Summary
Synopsis: Ceftazidime 1 is a new ‘third generation’ cephalosporin administered intravenously or intramuscularly. Similarly to other third generation cephalosporins it has a broad spectrum of in vitro activity against Gram-positive and Gram-negative aerobic bacteria, is particularly active against Enterobacteriaceae (including β-lactamase-positive strains) and is resistant to hydrolysis by most β-lactamases. Importantly, in vitro ceftazidime is presently the most active cephalosporin available against Pseudomonas aeruginosa, but it is less active against Staphylococcus aureus than first and second generation cephalosporins. Only larger comparative trials are likely to discern any statistically significant differences in clinical efficacy which may exist between ceftazidime and other antibiotics, but ceftazidime appears to be similar in efficacy to ‘standard’ comparative drugs in lower respiratory tract infections and complicated and/or chronic urinary tract infections among debilitated or hospitalised patients. Thus, in patients having Gram-negative infections at these sites and in whom the potential toxicity of the aminoglycosides is a concern, ceftazidime may be a valuable alternative in that it apparently lacks serious side effects and does not require routine drug plasma concentration monitoring. In fibrocystic patients having acute respiratory tract infections, ceftazidime is highly effective at both reducing symptoms of infection and temporarily reducing the sputum counts of Pseudomonas species. However, in these patients resistance to ceftazidime may develop, as seen with other β-lactam antibiotics. In the treatment of fever of unknown origin or documented infections in immunocompromised adults and children, ceftazidime appears to be similar in efficacy to various 2-or 3-drug combinations. Nevertheless, the coadministration of an antibiotic having greater efficacy against Gram-positive bacteria should be considered in immunocompromised patients. Results from a small number of comparative trials suggest that ceftazidime may be as effective as the aminoglycosides in intra-abdominal, obstetric and gynaecological, and skin and soft tissue infections. However, further clinical experience, particularly a few well designed comparative studies, is needed to clarify the comparative efficacy in these conditions as well as in septicaemia/bacteraemia, meningitis, and bone and joint infections.
Antibacterial Activity: Ceftazidime is highly resistant to β-lactamases and has a broad spectrum of in vitro antibacterial activity, including Pseudomonas aeruginosa.
Like other third generation cephalosporins, ceftazidime is active in vitro at therapeutic concentrations against most Enterobacteriaceae, including β-lactamase-positive strains and those resistant to first generation cephalosporins or other antibiotics. 90% of Serratia marcescens, Providencia species and indole-positive Proteus species are usually inhibited by 8 mg/L or less, and 90% of most other Enterobacteriaceae are usually inhibited by 2 mg/L or less. However, the activity of ceftazidime against Enterobacter and Citrobacter species (except C. diversus) varies widely between studies [minimum concentration inhibitory of 90% of tested strains (MIC90) of <1 mg/L to >32 mg/L].
90% of tested strains of P. aeruginosa including β-lactamase-positive, as well as cefsulodin-, piperacillin-, carbenicillin-or aminoglycoside-resistant isolates, are sensitive or moderately sensitive to ceftazidime (MIC90 0.5 to 32 mg/L). Ceftazidime is also active in vitro against P. cepacia, P. alcali genes, P. putida and VA-1 (P. picketti), but some other Pseudomonas species are resistant.
In common with most other third generation cephalosporins, ceftazidime is highly active against Haemophilus influenzae, Neisseria gonorrhoeae and Neisseria meningitidis (including β-lactamase-positive strains) [MIC90 < 1 mg/L]. 90% of tested strains of Acinetobacter species are usually at least moderately sensitive to ceftazidime.
Ceftazidime is generally active in vitro against Staphylococcus aureus (range of reported MIC90 8 to 64 mg/L) except oxacillin/methicillin-resistant strains, and against streptococci except enterococci. Ceftazidime is less active than first and second generation cephalosporins against S. aureus.
Clostridium perfringens is generally sensitive or moderately sensitive to ceftazidime, but most other clinically important anaerobic bacteria tested (including Bacteroides fragilis and Clostridium difficile) are resistant to or at best only moderately sensitive to ceftazidime.
