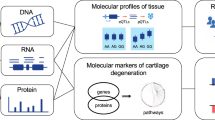
Abstract
The post-genomic era of functional genomics and target validation will allow us to narrow the bridge between clinically correlative data and causative data for complex diseases, such as arthritis, for which the etiological agent remains elusive. The availability of human and other annotated genome sequences, and parallel developments of new technologies that allow analysis of minute amounts of human and animal cells (peripheral blood cells and infiltrating cells) and tissues (synovium and cartilage) under different pathophysiological conditions, has facilitated high-throughput gene mining approaches that can generate vast amounts of clinically correlative data. Characterizing some of the correlative/causative genes will require reverting to the hypothesis-driven, low throughput method of complementary experimental biology using genomic approaches as a tool. This will include in silico gene expression arrays, genome-wide scans, comparative genomics using various animal models (such as rodents and zebrafish), bioinformatics and a team of well trained translational scientists and physicians.
For the first time, the ‘genomic tools’ will allow us to analyze small amounts of surgical samples (such as needle biopsies) and clinical samples in the context of the whole genome. Preliminary genomic analysis in osteoarthritis has already resurrected the debate on the semantic issues in the definition of inflammation. Further analyses will not only facilitate the development of unbiased hypotheses at the molecular level, but also assist us in the identification and characterization of novel targets and disease markers for pharmacological intervention, gene therapy, and diagnosis.






Similar content being viewed by others
References
McCarty DJ. Differential diagnosis of arthritis: analysis of signs and symptoms. In: Koopman WJ, editor. Arthritis and allied conditions: a textbook of rheumatology. Philadelphia (PA): Lippincott, 1998: 39–50
Riley JH, Allan CJ, Lai E, et al. The use of single nucleotide polymorphisms in the isolation of common disease genes. Pharmacogenomics 2000; 1: 39–47
Jimenez-Sanchez G, Childs B, Valle D. Human disease genes. Nature 2001; 409: 853–5
Peltonen L, McKusick VA. Genomics and medicine: dissecting human disease in the postgenomic era. Science 2001; 291: 1224–9
Frary A, Nesbitt TC, Grandillo S, et al. Fw2.2: a quantitative trait locus key to the evolution of tomato fruit size. Science 2000; 289: 85–8
Masinde GL, Li X, Gu W, et al. Identification of wound healing/regeneration quantitative trait loci (QTL) at multiple time points that explain seventy percent of variance in (MRL/MpJ and SJL/J) mice F2 population. Genome Res 2001; 11: 2027–33
Brockmann GA, Bevova MR. Using mouse models to dissect the genetics of obesity. Trends Genet 2002; 18: 367–76
Korstanje R, Paigen B. From QTL to gene: the harvest begins. Nat Genet 2002; 31: 235–6
Hamet P, Tremblay J. Genetic determinants of the stress response in cardiovascular disease. Metabolism 2002; 51: 15–24
Griffiths MM, Wang J, Joe B, et al. Identification of four new quantitative trait loci regulating arthritis severity and one new quantitative trait locus regulating autoantibody production in rats with collagen-induced arthritis. Arthritis Rheum 2000; 43: 1278–89
Joe B, Remmers EF, Dobbins DE, et al. Genetic dissection of collagen-induced arthritis in Chromosome 10 quantitative trait locus speed congenic rats: evidence for more than one regulatory locus and sex influences. Immunogenetics 2000; 51: 930–44
