Abstract
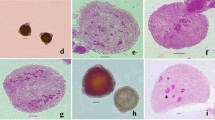
Thalictrum foetidum L. (Ranunculaceae), a morphologically variable and widely distributed species of temperate and alpine Himalayas is worked out cytologically for the first time from India. Earlier studies from outside India were restricted to chromosome counts and karyotypic analysis. We studied the male meiosis, microsporogenesis and pollen viability in the wild accessions from the cold deserts of Lahaul-Spiti, Kinnaur and Pangi Valley of Himachal Pradesh. Present cytomorphological surveys in the species record the existence of two distinct morphotypes involving plant size; colour and size of leaf/leaflet; dentation of leaflet lobes; and degree of leaf pubescence. All the accessions in the two morphovariants share the same meiotic chromosome number (n = 21) and adds a new intraspecific hexaploid cytotype. The accessions show the phenomenon of cytomixis involving transfer of chromatin material among proximate pollen mother cells (PMCs) and associated meiotic abnormalities like, out of plate bivalents, interchromosomal connections, and laggards, bridges and micronuclei at anaphases/telophases. Microsporogenesis results into abnormal sporads (tetrads with micronuclei, dyads, triads and polyads). The products of such sporads resulted into some pollen sterility and pollen grains of heterogeneous sizes. The persistent occurrence of phenomenon of cytomixis and associated meiotic abnormalities and consequently pollen sterility and pollen grains of heterogeneous sizes in the hexaploid cytotype of T. foetidum seems to be under some genetic factors associated with the genome.
Similar content being viewed by others
References
Aswal B. S. & Mehrotra B. N. 1994. Flora of Lahaul-Spiti (a Cold desert in North West Himalaya). Bishan Singh Mahendra Pal Singh, Dehra Dun, India.
Boldrini K.R., Pagliarini M.S. & Valle C.B. 2006. Cell fusion and cytomixis during microsporogenesis in Brachiaria humidicola (Poaceae). South Afr. J. Bot. 72:478–481.
Consolaro M.E.L. & Pagliarini M.S. 1995. Cytomixis in pollen mother cells of Centella asiatica L. Nucleus 38: 80–85.
Davlianidze M.T. 1985. Chromosome numbers in the representatives of the flora from Georgia. Bot. Žurn. (Moscow & Leningrad) 70: 698–700.
De M. & Sharma A.K. 1983. Cytomixis in pollen mother cells of an apomictic ornamental Ervatamia divaricata (Linn.) Alston. Cytologia 48: 201–207.
Friesen N.V. 1991. Chromosome numbers in members of the Ranunculaceae family from Siberia. Bot. Žurn. (Moscow & Leningrad) 76: 905–907.
Hand R. 2000. In: Dobes C. & Vitek E. (eds), Documented Chromosome Numbers Checklist of Austrian Vascular Plants. Verlog des Naturhistorischen Museums Wien, Vienna.
Haroun S.A. 1996. Induced cytomixis and male sterility in pollen mother cells of Hordeum vulgare L. Delta. J. Sci. 20:172–183.
Haroun S.A., Al Shehri A.M. & Al Wadie H.M. 2004. Cytomixis in the microsporogenesis of Vicia faba L. (Fabaceae). Cytologia 69: 7–11.
Holmgren P.K. & Holmgren N.H. 1998. Index Herbariorum: A global directory of public herbaria and associated staff. New York Botanical Garden’s Virtual Herbarium. http://sweetgum.nybg.org/ih/herbariumlist.php.
Krasnikov A.A. 1993. In: Takhtajan A. (ed), Numeri Chromosomatun Magnoliophytonum Florae USSR, Moraceae-Zygophyllaceae. Nauka, Petropoli.
Karsnikov A.A. & Schaulo D.N. 1986. Chromosome numbers of some representatives of families Apiaceae and Ranunculaceae from the South of Siberia. Bot. Žurn. (Moscow & Leningrad) 71: 116–117.
Krogulevich R.E. 1978. Kariologicheskij analiz vidov flory Vostochnogo Sajana. V Flora Pribajkal’ja. Nauka, Novosibirsk, pp.19–48.
Krogulevich R.E. 1984. In: R.E. Krogulevich & T.S. Rostovtseva (eds), Khromosomnye Chisla Tsvetkovykh Rastenii i Sibiri Dal’nego Vostoka. Izdatel’stvo “Nauka”, Sibirskoe Otdelenie, Novosibirsk.
Kuhn E. 1928. Zur Zytologie von Thalictrum. Jahrb. Wiss. Bot. 68: 382–430.
Kuhn E. 1930. Uber Kreuzungen des getrenntgeschlechtigen Thalictrum fendleri mit gemischtgeschlechtigen Arten der gleichen Gattung. Biol. Zentralbl. 50: 79–102.
