Abstract
Background and Objective: In chronic disease, treatment effects and costs accumulate over time; hence, the choice of time horizon in cost-effectiveness analysis can be particularly important. In this article we analyse the dynamic changes in cumulative costs, effects and incremental cost effectiveness of two competing drug strategies in patients with early Parkinson’s disease (PD).
Methods: Three hundred and one subjects with PD were randomised to initial pramipexole or levodopa and followed every 3 months over a 4-year period. Healthcare resource use was recorded in patient diaries and valued using a variety of sources at year 2002 $US values. Health-related quality of life (HRQoL) was measured using the EuroQoL EQ-5D. The study was conducted from a US societal perspective. Missing data were imputed using a multivariate fixed-effects model. Additional quality adjusted life years (QALY) gained by using pramipexole compared with levodopa were estimated as the area between the normalised treatment HRQoL profiles. The QALYs and costs for each treatment arm were calculated for various study horizons.
The incremental cost-effectiveness ratio (ICER) and the net monetary benefit (NB) [using $US50 000, $US100 000 and $US150 000 as the value of a QALY] were estimated, and were bootstrapped to calculate the standard errors. Cost-effectiveness acceptability curves (CEAC) were built to estimate the probability that pramipexole was cost effective given different societal values of QALY, for various study horizons.
We conducted sensitivity analyses on the ICER and the NB to test their robustness to various assumptions about missing data, for various subpopulations and under changes in the drug prices.
Results: Under the base-case assumptions, the ICER for pramipexole was $US42 989 per QALY. Using the CEAC approach, the probability that pramipexole was cost effective relative to levodopa over the first 4 years was 0.57, 0.77 and 0.82 when a QALY was valued at $US50 000, $US100 000, and $US150 000, respectively. Over time, the ICER for pramipexole improved and uncertainty around the ICER decreased. If, after treatment withdrawal, HRQoL improved in pramipexole subjects and declined in levodopa subjects (best-case scenario for pramipexole), the probability of pramipexole being cost effective increased to 0.88, 0.96 and 0.98, respectively. Factors that improved the ICER of pramipexole were a decrease in the relative price of pramipexole and having low HRQoL or depression at baseline.
Conclusions: The cost effectiveness of pramipexole compared with levodopa in the treatment of early PD increased as the time horizon of the clinical trial extended from 2 to 4 years. Our results suggest that pramipexole is more cost effective for patients with depression and low baseline HRQoL than in other patient subgroups.







Similar content being viewed by others
Notes
The use of tradenames is for product identification purposes only and does not imply endorsement.
Using the multivariate imputation model described above, we estimated a one-time deterioration of 0.078 QALYs in the pramipexole arm and 0.065 QALYs in the levodopa arm at the last visit before the subject dropped out of the study.
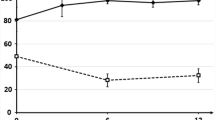
EQ-5D is expressed in units of utility, and is assessed using preference-based methods (time trade-off). It uses external weights (obtained on the sample of adult population in the UK) to provide a single score based on its five health domains that is bound by −0.594 and 1. The EQ-VAS assesses preference using internal (patient’s own) ‘health domains’ and weights and provides a HRQoL score on the scale 0 to 1. Changes in the EQ-5D and -VAS over time represent QALYs[52]
References
Marras C, Lang A, Krahn M, et al. Quality of life in early Parkinson’s disease: the impact of dyskinesias and motor fluctuations. Mov Disord 2004; 19 (1): 22–8
Hoerger TJ, Bala MV, Rowland C, et al. Cost effectiveness of pramipexole in Parkinson’s disease in the US. Pharmacoeconomics 1998; 15 (5): 541–67
LePen C, Wait S, Moutard-Martin F, et al. Cost of illness and disease severity in a cohort of French patients with Parkinson’s disease. Pharmacoeconomics 1999; 16 (1): 59–69
Rubenstein LM, DeLeo A, Chrischilles EA. Economic and health-related quality of life considerations of new therapies in Parkinson’s disease. Pharmacoeconomics 2001; 19 (7): 729–52
Dodel RC, Berger K, Oertel WH. Health-related quality of life and healthcare utilization in patients with Parkinson’s disease: impact of motor fluctuations and dyskinesias. Pharmacoeconomics 2001; 19 (10): 1013–38
US Food and Drug Administration, Department of Health and Human Services. Approvals of FDA-regulated products [online]. Available from URL: http://www.fda.gov/opacom/7ap provl.html [Accessed 2005 Sep 22]