Gulko PS, Kawahito Y, Remmers EF, et al. Identification of a new non-major histocompatibility complex genetic locus on chromosome 2 that controls disease severity in collagen-induced arthritis in rats. Arthritis Rheum 1998; 41: 2122–31
Kawahito Y, Cannon GW, Gulko PS, et al. Localization of quantitative trait loci regulating adjuvant-induced arthritis in rats: evidence for genetic factors common to multiple autoimmune diseases. J Immunol 1998; 161: 4411–9
Patel IR, Attur MG, Patel RN, et al. TNF-α convertase from human arthritis-affected cartilage: isolation of CDNA by differential display, expression of the active enzyme, and regulation of TNF-α. J Immunol 1998; 160: 4570–9
Attur MG, Dave MN, Stuchin S, et al. Osteopontin: an intrinsic inhibitor of inflammation in cartilage. Arthritis Rheum 2001; 44: 578–84
Hu SI, Carozza M, Klein M, et al. Human HtrA, an evolutionarily conserved serine protease identified as a differentially expressed gene product in osteoarthritic cartilage. J Biol Chem 1998; 273: 34406–12
Heller RA, Schena M, Chai A, et al. Discovery and analysis of inflammatory disease-related genes using CDNA microarrays. Proc Natl Acad Sci U S A 1997; 94: 2150–5
Khan IM, Salter DM, Bayliss MT, et al. Expression of clusterin in the superficial zone of bovine articular cartilage. Arthritis Rheum 2001; 44: 1795–9
Kumar S, Connor JR, Dodds RA, et al. Identification and initial characterization of 5000 expressed sequenced tags (ESTs) each from adult human normal and osteoarthritic cartilage CDNA libraries. Osteoarthritis Cartilage 2001; 9: 641–53
Justen HP, Grunewald E, Totzke G, et al. Differential gene expression in synovium of rheumatoid arthritis and osteoarthritis. Mol Cell Biol Res Commun 2000; 3: 165–72
Seki T, Selby J, Haupl T, et al. Use of differential subtraction method to identify genes that characterize the phenotype of cultured rheumatoid arthritis synoviocytes. Arthritis Rheum 1998; 41: 1356
Amin AR. Dissecting human musculoskeletal diseases in the post-genomic era of medicne: following the scent in search for the the etiological gene(s). In: Rosier RN, Evans CH, editor. Molecular biology in orthopaedic. Rosemont (IL): American Academy of Orthopaedic Surgeons, 2003: 3–12
Attur MG, Dave MN, Tsunoyama K, et al. A “system biology” approach to bioinformatics and functional genomics in complex human diseases: arthritis. Curr Issues Mol Biol 2002; 4: 129–46
Attur MG, Bingham III CO, Dave MN, et al. Model protocol to study pharmacogenomics in inflammatory diseases: human rheumatoid arthritis. Drug Dev Res 2000; 49: 29–33
Rioux JD, Daly MJ, Silverberg MS, et al. Genetic variation in the 5q31 cytokine gene cluster confers susceptibility to Crohn disease. Nat Genet 2001; 29: 223–8
Ingvarsson T, Stefansson SE, Gulcher JR, et al. A large icelandic family with early osteoarthritis of the hip associated with a susceptibility locus on chromosome 16p. Arthritis Rheum 2001; 44: 2548–55
Jawaheer D, Seldin MF, Amos CI, et al. A genomewide screen in multiplex rheumatoid arthritis families suggests genetic overlap with other autoimmune diseases. Am J Hum Genet 2001; 68: 927–36
Knight J. When the chips are down. Nature 2001; 410: 860–1
Hastie T, Tibshirani R, Eisen MB, et al. ‘Gene shaving’ as a method for identifying distinct sets of genes with similar expression patterns. Genome Biol 2000; 1: RESEARCH0003. Epub 2000 aug 04.
