Abstract
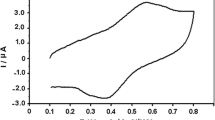
The characteristics of anodic electrochemical dissolution of chalcopyrite (CuFeS2) powder in hydrochloric acid medium with sodium chloride have been studied. Cyclic voltammetry and chronopotentiometry of immobilized microparticles using paraffin-impregnated graphite electrode was employed. Present work is focused on electrochemical identification of chalcopyrite cathodic and anodic reaction products within the potential range of −0.7 to +0.8 V (vs. SCE) in hydrochloric acid solution containing sodium chloride and/or copper(II) chloride.
Similar content being viewed by others

References
Hiroyoshi, N., Kuroiwa, S., Miki, H., Tsunekawa, M., and Hirajima, T., Hydrometallurgy 74, 103 (2004).
Bartlet, R. W., Willson, D. B., Savage, B. J., and Weseley, R. J., in Hydrometallurgy Reactor Design and Kinetics, pp. 227. The American Institute of Mining, Metallurgical and Petroleum Engineers (AIME), New York, 1986.
Arce, M. E. and Gonzáles, I., Int. J. Miner. Processes 67, 17 (2002).
Antonijević, M. M. and Bogdanović, G. D., Hydrometallurgy 73, 254 (2004).
Lu, Z. Y., Jeffrey, M. I., and Lawson, F., Hydrometallurgy 56, 189 (2000).
Vargas, T. and Inman, D., J. Electroanal. Chem. 119, 25 (1981).
Lázaro, I., Martínez-Medina, N., Rodríguez, I., Arce, E., and Gonzáles, I., Hydrometallurgy 38, 277 (1995).
Elsherief, A. E., Miner. Eng. 15, 5 (2002).
Hiroyoshi, N., Miki, H., Hirajima, T., and Tsunekawa, M., Hydrometallurgy 57, 31 (2000).
Mikhlin, Y. L., Tomashevich, Y. V., Asanov, I. P., Okotrub, A. V., Varnek, V. A., and Vyalikh, D. V., Appl. Surf. Sci. 225, 395 (2004).
Lamache, M. and Bauer, D., Anal. Chem. 51, 1320 (1979).
McMillan, R. S., MacKinnon, D. J., and Dutrizac, J. E., J. Appl. Electrochem. 12, 743 (1982).
Hiroyoshi, N., Miki, H., Hirajima, T., and Tsunekawa, M., Hydrometallurgy 60, 185 (2001).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pikna, L., Lux, L. & Grygar, T. Electrochemical dissolution of chalcopyrite studied by voltammetry of immobilized microparticles. Chem. Pap. 60, 293–296 (2006). https://doi.org/10.2478/s11696-006-0051-7
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.2478/s11696-006-0051-7