Against most susceptible bacteria the ceftazidime minimum bactericidal concentration (MBC) is equal to or twice the MIC. However, the MBC/MIC ratio is greater than 2 for some strains of P. aeruginosa, Enterobacteriaceae, and Acinetobacter species.
Generally, the MIC of ceftazidime for most strains is little affected by increases in inoculum size up to 106 colony-forming units/ml, by different media, or by the addition of up to 75% human serum.
Synergy between ceftazidime and various aminoglycosides or other antipseudomonal β-lactam antibiotics (cefsulodin or ureidopenicillins) has been demonstrated against P. aeruginosa and Enterobacteriaceae. However, the degree of synergistic activity varies widely with the drug and bacterial strain. As occurs with other cephalosporins, the combination of ceftazidime and cefoxitin is antagonistic in Pseudomonas aeruginosa, probably due to derepression (induction) of type I β-lactamases by cefoxitin.
Ceftazidime has marked stability in vitro against a variety of β-lactamases, including most R-piasmid-mediated enzymes. The β-lactamase stability pattern of ceftazidime is similar to those of cefoxitin and moxalactam (latamoxef) which are hydrolysed by fewer β-lactamases than most other cephalosporins. However, in some instances the stability of ceftazidime to hydrolysis by the β-lactamase of a species does not correlate with a low MIC for that species.
The in vitro activity of ceftazidime has been confirmed in vivo against experimental infections in rodents, guinea-pigs and rabbits.
Pharmacokinetics: Peak ceftazidime serum concentrations of 70 to 72 mg/L are attained immediately after a lg infusion administered over 20 to 60 minutes. Peak values immediately after administration of 1 g by intravenous bolus or approximately 1 hour after intramuscular injection are 120 to 146 and 37 to 43 mg/L, respectively. Ceftazidime mean peak plasma concentrations and the area under the concentration/time curve (AUC) appear to increase linearly with dose. Multiple-dose studies reveal no accumulation of drug.
The volume of distribution after intravenous or intramuscular administration is usually between 15 and 20L. Therapeutic concentrations of ceftazidime are achieved in a variety of tissues and body fluids —including female genital tissue, bone, pleural effusions, sputum, bile and cerebrospinal fluid (inflamed meninges), and peritoneal, extravascular, lymphatic and amniotic fluid. Mean ceftazidime concentrations of 5.2 mg/L are attained in human breast milk 1 hour after administration of 2g intravenously. Plasma protein binding of ceftazidime ranges from 5 to 22.8% and is independent of concentration.
In healthy subjects the elimination half-life of ceftazidime is 1.5 to 2.8 hours. The drug is not metabolised and is eliminated largely via the urine. As the pharmacokinetics are not significantly affected by probenecid, glomerular filtration and not tubular secretion appears to be the mechanism of renal elimination. Mean renal clearance of ceftazidime from healthy subjects ranges from 72 to 141 ml/min. There is a direct correlation between creatinine clearance and elimination rate. Thus, the half-life is prolonged in patients with moderate to severe renal impairment. In patients having creatinine clearance <12 ml/min the mean elimination half-life is between 14 and 30 hours. Ceftazidime is readily removed from the body by haemodialysis. The half-life of ceftazidime is prolonged slightly in neonates (mean half-life of 4 to 5 hours).
Therapeutic Trials: Ceftazidime (usual dosage 1 to 6g daily administered at 8-to 12-hourly intervals) has been shown to be effective in a wide range of infections caused by Gram-negative and Gram-positive bacteria. The infections, which were usually of moderate or greater severity, included lower respiratory tract infections (including acute infections in fibrocystic patients), urinary tract infections, septicaemia/bacteraemia, skin, soft tissue, bone and joint infections, intra-abdominal, obstetric and gynaecological infections, meningitis, and infections in febrile neutropenic patients.
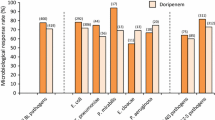
Frequently the infecting organism was a multidrug-resistant (but ceftazidime-sensitive) species of Enterobacteriaceae or a Pseudomonas species, or the infection had failed to resolve on alternative antibiotics). Bacteriological response rates varied. In complicated urinary tract infections in particular, from 90 to 100% of most Gram-negative and Gram-positive pathogens were eradicated, but percentages were lower for Enterobacter species (85%), Citrobacter species (79%), P. aeruginosa (76%), Staphylococcus epidermidis (73%) and Streptococcus faecalis (67%). Superinfection occurred in 2.5% of ceftazidime-treated patients and the development of resistance during therapy has been reported in a few Pseudomonas and Enterobacter species.
