Abstract
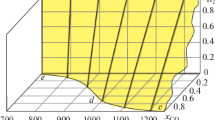
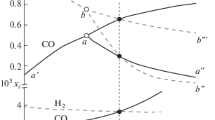
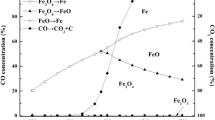
The reduction of chromium, nickel, and manganese oxides by hydrogen, CO, CH4, and model syngas (mixtures of CO + H2 or H2 + CO + CO2) and oxidation by water vapor has been studied from the thermodynamic and chemical equilibrium point of view. Attention was concentrated not only on the convenient conditions for reduction of the relevant oxides to metals or lower oxides at temperatures in the range 400–1000 K, but also on the possible formation of soot, carbides, and carbonates as precursors for the carbon monoxide and carbon dioxide formation in the steam oxidation step. Reduction of very stable Cr2O3 to metallic Cr by hydrogen or CO at temperatures of 400–1000 K is thermodynamically excluded. Reduction of nickel oxide (NiO) and manganese oxide (Mn3O4) by hydrogen or CO at such temperatures is feasible. The oxidation of MnO and Ni by steam and simultaneous production of hydrogen at temperatures between 400 and 1000 K is a difficult step from the thermodynamics viewpoint. Assuming the Ni—NiO system, the formation of nickel aluminum spinel could be used to increase the equilibrium hydrogen yield, thus, enabling the hydrogen production via looping redox process. The equilibrium hydrogen yield under the conditions of steam oxidation of the Ni—NiO system is, however, substantially lower than that for the Fe—Fe3O4 system. The system comprising nickel ferrite seems to be unsuitable for cyclic redox processes. Under strongly reducing conditions, at high CO concentrations/partial pressures, formation of nickel carbide (Ni3C) is thermodynamically favored. Pressurized conditions during the reduction step with CO/CO2 containing gases enhance the formation of soot and carbon-containing compounds such as carbides and/or carbonates.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Messerschmitt, A., Ger. DE 266863 (1911).
Fraser, S. and Hacker, V., Sponge Iron Process for Manned Space Exploration, Final Project Report, Technical Officer: Dr. Tiziana Pipoli, Ariadna Contract No. 18461/04/NL/MV, 2005.
Hacker, V., Fankhauser, R., Faleschini, G., Fuchs, H., Friedrich,, K., Muhr, M., and Kordeschet, K., J. Power Sources 86, 531 (2000).
Hacker, V., Faleschini, G., Fuchs, H., Fankhauser, R., Simader, G., Ghaemi, M., Spreitz, B., and Friedrichet, K., J. Power Sources 71, 226 (1998).
Lundberg, M., Int. J. Hydrogen Energy 18, 369 (1993).
Inoue, M., Hasegawa, N., Uehara, R., Gokon, N., Kaneko, H., and Tamaura, Y., Sol. Energy 76, 309 (2004).
Adanez, J., de Diego, L. F., Garcia-Labiano, F., Gayan, P., Abad, A., and Palacios, J. M., Energy Fuels 18, 371 (2004).
Son, S. R. and Kim, S. D., Ind. Eng. Chem. Res. 45, 2689 (2006).
Cao, Y. and Pan, W.-P., Energy Fuels 20, 1836 (2006).
Mattisson, T., Johansson, M., and Lyngfelt, A., Fuel 85, 736 (2006).
Hacker, V., J. Power Sources 118, 311 (2003).
Otsuka, K., Kaburagi, T., Yamada, C., and Takenaka, S., J. Power Sources 122, 111 (2003).
Takenaka, S., Sou, V. T. D., and Otsuka, K., Energy Fuels 18, 820 (2004).
Urasaki, K., Tanimoto, N., Hayashi, T., Sekine, Y., Kikuchi, E., and Matsukata, M., Appl. Catal., A 228, 143 (2005).
Hui, W., Takenaka, S., and Otsuka, K., Int. J. Hydrogen Energy 31, 1732 (2006).
Cho, P., Mattisson, T., and Lyngfeld, A., Ind. Eng. Chem. Res. 44, 668 (2005).
Barin, I. and Knacke, O., Thermochemical Data of Pure Substances, 3rd Edition. VCH, Weinheim, 1995.
Tseng, W. J., Hsu, C.-K., Chi, C.-C., and Teng, K.-H., Mater. Lett. 52, 313 (2002).
Kenfack, F. and Langbein, H., Cryst. Res. Technol. 41, 748 (2006).
Bučko, M. and Haberko, K., J. Eur. Ceram. Soc., in press.
Bolt, P. H., Habraken, F. H. P. M., and Geus, J. W., J. Solid State Chem. 135, 59 (1998).
Twigg, M. V. and Richardson, J. T., Appl. Catal., A 190, 61 (2000).
Readman, J. E., Olafsen, A., Smith, J. B., and Blom, R., Energy Fuels 20, 1382 (2006).
Svoboda, K., Slowinski, G., Rogut, J., and Siewiorek, A., 17th International Congress of Chemical and Process Engineering CHISA, Prague, 2006.
Villa, R., Cristiani, C., Groppi, G., Lietti, L., Forzatti, P., Cornaro, U., and Rossini, S., J. Mol. Catal. A: Chem. 204, 637 (2003).
Li, G. H., Hu, L. J., and Hill, J. M., Appl. Catal., A 301, 16 (2006).
Stobbe, E. R., de Boer, B. A., and Geus, J. W., Catal. Today 47, 161 (1999).
Terayama, K., Ikeda, M., and Taniguchi, M., Trans. Jpn. Inst. Met. 24, 24 (1983).
Malavasi, L., Ghigna, P., Chiodelli, G., Maggi, G., and Flor, G., J. Solid State Chem. 166, 171 (2002).
Christel, L., Pierre, A., and Abel, D. A. M. R., Thermochim. Acta 306, 51 (1997).
Guillemet-Fritsch, S., Navrotsky, A., Tailhades, P., Coradin, H., and Wang, M., J. Solid State Chem. 178, 106 (2005).
Jablonski, G. A., Geurts, F. W., Sacco, A., and Biederman, R. R., Carbon 30, 87 (1992).
Grabke, H. J. and Wolf, I., Mater. Sci. Eng. 87, 23 (1987).
Mallon, C. and Kendall, K., J. Power Sources 145, 154 (2005).
Li, Y., Zhang, B. C., Xie, X. W., Liu, J. L., Xu, Y. D., and Shen, W. J., J. Catal. 238, 412 (2006).
Zeng, Z. and Natesan, K., Solid State Ionics 167, 9 (2004).
Author information
Authors and Affiliations
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Svoboda, K., Siewiorek, A., Baxter, D. et al. Thermodynamic possibilities and constraints of pure hydrogen production by a chromium, nickel, and manganese-based chemical looping process at lower temperatures. Chem. Pap. 61, 110–120 (2007). https://doi.org/10.2478/s11696-007-0007-6
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.2478/s11696-007-0007-6