Bassett Jr DE, Eisen MB, Boguski MS. Gene expression informatics: it’s all in your mine. Nat Genet 1999; 21(1 Suppl.): 51–5
Rivera MC, Jain R, Moore JE, et al. Genomic evidence for two functionally distinct gene classes. Proc Natl Acad Sci U S A 1998; 95: 6239–44
Eisen MB, Spellman PT, Brown PO, et al. Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci U S A 1998; 95: 14863–8
Attur MG, Dave M, Akamatsu M, et al. Osteoarthritis or osteoarthrosis: the definition of inflammation becomes a semantic issue in the genomic era of molecular medicine. Osteoarthritis Cartilage 2002; 10: 1–4
Attur MG, Dave MN, Leung MY, et al. Functional genomic analysis of type II IL-1β decoy receptor: potential for gene therapy in human arthritis and inflammation. J Immunol 2002; 168: 2001–10
Wang FL, Connor JR, Dodds RA, et al. Differential expression of Egr-1 in osteoarthritic compared to normal adult human articular cartilage. Osteoarthritis Cartilage 2000; 8: 161–9
Tetlow LC, Adlam DJ, Woolley DE. Matrix metalloproteinase and proinflammatory cytokine production by chondrocytes of human osteoarthritic cartilage: associations with degenerative changes. Arthritis Rheum 2001; 44: 585–94
Freemont AJ, Hampson V, Tilman R, et al. Gene expression of matrix metalloproteinases 1, 3, and 9 by chondrocytes in osteoarthritic human knee articular cartilage is zone and grade specific. Ann Rheum Dis 1997; 56: 542–9
Goldring MB, Birkhead JR, Suen L-F, et al. Interleukin-1β-modulated gene expression in immortalized human chondrocytes. J Clin Invest 1994; 94: 2307–16
Hauselmann HJ, Aydelotte MB, Schumacher BL, et al. Synthesis and turnover of proteoglycans by human and bovine adult articular chondrocytes cultured in alginate beads. Matrix 1992; 12: 116–29
Lemare F, Steimberg N, Le Griel C, et al. Dedifferentiated chondrocytes cultured in alginate beads: restoration of the differentiated phenotype and of the metabolic responses to interleukin-1beta. J Cell Physiol 1998; 176: 303–13
Perka C, Spitzer RS, Lindenhayn K, et al. Matrix-mixed culture: new methodology for chondrocyte culture and preparation of cartilage transplants. J Biomed Mater Res 2000; 49: 305–11
Yang BB, Zhang Y, Cao L, et al. Aggrecan and link protein affect cell adhesion to culture plates and to type II collagen. Matrix Biol 1998; 16: 541–61
Aigner T, Dertinger S, Vornehm SI, et al. Phenotypic diversity of neoplastic chondrocytes and extracellular matrix gene expression in cartilaginous neoplasms. Am J Pathol 1997; 150: 2133–41
Haas C, Aicher WK, Dinkel A, et al. Characterization of SV40T antigen immortalized human synovial fibroblasts: maintained expression patterns of EGR-1, HLA-DR and some surface receptors. Rheumatol Int 1997; 16: 241–7
Rosier RN, Evans CH, editor. Molecular biology in orthopaedic. Rosemont (IL): American Academy of Orthopaedic Surgeons, 2002: 107–14
Marcelino J, Carpten JD, Suwairi WM, et al. CACP, encoding a secreted proteoglycan, is mutated in camptodactyly-arthropathy-coxa vara-pericarditis syndrome. Nat Genet 1999; 23: 319–22
Chan D, Weng YM, Graham HK, et al. A nonsense mutation in the carbosylterminal domain of type X collagen causes haploinsufficiency in schmid metaphyseal chondrodysplasia. J Clin Invest 1998; 101: 1490–9
Martignetti JA, Aqeel AA, Sewairi WA, et al. Mutation of the matrix metalloproteinase 2 gene (MMP2) causes a multicentric osteolysis and arthritis syndrome. Nat Genet 2001; 28: 261–5
Matsumoto I, Staub A, Benoist C, et al. Arthritis provoked by linked T and B cell recognition of a glycolytic enzyme. Science 1999; 286: 1732–5
Jorgensen C, Gay S. Gene therapy in osteoarticular diseases: where are we? Immunol Today 1998; 19: 387–91
Muller-Ladner U, Roberts CR, Franklin BN, et al. Human IL-1Ra gene transfer into human synovial fibroblasts is chondroprotective. J Immunol 1997; 158: 3492–8
