Abstract
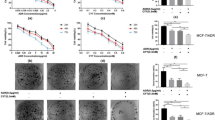
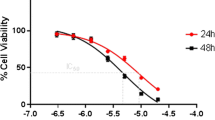
2DG causes cytotoxicity in cancer cells by disrupting thiol metabolism while Doxorubicin (DOX) induces cytotoxicity in tumor cells by generating reactive oxygen species (ROS). Here we examined the combined cytotoxic action of 2DG and DOX in rapidly dividing T47D breast cancer cells vs. slowly growing MCF-7 breast cancer cells. T47D cells exposed to the combination of 2DG/DOX significantly decreased cell survival compared to controls, while 2DG/DOX had no effect on MCF-7 cells. 2DG/DOX also disrupted the oxidant status of T47D treated cells, decreased intracellular total glutathione and increased glutathione disulfide (%GSSG) compared to MCF-7 cells. Lipid peroxidation increased in T47D cells treated with 2DG and/or DOX, but not in MCF-7 cells. T47D cells were significantly protected by NAC, indicating that the combined treatment exerts its action by increasing ROS production and disrupting antioxidant stores. When we inhibited glutathione synthesis with BSO, T47D cells became more sensitive to 2DG/DOX-induced cytotoxicity, but NAC significantly reversed this cytotoxic effect. Finally, 2DG/DOX, and BSO significantly increased the %GSSG in T47D cells, an effect which was also reversed by NAC. Our results suggest that exposure of rapidly dividing breast cancer cells to 2DG/DOX enhances cytotoxicity via oxidative stress and via disruptions to thiol metabolism.
Similar content being viewed by others
References
Weber G. Enzymology of cancer cells (second of two parts), N. Engl. J. Med., 1977, 296, 541–551
Warburg O. On the Origin of Cancer Cells, Science, 1956, 123, 309–314
Weber G. Enzymology of cancer cells (first of two parts), N. Engl. J. Med., 1977, 296, 486–492
Lee Y.J., Galoforo S.S., Berns C.M., Chen J.C., Davis B.H., Sim J.E., et al. Glucose Deprivation-induced Cytotoxicity and Alterations in Mitogen-activated Protein Kinase Activation Are Mediated by Oxidative Stress in Multidrug-resistant Human Breast Carcinoma Cells, J. Biol. Chem., 1998, 273, 5294–5299
Spitz D.R., Sim J.E., Ridnour L.A., Galoforo S.S., Lee Y.J. Glucose Deprivation-Induced Oxidative Stress in Human Tumor Cells: A Fundamental Defect in Metabolism?, Ann. N. Y. Acad Sci., 2000, 899, 349–362
Galoforo S.S., Berns C.M., Erdos G., Corry P.M., Lee Y.J. Hypoglycemia-induced AP-1 transcription factor and basic fibroblast growth factor gene expression in multidrug resistant human breast carcinoma MCF-7/ADR cells, Mol. Cell. Biochem., 1996, 155, 163–171
Liu X., Gupta A.K., Corry P.M., Lee Y.J. Hypoglycemia-induced c-Jun Phosphorylation Is Mediated by c-Jun N-terminal Kinase 1 and Lyn Kinase in Drug-resistant Human Breast Carcinoma MCF-7/ADR Cells, J. Biol. Chem., 1997, 272, 11690–11693
Wick A.N., Drury D.R., Nakada H.I., Wolfe J.B. Localization of The Primary Metabolic Block Produced by 2-Deoxyglucose, J. Biol. Chem., 1957, 224, 963–969
Kern K., Norton J. Inhibition of established rat fibrosarcoma growth by the glucose antagonist 2-deoxy-D-glucose, Surgery, 1987, 102, 380–385
Dwarkanath B.S., Zolzer F., Chandana S., Bauch T., Adhikari J.S., Muller W.U., et al. Heterogeneity in 2-deoxy-D-glucose-induced modifications in energetics and radiation responses of human tumor cell lines, Int. J. Radiat. Oncol. Biol. Phys., 2001, 50, 1051–1061
