Summary
The usefulness of many anti-Gram-positive antibiotics is being compromised by the spread of antibiotic resistance in bacteria. The most reliable agents for serious infections are the glycopeptide agents vancomycin and teicoplanin.
The appropriate maintenance dosage for teicoplanin in serious infections is 6 mg/kg/day, i.e. usually 400 mg/day. There are 3 exceptions for which the daily maintenance dosage should be 12 mg/kg/day and these are intravenous drug abusers, septic arthritis (but not osteomyelitis), and Staphylococcus aureus endocarditis treated with teicoplanin monotherapy.
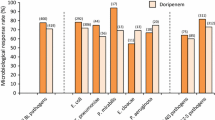
When teicoplanin is given at these doses, it achieves clinical and bacteriological results that are equivalent to those obtained with vancomycin, irrespective of pathogen or type of infection. The toxicity profile favours teicoplanin over vancomycin, especially when other, potentially toxic, drugs are coadministered. Teicoplanin also has an advantage in terms of ease and convenience of administration, which, together with its lack of need for routine blood level monitoring, facilitates its use outside hospital.
New agents hold some promise for the future; however, oral agents, if developed, could present the risk of being overused, which might compromise their long term utility.
Similar content being viewed by others
References
Stickler D. Prosthetic device-associated infections: what’s new? Curr Opin Infect Dis 1996; 9: 265–9
Eykyn SJ, Grandsen WR, Phillips I. The causative organisms of septicaemia and their epidemiology. J Antimicrob Chemother 1990; 25 Suppl. C: 41–58
Voss A, Milatovic D, Wallrauch-Schwarz C, et al. Methicillin-resistant Staphylococcus aureus in Europe. Eur J Clin Microbiol Infect Dis 1994; 13: 50–5
Spencer RC, Goering R. A critical review of the in vitro activity of teicoplanin. Int J Antimicrob Agents 1995; 5: 169–77
Goldstein F, Acar J. Antimicrobial resistance among lower respiratory tract isolates of Streptococcus pneumoniae — results of a 1992–93 Western European and United States collaborative surveillance study. J Antimicrob Chemother 1996; 38 Suppl. A: 71–84
Jones RN, Sader HS, Erwin ME, et al. Emerging multiply resistant enterococci among clinical isolates. I. Prevalence data from a 97 medical center surveillance study in the United States. Enterococcus Study Group. Diagn Microbiol Infect Dis 1995; 21: 85–93
Brown DFJ, Courvalin P, European Glycopeptide Resistance Group. European Glycopeptide Susceptibility Survey: susceptibility of Enterococcus spp to teicoplanin and vancomycin [abstract no. E026]. In: Program and Abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1996 Sep 15–18; New Orleans
Michel M, Gatmann L. Methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci: therapeutic realities and possibilities. Lancet 1997 Jun 28; 349: 1901–6
Lewis P, Garaud JJ, Parenti F. A multicentre open clinical trial of teicoplanin in infections caused by Gram-positive bacteria. J Antimicrob Chemother 1988; 21 Suppl. A: 61–7
Glupczynski Y, Lagast H, Van der Auwera P, et al. Clinical evaluation of teicoplanin for therapy of severe infections caused by Gram-positive bacteria. Antimicrob Agents Chemother 1986; 29: 52–7
Galanakis N, Giamarellou H, Vlachogiannis N, et al. Poor efficacy of teicoplanin in treatment of deep-seated staphylococcal infections. Eur J Clin Microbiol Infect Dis 1988; 7(2): 130–4
Calain P, Krause KH, Vaudaux P, et al. Early termination of a prospective randomised trial comparing teicoplanin and flucloxacillin for treating severe staphylococcal infections. J Infect Dis 1987; 155: 187–91
Gilbert DN, Wood CA, Kimbrough RC. The Infectious Disease Consortium of Oregon. Failure of treatment with teicoplanin at 6 milligrams/kilogram/day in patients with Staphylococcus aureus intravascular infection. Antimicrob Agents Chemother 1991; 35: 79–87