Most of the comparative trials (generally open and randomised) were small and hence unlikely to identify small differences between drugs that may have existed, but as indicated below, ceftazidime appeared to be similar in efficacy to the ‘standard’ antibiotics with which it was compared.
In patients with bacteriologically confirmed pneumonia or bronchitis, usually complicated by underlying diseases, mean clinical response rates (cured) of 51 to 88% have been reported in various comparative studies following treatment with ceftazidime (3 to 6 g/day as 2 to 4 divided doses). Mean clinical response rates of 53 to 88% were reported for patients in the comparative groups (cefamandole 1.5g 8-hourly or 1g 6-hourly, cefazolin 1.5g plus tobramycin 1.7 mg/kg 8-hourly, or ticarcillin 3g 4-hourly plus tobramycin 1.5 mg/kg 8-hourly). In patients having acute exacerbations of chronic bronchitis, clinical response rates were lower: 40% of patients receiving ceftazidime 2g 12-hourly and 31% of patients receiving cefazolin 2g plus tobramycin 40 to 80mg 12-hourly responded. Fibrocystic patients having acute pseudomonal lower respiratory tract infections were assessed in a randomised double-blind comparison of ceftazidime, cefoperazone, cefsulodin (200 mg/kg daily, each), azlocillin and piperacillin (400 mg/kg daily, each). The patients treated with ceftazidime or cefsulodin had the greatest improvement in their clinical condition and the highest temporary reduction of sputum counts of Pseudomonas species. In these patients the development of resistance was seen regularly with all of the drugs, and with ceftazidime and carbenicillin in a second comparative trial (a reversible resistance), but no significant resistance developed when tobramycin was used as mono-therapy. However, the therapeutic implications of the resistance which developed in these patients are unclear.
In complicated and/or chronic urinary tract infections (22 to 100% of which were due to P. aeruginosa) ceftazidime 0.5 to 1g 12-hourly effected a clinical and/or bacteriological response in 81 to 96% of patients (mean values reported in 4 different studies). Response rates for netilmicin lOOmg twice daily (60%), or tobramycin 1 mg/kg 8-hourly (79 to 90%) were not statistically different from the ceftazidime values.
Ceftazidime 2g twice daily cured all 27 patients with gallbladder empyema undergoing cholecystectomy whereas 12 of 23 patients receiving cefamandole 2g thrice daily plus tobramycin 80mg twice daily required withdrawal from the study because the pathogens (8 mixed infections of Escherichia coli plus enterococci, and 4 pseudomonal infections) were not sensitive to the drugs.
34 of 38 patients having various types of obstetric and gynaecological infections responded to therapy with ceftazidime 2g 8-hourly versus 34 of 39 patients treated with clindamycin 600mg plus tobramycin 1.5 mg/kg 8-hourly.
In paediatric patients, ceftazidime (usual dosage 90 to 150 mg/kg daily) has demonstrated clinical efficacy in chronic or recurrent otitis media complicated by P. aeruginosa (65% cured), complicated pseudomonal urinary tract infections (81% bacterial eradication) and serious infections in neonates less than or greater than 48 hours old (100% and 87.5% cured, respectively).
In immunocompromised patients (neutropenic adults and children, and both neutropenic and non-neutropenic adult haematological cancer patients) having fever of unknown origin or documented infection, ceftazidime 3 to 6g daily (as 3 to 6 divided doses) appeared to be similar in efficacy to the various 2-or 3-drug regimens with which it was compared (an aminoglycoside plus a ureidopenicillin and/or a different cephalosporin). However, inadequate efficacy against Gram-positive bacteria (2 studies) or a tendency to superinfection with Gram-positive bacteria (1 study) prompted the investigators to recommend the coadministration of an antibiotic having greater Gram-positive activity.
Side Effects: Ceftazidime is generally well tolerated. The most commonly reported side effects are similar to those of other 0-lactam antibiotics and include skin rashes, fever, diarrhoea, transient eosinophilia and reversible elevation of liver function tests. Altered renal function has occurred in 1.2% of patients.