Muller-Ladner U, Evans CH, Franklin BN, et al. Gene transfer of cytokine inhibitors into human synovial fibroblasts in the SCID mouse model. Arthritis Rheum 1999; 42: 490–7
Arita M, Li SW, Kopen G, et al. Skeletal abnormalities and ultrastructural changes of cartilage in transgenic mice expressing a collagen II gene (COL2A1) with a Cys for Arg-alpha1-519 substitution. Osteoarthritis Cartilage 2002; 10: 808–15
Gustafsson E, Aszodi A, Ortega N, et al. Role of collagen type II and perclean in skeletal development. Ann N Y Acad Sci 2003; 995: 140–50
Helminen HJ, Saamanen AM, Salminen H, et al. Transgenic mouse models for studying the role of cartilage macromolecules in osteoarthrists. Rheumatology (Oxf) 2002; 41: 848–56
Mier RJ, Holderbaum D, Ferguson R, et al. Osteoarthritis in children associated with a muation in the type II procollagen gene (COL2A1). Mol Genet Metab 2001; 74; 338–41
Baldwin CT, Cupples LA, Joost O, et al. Absence of linkage or association for osteoarthritis with the vitamin D receptor/type II collagen locus: the Framingham Osteoarthritis Study. J Rheumatol 2002; 29: 161–5
Petit B, Freyria AM, van der Rest M, et al. Cartilage collagens. In: Adolphe M, editor. Biological regulation of the chondrocytes. Boca Raton (FL): CRC Press, 1998: 33–72
Kuivaniemi H, Tromp G, Prockop DJ. Mutations in fibrillar collagens (Types I, II, III, and XI), fibril-associated collagen (Type IX), and network-forming collagen (type X) cause a spectrum of diseases of bone, cartilage, and blood vessels. Hum Mutat 1997; 9: 300–15
Fassler R, Schnegelsberg PN, Dausman J, et al. Mice lacking alpha 1 (IX) collagen develop noninflammatory degenerative joint disease. Proc Natl Acad Sci U S A 1994; 91: 5070–4
Nakata K, Ono K, Miyazaki J, et al. Osteoarthritis associated with mild chondrodysplasia in transgenic mice expressing alpha 1(IX) collagen chains with a central deletion. Proc Natl Acad Sci U S A 1993; 90: 2870–4
Aszodi A, Hunziker EB, Olsen BR, et al. The role of collagen II and cartilage fibril-associated molecules in skeletal development. Osteoarthritis Cartilage 2001; 9Suppl. A: S150–9
Childs S, Chen JN, Garrity DM, et al. Patterning of angiogenesis in the zebrafish embryo. Development 2002; 129: 973–82
Westermarck J, Kahari VM. Regulation of matrix metalloproteinase expression in tumor invasion. FASEB J 1999; 13: 781–92
Chandler S, Coates R, Gearing A, et al. Matrix metalloproteinases degrade myelin basic protein. Neurosci Lett 1995; 201: 223–6
Rosenberg GA. Matrix metalloproteinases in brain injury. J Neurotrauma 1995; 12: 833–42
George SJ. Therapeutic potential of matrix metalloproteinase inhibitors in atherosclerosis. Expert Opin Investig Drugs 2000; 9: 993–1007
Longo GM, Xiong W, Greiner TC, et al. Matrix metalloproteinases 2 and 9 work in concert to produce aortic aneurysms. J Clin Invest 2002; 110: 625–32
Shahin M, Konturek JW, Pohle T, et al. Remodeling of extracellular matrix in gastric ulceration. Microsc Res Tech 2001; 53: 396–408
Boeker KH, Haberkorn CI, Michels D, et al. Diagnostic potential of circulating TIMP-1 and MMP-2 as markers of liver fibrosis in patients with chronic hepatitis C. Clin Chim Acta 2002; 316: 71–81
Mengshol JA, Mix KS, Brinckerhoff CE. Matrix metalloproteinases as therapeutic targets in arthritic diseases: bull’s-eye or missing the mark? Arthritis Rheum 2002; 46: 13–20
Bayliss MT, Hutton S, Hayward J, et al. Distribution of aggrecanase (ADAMts 4/5) cleavage products in normal and osteoarthritic human articular cartilage: the influence of age, topography and zone of tissue. Osteoarthritis Cartilage 2001; 9: 553–60
Brinckerhoff CE, Matrisian LM. Matrix metalloproteinases: a tail of a frog that became a prince. Nat Rev Mol Cell Biol 2002; 3: 207–14
Martel-Pelletier J, Welsch DJ, Pelletier JP. Metalloproteases and inhibitors in arthritic diseases. Best Pract Res Clin Rheumatol 2001; 15: 805–29
