytic mechanisms: aspects related to nomenclature and classification. Histochem J 17:773–785. https://doi.org/10.1007/BF01003313
Murata J, Satake M, Suzuki T (1963) Studies on snake venom. XII Distribution of proteinase activities among Japanese and Formosan snake venoms. J Biochem 53:431–443
Muruganandam S., Kamaraj E., Kadirvelmurugan V. & Ravikumar S. 2016. Ethnobotany_of_Gingee_Hills,_Villupuram_District,_ Tamil_Nadu,_India. 4
Nayak S, Nalabothu P, Sandiford S, Bhogadi V, Adogwa A (2006) Evaluation of wound healing activity of Allamanda cathartica. L. and Laurus nobilis. L. Extracts on rats. BMC complement. Altern. Med 6:12. https://doi.org/10.1186/1472-6882-6-12
Neuman RE, Logan MA (1950) The determination of Hydroxyproline. J Biol Chem 184:299–306
Niamke S, Kouame LP, Kouadio JP, Koffi D, Faulet BM, Dabonne S (2006) Effect of some chemicals on the accuracy of protein estimation by the Lowry method. Biokemistri. 17. https://doi.org/10.4314/biokem.v17i2.32591
Pastar I, Stojadinovic O, Yin NC, Ramirez H, Nusbaum AG, Sawaya A, Patel SB, Khalid L, Isseroff RR, Tomic-Canic M (2014) Epithelialization in wound healing: a comprehensive review. Adv Wound Care 3:445–464. https://doi.org/10.1089/wound.2013.0473
Rajesh R, Shivaprasad HV, Gowda CDR, Nataraju A, Dhananjaya BL, Vishwanath BS (2007) Comparative study on plant latex proteases and their involvement in hemostasis: a special emphasis on clot inducing and dissolving properties. Planta Med 73:1061–1067. https://doi.org/10.1055/s-2007-981575
Reinke JM, Sorg H (2012) Wound repair and regeneration. Eur Surg Res Eur Chir Forsch Rech Chir Eur 49:35–43. https://doi.org/10.1159/000339613
Rubinstein HM (1957) Coagulant action of proteolytic enzymes. Nature. 180:1202–1203. https://doi.org/10.1038/1801202a0
Saha SK, Moriya M, Ohinata H, Kuroshima A (1994) Lipid interference with Fluorometric assay of DNA in adipose tissues under various conditions. Jpn J Physiol 44:421–431. https://doi.org/10.2170/jjphysiol.44.421
Shah J, Mohd Y, Omar E, Pai DR, Sood S (2012) Cellular events and biomarkers of wound healing. Indian J Plast Surg Off Publ Assoc Plast Surg India 45:220–228. https://doi.org/10.4103/0970-0358.101282
Sumithira G, Ashma A, Rajamathanky H, Kavya V, Riyas KM (2017) A review on ethanobotanical uses and pharmacology of plecospermum spinosum 6:5
Thakur R, Jain N, Pathak R, Sandhu SS (2011) Practices in Wound Healing Studies of Plants. Evid-Based Complement Altern Med ECAM:2011. https://doi.org/10.1155/2011/438056
Travis BL, Ferguson JH (1951) Proteolytic enzymes and platelets in relation to blood coagulation 1. J Clin Invest 30:112–123. https://doi.org/10.1172/JCI102411
Uday P, Achar RR, Bhat PR, Rinimol RV, Bindu J, Nafeesa Z, Swamy NS (2015) Laticiferous plant proteases in wound care. Int J Pharm Pharm Sci 7:44–49
Upadhyay R (2011) Plant latex: a natural source of pharmaceuticals and pesticides. Int J Green Pharm 5:169. https://doi.org/10.4103/0973-8258.91222
van Hinsbergh VWM, Engelse MA, Quax PHA (2006) Pericellular proteases in angiogenesis and vasculogenesis. Arterioscler Thromb Vasc Biol 26:716–728. https://doi.org/10.1161/01.ATV.0000209518.58252.17
Venkatesh B.K., Achar R.R., Sharanappa P., Priya B.S. & Swamy S.N. 2015. Synergistic caseinolytic activity and differential fibrinogenolytic action of multiple proteases of Maclura spinosa (Roxb. ex Willd.) latex. Pharmacogn Mag 11: S457–S461. doi: https://doi.org/10.4103/0973-1296.168987
Wallace HA, Bhimji SS (2018) Wound healing phases. StatPearls Publishing, Treasure Island (FL), StatPearls
Walsh PN, Ahmad SS (2002) Proteases in blood clotting. Essays Biochem 38:95–111. https://doi.org/10.1042/bse0380095
Wilgus TA, Roy S, McDaniel JC (2013) Neutrophils and wound repair: positive actions and negative reactions. Adv. Wound Care. 2:379–388. https://doi.org/10.1089/wound.2012.0383
Xue M, Le NT, Jackson CJ (2006) Targeting matrix metalloproteases to improve cutaneous wound healing. Expert Opin Ther Targets 10:143–155. https://doi.org/10.1517/14728222.10.1.143
Yariswamy M, Shivaprasad HV, Joshi V, Urs ANN, Nataraju A, Vishwanath BS (2013) Topical application of serine proteases from Wrightia tinctoria R. Br. (Apocyanaceae) latex augments healing of experimentally induced excision wound in mice. J. Ethnopharmacol 7. https://doi.org/10.1016/j.jep.2013.06.056
Acknowledgements
The authors wish to thank the management of JSS Mahavidyapeetha, JSS Science and Technology University and JSS Research Foundation, Mysuru for their continued support in this endeavor. The authors acknowledge, TEQIP-III (Technical Education Quality Improvement Programme), a World-Bank-assisted project under the aegis of the Government of India for their financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest associated with this manuscript.
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution at which the studies were conducted.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kulkarni, V.B., Achar, R.R., Mahadevappa, M. et al. Hemostatically potent small molecular weight serine protease from Maclura spinosa (Roxb. ex Willd.) accelerates healing of subcutaneous dermal wounds in Swiss albino mice. Biologia 75, 139–149 (2020). https://doi.org/10.2478/s11756-019-00322-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-019-00322-y