Martinetto P, Gariglio M, Malcangi A, et al. Studio microbiologico-clinico sull’efficacia di un nuovo antibiotico: la teicoplanina. G Batteriol Virol Immunol 1985; 78: 86–94
Wilson APR, Grüneberg RN, Neu H. A critical review of the dose of teicoplanin in Europe and the USA. Int J Antimicrob Agents 1994; 4 Suppl. 1: Sl–30
Wilson APR, Grüneberg RN. Teicoplanin, the first decade. Abingdon: The Medicine Group, 1997: 57–64
Gerard M, Van der Auwera P, Meunier F, et al. A controlled clinical trial on efficacy and safety of teicoplanin versus van-comycin in the treatment of staphylococcal infections [abstract no. 1039]. In: Program and Abstracts of the 27th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1987 Oct 4–7; New York: 278
Hoechst Marion Roussel. US Trial 102-009 (Data on file)
Wood MJ. The comparative efficacy and safety of teicoplanin and vancomycin. J Antimicrob Chemother 1996; 37: 209–22
Choi JY, Kim YR, Shin WS, et al. A randomised study comparing clinical efficacy of ceftazidime plus teicoplanin or vancomycin containing regimen in febrile granulocytopenia patients. J Korean Soc Chemother 1992; 10: 165–71
Liu CY, Lee WS, Fung CP, et al. Comparative study of teicoplanin vs vancomycin for the treatment of methicillin-resistant Staphylococcus aureus bacteraemia. Clin Drug Invest 1996; 12: 80–7
Studena M, Hlavacova E, Helpianska L, et al. Teicoplanin plus pefloxacin versus teicoplanin plus netilmicin in empiric therapy of febrile neutropenic patients with cancer and neutropenia: a randomised study of two once daily regimens in patients with previously inserted catheters. Chemotherapy 1994; 40: 431–4
Hernandez L, Figuera A, Jimenez ML, et al. Empiric antibiotic regimen for febrile neutropenia (FN): imipenem plus vancomycin vs imipenem plus teicoplanin as initial therapy [abstract no. 271]. Bone Marrow Transplant 1994; Suppl. 162
Lewis PJ, Martino P, Mosconi G, et al. Teicoplanin in endocarditis: a multicentre, open European study. Chemotherapy 1995; 41: 399–411
Leport C, Perronne C, Massip P, et al. Evaluation of teicoplanin for treatment of endocarditis caused by Gram-positive cocci in 20 patients. Antimicrob Agents Chemother 1989; 33: 871–6
Martino P, Venditti M, Micozzi A, et al. Teicoplanin in the treatment of Gram-positive bacterial endocarditis. Antimicrob Agents Chemother 1989; 33: 1329–34
Presterl B, Graninger W, Georgopoulos A. The efficacy of teicoplanin in the treatment of endocarditis caused by Gram-positive bacteria. J Antimicrob Chemother 1993; 31: 755–66
Mehtar S, Walsh B, Drabu YJ, et al. Teicoplanin v flucloxacillin + fusidic acid in Gram-positive infections — a multi-centre randomised study [abstract no. 932]. In: Program and Abstracts of the 28th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1988 Oct 23–26; Los Angeles
Le Frock JL. Teicoplanin Study Group of North America. Safety and efficacy of teicoplanin (Teico) vs cefazolin (Cef) for the treatment of skin/soft tissue infections due to Gram-positive pathogens [abstract no. 1335]. In: Programme and Abstracts of the 18th International Congress of Chemotherapy; 1993 Jun 27–Jul 2; Stockholm: 345
Jemsek JG, Teicoplanin Study Group of North America. Safety and efficacy of teicoplanin (Teico) vs cefazolin (Cef) for the treatment of skin/soft tissue infections due to Staphylococcus aureus [abstract no. 1336]. In: Programme and Abstracts, 18th International Congress of Chemotherapy; 1993 Jun 27–Jul 2; Stockholm: 345
Wilson APR, Grüneberg RN. Teicoplanin, the first decade. Abingdon: The Medicine Group, 1997: 83–4
Le Frock JL, Ristuccia AM, Ristuccia PA, et al. Teicoplanin in the treatment of bone and joint infection. Eur J Surg 1992; S567: 9–13
Davey PG, Williams AH. A review of the safety profile of teicoplanin. J Antimicrob Chemother 1991; 27 Suppl. B: 69–73