Cawston T, Billington C, Cleaver C, et al. The regulation of MMPs and TIMPs in cartilage turnover. In: Greenwald RA, Zucker S, Golub LM, editors. Inhibition of matrix metalloproteinases therapeutic applications. New York: The New York Academy of Sciences, 1999: 120–9
Sternlicht MD, Werb Z. How matrix metalloproteinasesregulate cell behavior. Annu Rev Cell Dev Biol 2001; 17: 463–516
Lohi J, Wilson CL, Roby JD, et al. Epilysin, a novel human matrix metalloproteinase (MMP-28) expressed in testis and keratinocytes and in response to injury. J Biol Chem 2001; 276: 10134–44
Lander ES, Linton LM, Birren B, et al. Initial sequencing and analysis of the human genome. Nature 2001; 409: 860–921
Venter JC, Adams MD, Myers EW, et al. The sequence of the human genome. Science 2001; 291: 1304–51
Wolfsberg TG, Bazan JF, Blobel CP, et al. The precursor region of a protein active in sperm-egg fusion contains a metalloprotease and a disintegrin domain: structural, functional, and evolutionary implications. Proc Natl Acad Sci U S A 1993; 90: 10783–7
Abagyan RA, Totrov MM, Kuznetsov DA. ICM, a new method for structure modeling and design: applications to docking and structure prediction from the distorted native conformation. J Comp Chem 1994; 15: 488–506
Moss ML, White JM, Lambert MH, et al. TACE and other ADAM proteases as targets for drug discovery. Drug Discov Today 2001; 6: 417–26
Attur MG, Dave MN, Patel IR, et al. Regulation of inflammatory mediators by tetracyclines. In: Nelson M, Hillen W, Greenwald RA, editors. Tetracyclines in biology, chemistry and medicine. Boston (MA): Birkhauser Verlag, 2001: 295–310
Black RA, Rauch CT, Kozlosky CJ, et al. A metalloproteinase disintegrin that releases tumor-necrosis factor-α from cells. Nature 1997; 385: 729–33
Ganu V, Melton, R, Hu S, et al. Extracellular matrix proteins COMP and fibronectin are substrates for Htra, a novel serine protease upregulated in osteoarthritic cartilage. 47th Annual Meeting, Orthopaedic Research Society; 2001 Feb 25–28; San Francisco.
Holmbeck K, Bianco P, Caterina J, et al. MT1-MMP-deficient mice develop dwarfism, osteopenia, arthritis, and connective tissue disease due to inadequate collagen turnover. Cell 1999; 99: 81–92
Bakker AC. van de Loo, FA, van Beuningen HM, et al. Overexpression of active TGF-beta-1 in the murine knee joint: evidence for synovial-layer-dependent chondro-osteophyte formation. Osteoarthritis Cartilage 2001; 9: 128–36
Loeser RF. Chondrocyte integrin expression and function. Biorheology 2000; 37: 109–16
Attur MG, Dave MN, Clancy RM, et al. Functional genomic analysis in arthritis-affected cartilage: Yin-Yang regulation of inflammatory mediators by alpha5beta1 and alphaVbeta3 integrins. J Immunol 2000; 164: 2684–91
Heinegard D, Bayliss M, Lorenzo P. Pathogenesis of osteoarthritis. In: Brandt KD, Doherty M, Lohmander LS, editors. Osteoarthritis. Oxford: Oxford University Press, 1998: 74–84
Attur MG, Patel IR, Patel RN, et al. Autocrine production of IL-1β by human osteoarthritis-affected cartilage and differential regulation of endogenous nitric oxide, IL-6, prostaglandin E2 and IL-8. Proc Assoc Am Physicians 1998; 110: 1–8
Amin AR, Attur MG, Abramson SB. Regulation of nitric oxide and inflammatory mediators in human osteoarthritis-affected cartilage: implication for pharmacological intervention. In: Rubanyi GM, editor. The pathophysiology and clinical applications of nitric oxide. Richmond (CA): Harwood Academic Publishers, 1999: 397–413
Amin AR, Attur M, Abramson SB. Nitric oxide synthase and cyclooxygenases: distribution, regulation, and intervention in arthritis. Curr Opin Rheumatol 1999; 11: 202–9
Amin AR, Dave M, Attur M, et al. Cox-2, no, and cartilage damage and repair. Curr Rheumatol Rep 2000; 2: 447–53