Lin X., Zhang F., Bradbury C.M., Kaushal A., Li L., Spitz D.R., et al. 2-Deoxy-D-Glucose-induced Cytotoxicity and Radiosensitization in Tumor Cells Is Mediated via Disruptions in Thiol Metabolism, Cancer Res., 2003, 63, 3413–3417
Jain V.K., Kalia V.K., Sharma R., Maharajan V., Menon M. Effects of 2-deoxy-D-glucose on glycolysis, proliferation kinetics and radiation response of human cancer cells, Int. J. Radiat. Oncol. Biol. Phys., 1985, 11, 943–950
Burger C., Wick M., Brusselbach S., Muller R. Differential induction of ‘metabolic genes’ after mitogen stimulation and during normal cell cycle progression, J. Cell. Sci., 1994, 107, 241–252
Perez J.X., Roig T., Manzano A., Dalmau M., Boada J., Ventura F., et al. Overexpression of fructose 2,6-bisphosphatase decreases glycolysis and delays cell cycle progression, Am. J. Physiol. Cell Physiol., 2000, 279, C1359–C1365
Takanashi S., Bachur N.R. Adriamycin metabolism in man. Evidence from urinary metabolites, Drug Metab. Dispos., 1976, 4, 79–87
Quiles J.L., Huertas J.R., Battino M., Mataix J., Ramírez-Tortosa M.C. Antioxidant nutrients and adriamycin toxicity, Toxicology, 2002, 180, 79–95
Kong Q., Lillehei K.O. Antioxidant inhibitors for cancer therapy, Med. Hypotheses, 1998, 51, 405–409
Gille L., Nohl H. Analyses of the Molecular Mechanism of Adriamycin-Induced Cardiotoxicity, Free Radic. Biol. Med., 1997, 23, 775–782
Zhou S., Starkov A., Froberg M.K., Leino R.L., Wallace K.B. Cumulative and Irreversible Cardiac Mitochondrial Dysfunction Induced by Doxorubicin, Cancer Res, 2001, 61, 771–777
Shenoy M.A., Singh B.B. Non-nitro Radiation Sensitizers, Int. J. Radiat. Biol., 1985, 48, 315–326
Ahmad I.M., Abdalla M.Y., Aykin-Burns N., Simons A.L., Oberley L.W., Domann F.E., et al. 2-Deoxyglucose combined with wild-type p53 overexpression enhances cytotoxicity in human prostate cancer cells via oxidative stress, Free Radic. Biol. Med., 2008, 44, 826–834
Aykin-burns N., Ahmad I.M., Zhu Y., Oberley L.W., Spitz D.R. Increased levels of superoxide and H2O2 mediate the differential susceptibility of cancer cells versus normal cells to glucose deprivation, Biochem. J., 2009, 418, 29–37
Griffith O.W. Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine, Anal. Biochem., 1980, 106, 207–212
Anderson M. Determination of glutathione and glutathione disulfide in biological samples, Meth. Enzymol., 1985, 113, 548–554
Mosmann T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays, J. Immunol. Meth., 1983, 65, 55–63
Ohkawa H., Ohishi N., Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction, Anal. Biochem., 1979, 95, 351–358
Schafer F.Q., Buettner G.R. Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple, Free Radic. Biol. Med., 2001, 30, 1191–1212
Sies H., Cadenas E., Symons M.C.R., Scott G. Oxidative Stress: Damage to Intact Cells and Organs, Philos. Trans. R. Soc. Lond. B Biol. Sci., 1985, 311, 617–631
Oberley LW., Buettner G.R. Role of Superoxide Dismutase in Cancer: A Review, Cancer Res., 1979, 39, 1141–1149
Suzuki M., O’Dea J.D., Suzuki T., Agar N.S. 2-deoxyglucose as a substrate for glutathione regeneration in human and ruminant red blood cells, Comp. Biochem. Physiol. Part B: Bioch. Mol. Biol., 1983, 75, 195–197
Landau B.R., Lubs H.A. Animal responses to 2-Deoxy-d-Glucose administration, Proc. Soc. Exp. Biol. Med., 1958, 99, 124–127