Anon. Targoeid NDA50-681. Clinical/statistical data section, volume 60, p. 335. Marion Merrell Dow Inc., Cincinnati, USA, 1991
Wilson APR, Grüneberg RN. Teicoplanin, the first decade. Abingdon: The Medicine Group, 1997: 143
Grüneberg RN. The true cost of monitoring antibiotic levels. Hosp Formul 1993; 28: 55–8
Wilson APR, Grüneberg RN. Use of teicoplanin in community medicine. Eur J Clin Microbiol Infect Dis 1994; 13: 701–10
Nathwani D, Parker S, Davey PG, et al. A feasibility study of non-inpatient IV (NIPIV) antibiotic therapy in the UK [abstract no. 1295]. In: Programme and Abstracts, 7th European Congress of Clinical Microbiology and Infectious Diseases; 1995 Mar 26–30; Vienna: 252
Graninger W, Wenisch C, Wieslinger E, et al. Experience with outpatient intravenous teicoplanin therapy for chronic osteomyelitis. Eur J Clin Microbiol Infect Dis 1995; 14: 643–7
Davey PG, Rowley DR, Phillips GA. Teicoplanin — home therapy for prosthetic joint infections. Eur J Surg Suppl 1992; 567: 23–5
Pye KG, Kelsey SM, Newland AC. Outpatient treatment of Hickman catheter infections. Lancet 1992; 339: 1237
Ball LM, Siddal S, Van Saenen H. Teicoplanin in home therapy of the terminally ill child. Eur J Haematol 1993; Suppl. 51/54: 14–7
Venditti M, Gelfusa V, Serra P, et al. 4-week treatment of streptococcal native valve endocarditis with high dose teicoplanin. Antimicrob Agents Chemother 1992; 36: 723–6
Presterl E, Graninger W, Georgopoulos A. The efficacy of teicoplanin in the treatment of endocarditis caused by Gram-positive bacteria. J Antimicrob Chemother 1993; 31: 755–66
Rubinstein E. Teicoplanin — domiciliary use in surgical infections. Eur J Surg Suppl. 1992; 567: 27–9
Dagan R, Einhorn M, Howard CB, et al. Outpatient and inpatient teicoplanin treatment for serious Gram-positive infections in children. Pediatr Infect Dis J 1993; 12: S17–20
Noble WC, Virani Z, Cree RGA. Co-transfer of vancomycin and other resistant genes from Enterrococcus faecalis NCTC 12201 to Staphylococcus aureus. FEMS Microbiol Lett 1992; 93: 195–8
Biavasco F, Lupidi R, Manso E, et al. In vitro activity of LY333328 against Gram-positive bacteria resistant or moderately susceptible to clinically available glycopeptides (GPs) [abstract no. F202]. In: Program and Abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1996 Sep 15–18; New Orleans
Blumberg EA, Mandler HD, Fuchs AE, et al. Efficacy and toxicity of Synercid (quinupristin/dalfopristin) [SYN (Q/D)] for vancomycin-resistant Enterococcus faecium (VREF) infections [abstract no. L032]. In: Program and Abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1996 Sep 15–18; New Orleans
Jernigan JA, Hadziyannis E, Schwarzmann SC, et al. Vancomycin-resistant Enterococcus faecium (VRE) bacteremia (B) in severely neutropenic patients [abstract no. J008]. In: Program and Abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1996 Sep 15–18; New Orleans
Nepomuceno J, Case M, Packuchi JR, et al. In vitro activity of oxazolidinones U-100592 and U-100766 against thirty-five isolates of vancomycin resistant enterococci [abstract no. F231]. In: Program and Abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1996 Sep 15–18; New Orleans
Willey BM, Sachse L, Mustachi B, et al. Comparative in vitro activities of a new oligosaccharide, everninomicin (SH27899), against clinical enterococci In: Program and Abstracts of the 36th Interscience Conference on Antimicrobial Agents and Chemotherapy; 1996 Sep 15–18; New Orleans
Goldstein FW, Acar JF. Epidemiology of quinolone resistance: Europe and North and South America. Drugs 1995; 49 Suppl. 2: 36–42
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grüneberg, R.N. Anti-Gram-Positive Agents. Drugs 54 (Suppl 6), 29–38 (1997). https://doi.org/10.2165/00003495-199700546-00007
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-199700546-00007