Sodek J, Ganss B, McKee MD. Osteopontin. Crit Rev Oral Biol Med 2000; 11: 279–303
Sodek J, Zhu B, Huynh MH, et al. Novel functions of the matricellular proteins osteopontin and osteonectin/SPARC. Connect Tissue Res 2002; 43: 308–19
Pullig O, Weseloh G, Gauer S, et al. Osteopontin is expressed by adult human osteoarthritic chondrocytes: protein and MRNA analysis of normal and osteoarthritic cartilage. Matrix Biol 2000; 19: 245–55
Wright GD, Hughes AE, Regan M, et al. Association of two loci on chromosome 2q with nodal osteoarthritis. Ann Rheum Dis 1996; 55: 317–9
Saito S, Yamaji N, Yasunaga K, et al. The fibronectin extra domain a activates matrix metalloproteinase gene expression by an interleukin-1-dependent mechanism. J Biol Chem 1999; 274: 30756–63
Aplin AE, Howe A, Alahari SK, et al. Signal transduction and signal modulation by cell adhesion receptors: the role of integrins, cadherins, immunoglobulin-cell adhesion molecules, and selectins. Pharmacol Rev 1998; 50: 197–263
Longhurst CM, Jennings LK. Integrin-mediated signal transduction. Cell Mol Life Sci 1998; 54: 514–26
Denhardt DT, Noda M, O’Regan AW, et al. Osteopontin as a means to cope with environmental insults: regulation of inflammation, tissue remodeling, and cell survival. J Clin Invest 2001; 107: 1055–61
Attur MG, Dave M, Cipolletta C, et al. Reversal of autocrine and paracrine effects of interleukin 1 (IL-1) in human arthritis by type II IL-1 decoy receptor: potential for pharmacological intervention. J Biol Chem 2000; 275: 40307–15
Hurvitz JR, Suwairi WM, Van Hul W, et al. Mutations in the CCN gene family member WISP3 cause progressive pseudorheumatoid dysplasia. Nat Genet 1999; 23: 94–8
Feldmann M, Maini RN. Links discovery of TNF-alpha as a therapeutic target in rheumatoid arthritis: preclinical and clinical studies. Joint Bone Spine 2002 Jan; 69(1): 12–8
Cohen S, Hurd E, Cush J, et al. Treatment of rheumatoid arthritis with anakinra, a recombinant human interleukin-1 receptor antagonist, in combination with methotrexate: results of a twenty-four-week, multicenter, randomized, doubleblind, placebo-controlled trial. Arthritis Rheum 2002; 46: 614–24
Lin H, Ho AS, Haley-Vicente D, et al. Cloning and characterization of IL-1Hy2, a novel interleukin-1 family member. J Biol Chem 2001; 276: 20597–602
Kumar S, McDonnell PC, Lehr R, et al. Identification and initial characterization of four novel members of the interleukin-1 family. J Biol Chem 2000; 275: 10308–14
Mulero JJ, Pace AM, Nelken ST, et al. IL1HY1: a novel interleukin-1 receptor antagonist gene. Biochem Biophys Res Commun 1999; 263: 702–6
O’Neill LA, Greene C. Signal transduction pathways activated by the IL-1 receptor family: ancient signaling machinery in mammals, insects, and plants. J Leukoc Biol 1998; 63: 650–7
Geng Y, Valbracht J, Lotz M. Selective activation of the mitogen-activated protein kinase subgroups c-Jun NH2 terminal kinase and p38 by IL-1 and TNF in human articular chondrocytes. J Clin Invest 1996; 98: 2425–30
Loughlin J. Genetic epidemiology of primary osteoarthritis. Curr Opin Rheumatol 2001; 13: 111–6
Leppavuori J, Kujala U, Kinnunen J, et al. Genome scan for predisposing loci for distal interphalangeal joint osteoarthritis: evidence for a locus on 2q. Am J Hum Genet 1999; 65: 1060–7
Haukim N, Bidwell JL, Smith AJ, et al. Cytokine gene polymorphism in human disease: on-line databases, Supplement 2. Genes Immun 2002; 3: 313–30
El Omar EM, Carrington M, Chow WH, et al. Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature 2000; 404: 398–402
Loughlin J, Dowling B, Mustafa Z, et al. Association of the interleukin-1 gene cluster on chromosome 2q13 with knee osteoarthritis. Arthritis Rheum 2002; 46: 1519–27
Moos V, Rudwaleit M, Herzog V, et al. Association of genotypes affecting the expression of interleukin-1beta or interleukin-1 receptor antagonist with osteoarthritis [published erratum in Arthritis Rheum 2001; 44 (7): 1715]. Arthritis Rheum 2000; 43: 2417–22