Andringa K.K., Coleman MC., Aykin-Burns N., Hitchler M.J., Walsh S.A., Domann F.E., et al. Inhibition of Glutamate Cysteine Ligase Activity Sensitizes Human Breast Cancer Cells to the Toxicity of 2-Deoxy-D-Glucose, Cancer Res., 2006, 66, 1605–1610
Mohanti B.K., Rath G.K., Anantha N., Kannan V., Das B.S., Chandramouli B.A.R, et al. Improving cancer radiotherapy with 2-deoxy—glucose: phase I/II clinical trials on human cerebral gliomas, Int. J. Radiat. Oncol., Biol., Phys., 1996, 35, 103–111
Simons A.L., Ahmad I.M., Mattson D.M., Dornfeld K.J., Spitz D.R. 2-Deoxy-D-Glucose Combined with Cisplatin Enhances Cytotoxicity via Metabolic Oxidative Stress in Human Head and Neck Cancer Cells, Cancer Res., 2007, 67, 3364–3370
Singh G., Lakkis CL., Laucirica R., Epner D.E. Regulation of prostate cancer cell division by glucose, J. Cell. Physiol., 1999, 180, 431–438
Lo C-h., Cristofalo V.J., Morris H.P., Weinhouse S. Studies on Respiration and Glycolysis in Transplanted Hepatic Tumors of the Rat, Cancer Res., 1968, 28, 1–10
Wang T., Marquardt C., Foker J. Aerobic glycolysis during lymphocyte proliferation, Nature, 1976, 261, 702–705
Szatrowski T.P., Nathan C.F. Production of Large Amounts of Hydrogen Peroxide by Human Tumor Cells, Cancer Res., 1991, 51, 794–798
Maschek G., Savaraj N., Priebe W., Braunschweiger P., Hamilton K., Tidmarsh G.F., et al. 2-Deoxy-D-glucose Increases the Efficacy of Adriamycin and Paclitaxel in Human Osteosarcoma and Non-Small Cell Lung Cancers In Vivo, Cancer Res., 2004, 64, 31–34
Wold L.E., Aberle N.S., Ren J. Doxorubicin induces cardiomyocyte dysfunction via a p38 MAP kinase-dependent oxidative stress mechanism, Cancer Detect. Prev., 2005, 29, 294–299
Moadel R., Nguyen A., Lin E., Lu P., Mani J., Blaufox M.D., et al. Positron emission tomography agent 2-deoxy-2-[18F]fluoro-D-glucose has a therapeutic potential in breast cancer, Breast Cancer Res., 2003, 5, R199–R205
Tyagi A.K., Singh R.P., Agarwal C., Chan D.C.F., Agarwal R. Silibinin Strongly Synergizes Human Prostate Carcinoma DU145 Cells to Doxorubicininduced Growth Inhibition, G2-M Arrest, and Apoptosis, Clin. Cancer Res., 2002, 8, 3512–3519
Bailey H.H. L-S,R-buthionine sulfoximine: historical development and clinical issues, Chem. Biol. Interact., 1998, 111-112, 239–254
Hales B.F., Brown H. The effect of in vivo glutathione depletion with buthionine sulfoximine on rat embryo development, Teratology, 1991, 44, 251–257
Meister A., Anderson M.E. Glutathione, Annu. Rev. Biochem., 1983, 52, 711–760
Watanabe T., Sagisaka H., Arakawa S., Shibaya Y., Watanabe M., Igarashi I,, et al. A Novel Model of Continuous Depletion of Glutathione in Mice Treated With L-Buthionine (S,R)-Sulfoximine, J. Toxicol. Sci., 2003, 28, 455–469
Mattson D.M., Ahmad I.M., Dayal D., Parsons A.D., Aykin-Burns N., Li L., et al. Cisplatin combined with zidovudine enhances cytotoxicity and oxidative stress in human head and neck cancer cells via a thiol-dependent mechanism, Free Radic. Biol. Med., 2009, 46, 232–237
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ahmad, I.M., Mustafa, E.H., Mustafa, N.H. et al. 2DG enhances the susceptibility of breast cancer cells to doxorubicin. cent.eur.j.biol. 5, 739–748 (2010). https://doi.org/10.2478/s11535-010-0060-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11535-010-0060-y