Zielinski SL. FDA attempting to overcome major roadblocks in monitoring drug safety. J Nall Cancer Inst 2005 Jun 15; 97 (12): 872–3
Noyes K, Dick AW, Holloway RG, et al. Pramipexole vs levodopa as initial treatment for Parkinson’s disease: a randomized clinical-economic trial. The Parkinson Study Group. Med Decis Making 2004; 24: 472–85
Parkinson Study Group. Pramipexole vs levodopa as initial treatment for Parkinson disease: a randomized controlled trial. JAMA 2000; 284: 1931–8
Parkinson Study Group. Pramipexole versus levodopa as initial treatment for Parkinson disease: a 4-year randomized controlled trial. Arch Neurol 2004; 61: 1044–53
Noyes K, Dick AW, Holloway RG, et al. Quality of life assessment of early Parkinson. Med Decis Making 2003; 23 (6): 248
Parkinson Study Group. A randomized controlled trial comparing pramipexole with levodopa in early Parkinson’s disease: design and methods of the CALM-PD study. Clin Neuropharmacol 2000; 23 (1): 34–44
Ramsey SD, McIntosh M, Sullivan SD. Design issues for conducting cost-effectiveness analyses alongside clinical trials. Annu Rev Public Health 2001; 22: 129–41
Fahn S, Elton RL, Members of the UPDRS Development Committee. Unified Parkinson’s disease rating scale. In: Fahn S, Marsden CD, Calne DB, et al., editors. Recent developments in Parkinson’s disease. Vol 2. Macmillian Health Care Information; Florman Park (NJ), 1987: 153–64
Dolan P. Modelling valuations for EuroQol health states. Med Care 1997; 11: 1095–108
Shaw JW, Johnson JA, Coons SJ. US valuation of the EQ-5D health states: development and testing of the DI valuation model. Med Care 2005; 43: 203–20
Drummond MF, O’Brien BJ, Stoddart GL, et al. Methods for economic evaluation of health care programmes. 2nd ed. Oxford, UK: Oxford University Press, 1997
Gold MR, Siegel JE, Russell LB, et al. Cost-effectiveness in health and medicine. New York: Oxford University Press, 1996
Siderowf A, Ravina B, Glick HA. Preference-based quality-oflife in patients with Parkinson’s disease. Neurology 2002; 59 (1): 103–8
Brazier JE, Walters SJ, Nicholl JP, et al. Using the SF-36 and EuroQoL on an elderly population. Qual Life Res 1996; 5 (2): 195–204
Federal Register (1994) Part II: 42 CFR Parts 410 and $14 Medicare program. Physician Fee Schedule for Calendar Year 1995; Payment Policies and Relative Value Unit Adjustments. Vol. 59, No. 235. Washington, DC: US Government Printing Office, 1994
Center for Medicare and Medicaid Services. DRG definitions manual version 18, effective 1-1-2001. St. Paul (MN): 3M Health Information Systems, 2000
US Census Bureau. Statistical abstract of the United States: 2000. Table no. 750: money income of persons. Washington, DC: Government Printing Office, 2000
Gabrel CS. An overview of nursing home facilities: data from the 1997 National Nursing Home Survey. Hyattsville (MD): National Center for Health Statistics, 2000
The Red Book Drug Topics. Montvale (NJ): Medical Economics Company Inc, 2001
Williams RM. The costs of visits to emergency departments. N Engl J Med 1996; 334: 642–6
Torrance GW, Siegel J, Luce BR. Framing and designing the cost-effectiveness analysis. In: Gold MR, Russell LB, Siegel JE, et al., editors. Cost-effectiveness in health and medicine. Oxford: Oxford University Press, 1996: 54–81
Meltzer D, Johannesson M. Inconsistencies in the ’societal perspective’ on costs of the panel on cost-effectiveness in heath and medicine. Med Decis Making 1999; 19: 371–7
Engels JM, Diehr P. Imputation of missing longitudinal data: a comparison of methods. J Clin Epidemiol 2003; 56: 968–76
Duan N, Manning WG, Morris CN, et al. Choosing between the sample selection model and multi-part model. J Bus Econ Stat 1984; 2: 283–9
Torrance GW, Tugwell P, Amorosi S, et al. Improvement in health utility among patients with rheumatoid arthritis treated with adalimumab (a human anti-TNF monoclonal antibody) plus methotrexate. Rheumatology 2004; 43: 712–8
Consumer price index. Washington, DC: US Department of Labor, Bureau of Labor Statistics [online]. Available from URL: http://www.bl s.gov/cpi [Accessed 2005 Nov 4]
Hoch JS, Briggs AH, Willan AR. Something old, something new, something borrowed, something blue: a framework for the marriage of health econometrics and cost-effectiveness analysis. Health Econ 2002; 11 (5): 415–30
Stinnett AA, Mullahy J. Net health benefit: a new framework for the analysis of uncertainty in cost-effectiveness analysis. Med Decis Making 1998; 18 (2 Suppl.): S68–80
Fenwick E, O’Brien BJ, Briggs A. Cost-effectiveness acceptability curves: facts, fallacies and frequently asked questions. Health Econ 2004; 13 (5): 405–15