Amin AR, Thakker G, Patel IR, et al. Regulation of IL-1 gene transcription in human chondrocytes and macrophages: preferential susceptibility of chondrocytes to IL-1 and diverse signaling pathways [abstract]. Arthritis Rheum 2000; 43Suppl. S: 599
Meulenbelt I, Bijkerk C, Miedema HS, et al. A genetic association study of the IGF-1 gene and radiological osteoarthritis in a population-based cohort study (the Rotterdam Study). Ann Rheum Dis 1998; 57: 371–4
Yamada Y, Okuizumi H, Miyauchi A, et al. Association of transforming growth factor beta1 genotype with spinal osteophytosis in japanese women. Arthritis Rheum 2000; 43: 452–60
Ghivizzani SC, Kang R, Georgescu HI, et al. Constitutive intra-articular expression of human il-1 beta following gene transfer to rabbit synovium produces all major pathologies of human rheumatoid arthritis. J Immunol 1997; 159: 3604–12
Van den Berg WB. Growth factors in experimental osteoarthritis: transforming growth factor beta pathogenic? J Rheumatol Suppl 1995; 43: 143–5
Smith P, Shuler FD, Georgescu HI, et al. Genetic enhancement of Matrix synthesis by articular chondrocytes: comparison of different growth factor genes in the presence and absence of interleukin-1. Arthritis Rheum 2000; 43: 1156–64
Mi Z, Ghivizzani SC, Lechman ER, et al. Adenovirus-mediated gene transfer of insulin-like growth factor 1 stimulates proteoglycan synthesis in rabbit joints. Arthritis Rheum 2000; 430: 2563–70
Woods JM, Katschke KJ, Volin MV, et al. IL-4 adenoviral gene therapy reduces inflammation, proinflammatory cytokines, vascularization, and bony destruction in rat adjuvant-induced arthritis. J Immunol 2001; 166: 1214–22
Bardos T, Kamath RV, Mikecz K, et al. Anti-inflammatory and chondroprotective effect of TSG-6 (tumor necrosis factor-alpha-stimulated gene-6) in murine models of experimental arthritis. Am J Pathol 2001; 159: 1711–21
Taniguchi K, Kohsaka H, Inoue N, et al. Induction of the P16INK4a Senescence gene as a new therapeutic strategy for the treatment of rheumatoid arthritis. Nat Med 1999; 5: 760–7
Iyama S, Okamoto T, Sato T, et al. Treatment of murine collagen-induced arthritis by ex vivo extracellular superoxide dismutase gene transfer. Arthritis Rheum 2001; 44: 2160–7
Yaspo ML. Taking a functional genomics approach in molecular medicine. Trends Mol Med 2001; 7: 494–501
Bumol TF, Watanabe AM. Genetic information, genomic technologies, and the future of drug discovery. JAMA 2001; 285: 551–5
Drews J. Drug discovery: a historical perspective. Science 2000; 287: 1960–4
Stuhlmuller B, Ungethum U, Scholze S, et al. Identification of known and novel genes in activated monocytes from patients with rheumatoid arthritis. Arthritis Rheum 2000; 43: 775–90
Gerber HP, Vu TH, Ryan AM, et al. VEGF couples hypertrophic cartilage remodeling, ossification and angiogenesis during endochondral bone formation. Nat Med 1999; 5: 623–8
Acknowledgements
We thank Ms Andrea L. Barrett and Ms Cari Reiner for the preparation of the manuscript and Ms Sonali Trivedi for preparation of the figures. We would like to thank Dr Smita Palejwala (University of Medicine and Dentistry of New Jersey) for critically reviewing the manuscript and for her constructive suggestions. We would like to thank NDRI (Philadelphia, PA) for some of the cartilage samples. ## We would like to thank the American Association of Orthopedic Surgeons and the Journal of Immunology for allowing us to republish Fig 1
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Attur, M.G., Dave, M.N. & Amin, A.R. Functional Genomics Approaches in Arthritis. Am J Pharmacogenomics 4, 29–43 (2004). https://doi.org/10.2165/00129785-200404010-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00129785-200404010-00004