Kumar P. & Singhal V.K. 2008. Cytology of Caltha palustris L. (Ranunculaceae) from cold regions of Western Himalayas. Cytologia 73: 137–143.
Kumar P., Singhal V.K. & Kaur D. 2008a. Meiotic studies in species from the cold deserts of Lahaul-Spiti and adjoining areas (Northwest Himalayas). Cytologia 73: 463–470.
Kumar P., Singhal V.K. & Kaur J. 2008b. Cytomixis induced meiotic abnormalities in pollen mother cells of Clematis flammula L. (Ranunculaceae). Cytologia 73: 381–385.
Kumar P., Singhal V.K., Kaur D. & Kaur S. 2010. Cytomixis and associated meiotic abnormalities affecting pollen fertility in Clematis orientalis. Biol. Plantarum 54: 181–184.
Kuzmanova B., Evstatieva L. & Georgieva S. 1981. Chromosome number reports LXXII. Taxon 30: 702.
Kuzmanova B., Robeva P. & Georgieva S. 1987. Cytotaxonomic study of Bulgarian species of Thalictrum L. Fitologia (Sofia) 33: 14–21.
Langlet O.F.J. 1927. Beitrage zur Zytologie der Ranunculazeen. Sven. Bot. Tidskr. 21: 1–17.
Löve á. & Löve D. 1982. IOPB chromosome number reports LXXV. Taxon 31: 344–360.
Magulaev A. Vu. 1984. Cytotaxonomic study in some flowering plants of the North Caucasus. Bot. Zhurn. (Moscow & Leningrad) 69: 511–517.
Malakhova L.A. 1990. Kariologocheskij analiz prirodnykh populjacij redkich i ischezajushchikh rastenij na juge Tomskoj Oblasti. Biull. Glavn. Bot. Sada. 155: 60–66.
Pierozzi N.I. & Benatti R. 1998. Cytological analysis in the microsporogenesis of ramie Boehmeria nivea Gaud. (Urticaceae) and the effect of colchicine on the chiasma frequency. Cytologia 63: 213–221.
Rostovtseva T.S. 1981. Chromosome numbers of some species of the family Ranunculaceae II. Bot. Zhurn. (Moscow & Leningrad) 66: 1751–1755.
Rostovtseva T.S. 1984. Khromosomnye Chisla Tsvetkovykh Rastenii i Sibiri Dal’nego Vostoka.
Singh H. & Sharma M. 2006. Flora of Chamba District (Himachal Pradesh). Bishan Singh Mahendra Pal Singh, Dehra Dun, India.
Singhal V.K., Kaur D. & Kumar P. 2008. Effect of cytomixis on the pollen size in ’seabuckthorn’ (Hippophae rhamnoides L., Elaeagnaceae). Cytologia 73: 167–172.
Singhal V.K., Kumar P., Kaur D. & Rana P.K. 2009a. Chromatin transfer during male meiosis resulted into heterogeneous sized pollen grains in Anemone rivularis Buch.-Ham. ex DC. from Indian cold deserts. Cytologia 74: 229–234.
Singhal V.K., Kaur S., Kaur D. & Kumar P. 2009b. New detection of haploid chromosomes, pollen size and sterility in Lychnis indica Benth. var. fimbriata Wall. Chrom. Bot. 4: 53–56
Singhal V.K. & Kumar P. 2008a. Impact of cytomixis on meiosis, pollen viability and pollen size in wild populations of Himalayan poppy (Meconopsis aculeata Royle). J. Biosci. 33: 371–380.
Singhal V.K. & Kumar P. 2008b. Cytomixis during microsporogenesis in the diploid and tetraploid cytotypes of Withania somnifera (L.) Dunal, 1852 (Solanaceae). Comp. Cytogenet. 2: 85–92.
Singhal V.K., Kaur S. & Kumar P. 2010. Aberrant male meiosis, pollen sterility and heterogenous sized pollen grains in Clematis montana Buch.-Ham. ex DC. from Dalhousie hills, Himachal Pradesh. Cytologia 75: 31–36.
Tyagi B. R. 2003. Cytomixis in pollen mother cells of spearmint (Mentha spicata L.). Cytologia 68: 67–73.
Yurtsev B.A. & Zhukova P.G. 1982. Chromosome numbers of some plants of the northeastern Yakutia (the drainage of the Indigirka River in its middle reaches). Bot. Zhurn. (Moscow & Leningrad) 67: 778–787.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singhal, V.K., Rana, P.K., Kumar, P. et al. Persistent occurrence of meiotic abnormalities in a new hexaploid cytotype of Thalictrum foetidum from Indian cold deserts. Biologia 66, 458–464 (2011). https://doi.org/10.2478/s11756-011-0033-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11756-011-0033-2