Van Hout BA, Alm J, Gordon GS, et al. Cost, effects and C/E ratios alongside a clinical trial. Health Econ 1994; 3: 309–19
Hariz GM, Lindberg M, Hariz MI, et al. Gender differences in disability and health-related quality of life in patients with Parkinson’s disease treated with stereotactic surgery. Acta Neurol Scand 2003; 108 (1): 28–37
Karlsen KH, Larsen JP, Tandberg E, et al. Influence of clinical and demographic variables on quality of life in patients with Parkinson’s disease. J Neurol Neurosurg Psychiatry 1999; 66 (4): 431–5
Karlsen KH, Tandberg E, Arsland D, et al. Health-related quality of life in Parkinson’s disease: a prospective longitudinal study. J Neurol Neurosurg Psychiatry 2000; 69 (5): 584–9
Stason WB, Weinstein MC. Allocation of resources to manage hypertension. N Engl J Med 1977; 296 (13): 732–9
Sanders GD, Hlatky MA, Every NR, et al. Potential costeffectiveness of prophylactic use of the implantable cardiac defibrillators and amiodarone after myocardial infarction. Ann Intern Med 2001; 135: 870–83
Weinstein MC, Stason WB. Cost-effectiveness of coronary artery bypass graft surgery. Circulation 1982; 66 (5 pt 2): III56–66
Williams A. Economics of coronary bypass grafting. BMJ 1985; 291 (6491): 326–9
Ubel PA, Hirth RA, Chernew ME, et al. What is the price of life and why doesn’t it increase at the rate of inflation? Arch Intern Med 2003; 163 (14): 1637–41
Rascol O, Brooks DJ, Korczyn AD, et al. A five-year study of the incidence of dyskinesia in patients with early Parkinson’s disease who were treated with ropinirole or levodopa. N Eng J Med 2000; 342: 1484–91
Baker CB, Johnsrud MT, Crismon ML, et al. Quantitative analysis of sponsorship bias in economic studies of antidepressants. Br J Psychiatry 2003 Dec; 183: 498–506
Briggs AH. A Bayesian approach to stochastic cost-effectiveness analysis: an illustration and application to blood pressure control in type 2 diabetes. Int J Technol Assess Health Care 2001; 17 (1): 69–82
Angell M. The truth about the drug companies: how they deceive us and what to do about it. New York: Random House, 2004
Greider K. The big fix: how the pharmaceutical industry rips off American consumers. Cambridge (MA): Public Affairs, 2003
Salomon JA, Weinstein MC, Goldie SJ, et al. Taking account of future technology in cost-effectiveness analysis. BMJ 2004; 329: 733–6
Coyle D, Barbeau M, Guttman M, et al. The economic evaluation of pharmacotherapies for Parkinson’s disease. Parkinsonism Relat Disord 2003; 9 (5): 301–7
Gold MR, Stevenson D, Fryback DG. HALYs and QALYs and DALYs, oh my: similarities and differences in summary measures of population health. Annu Rev Public Health 2002; 23: 115–34
Goldberg JF, Frye MA, Dunn RT. Pramipexole in refractory bipolar depression [letter]. Am J Psychiatry 1999; 156 (5): 798
Maggio R, Scarselli M, Novi F, et al. Potent activation of dopamine D3/D2 heterodimers by the antiparkinsonian agents, S32504, pramipexole and ropinirole. J Neurochem 2003; 87 (3): 631–41
Acknowledgements
The randomized clinical trial (but not the presented analyses) was supported by Pharmacia, Corp. (Peacock, NJ, USA) and Boehringer Ingelheim Pharma (Ingelheim, Germany). The additional analyses and research efforts for the cost-effectiveness study were supported by research grants K01 AG 20980 from the National Institute of Aging (Katia Noyes) and K24 NS4 2098 from the National Institute of Neurological Disorders and Stroke (Robert Holloway).
Author contribution:
KN: conception and design, obtaining funding, acquisition, analysis and interpretation of data, drafting and revisions of the manuscript.
AD: conception and design, analysis and interpretation of data, revisions of the manuscript.
RH: conception and design, obtaining funding, acquisition and interpretation of data, drafting and revisions of the manuscript.
The authors have no conflicts of interest that are directly relevant to the content of this study.
The earlier version of the manuscript was presented at the 9th Annual Meeting of the International Society for Pharmacoeconomics and Outcomes Research in Arlington, VA, May 2004, where it received the Best Podium Presentation Award. The authors would like to thank Drs. Andrew Briggs and Christopher McCabe for their comments on the earlier version of the manuscript, Carolynn O’Connell and Patricia Klein with assistance in preparing the manuscript, and all participating sites for collecting the data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noyes, K., Dick, A.W. & Holloway, R.G. Pramipexole and levodopa in early parkinson’s disease. Pharmacoeconomics 23, 1257–1270 (2005). https://doi.org/10.2165/00019053-200523120-00009
Published:
Issue Date:
DOI: https://doi.org/10.2165/00019053-200523120-00009