Dosage and Administration: The recommended adult dosage is 1 to 6g daily in divided doses by intravenous or intramuscular injection. In urinary tract and less severe infections 500mg or 1g 12-hourly is usually adequate. In children 30 to 100 mg/kg daily and in neonates 25 to 60 mg/kg daily, as 2 to 3 divided doses, is recommended.
In patients with renal impairment the dosage must be decreased according to creatinine clearance.
Similar content being viewed by others
References
Abbas, A.M.A.; Taylor, M.C.; Newby, D.; Joncs, R.B.; Rigby, CC; Haste, A.R. and Cantor, A.M.: Ceftazidime: A new approach in the treatment of moderate and severe infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 147–152 (1983).
Acred, P.; Harding, S.M. and Ryan, D.M.: In vivo properties of GR 20263 (ceftazidime) a highly active broad spectrum cepha-losporin with antipseudomonal activity. Drugs Under Experimental and Clinical Research 7: 189–199 (1981a).
Acred, P.; Ryan, D.M. and Collard, A.E.: Comparative activity of ceftazidime (GR 20263) in experimental urinary tract infections; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July, 1981) pp. 491–493 (American Society for Microbiology, Washington D.C. 1982).
Acred, P.; Ryan, D.M.; Sowa, M.A. and Watts, CM.: In vivo antibacterial activity of ceftazidime (GR 20263) —a comparison with other new β-lactam antibiotics and gentamicin. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 247–255 (1981b).
Acuna, G.; Johnston, J.; Young, L.S. and Martin, W.J.: In vitro studies with ceftazidime against aerobic Gram-negative bacilli and Bacteroides fragilis group. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 83–89 (1981).
Adam, P.; Reichart, B. and Williams, K.J.: Penetration of ceftazidime into human tissue in patients undergoing cardiac surgery. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 269–273 (1983).
Adamsson, L.; Dornbusch, K. and Hallander, H.O.: Influence of bacterial β-lactamase production on susceptibility in agar or broth to new cephalosporins. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 153–162 (1981).
Alestig, K.; Andersson, R.; Olaisson, L. and Trollfors, B.: Dosage of ceftazidime in patients with renal impairment. (Abstract no. 802) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Alestig, K.; Trollfors, B.; Andersson, R.; Olaison, L.; Suurküla, M. and Norrby, S.R.: Ceftazidime and renal function. Journal of Antimicrobial Chemotherapy 13: 177–181 (1984).
Al-Khouri, S.; Bakhtiar, M. and Selwyn, S.: Comparative in vitro studies of 10 antipseudomonal β-lactam antibiotics; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July, 1981) pp. 78–80 (American Society for Microbiology, Washington D.C. 1982).
Aoyama, R.; Waga, S.; Ohnishi, A.; Izumi, Y.; Kuronuma, T.; Kakizaki, Y.; Nagata, K.; Kudo, M.; Okaniwa, S.; Sawaguchi, H. and Okamoto, T.: Clinical studies of ceftazidime in pediatric field. Japanese Journal of Antibiotics 37: 349–353 (1984).
Assael, B.M.; Boccazzi, A.; Caccamo, MX.; Giunta, A.; Marini, A.; Padoan, R.; Rusconi, F. and Sereni, F.: Clinical pharmacology of ceftazidime in paediatrics. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 341–346 (1983).
Axelrod, J.L.; Kochman, R.S.; Horowitz, M.A. and Youngworth, L.: Ceftazidime concentrations in human aqueous human. Archives of Ophthalmology 102: 923–925 (1984).
Ayrton, J.: Assay of ceftazidime in biological fluids using high-pressure liquid chromatography. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 227–231 (1981).
Baker, R.L. and Fass, R.J.: Correlation of in vitro activities of cephalothin and ceftazidime with their efficacies in the treatment of Staphylococcus aureus endocarditis in rabbits. Antimicrobial Agents and Chemotherapy 26: 231–234 (1984).
Bakker-Woudenberg, I.A.J.M.; van den Berg, J.C. and Michel, M.F.: Therapeutic activities of cefazolin, cefotaxime and ceftazidime against experimentally induced Klebsiella pneumoniae pneumonia in rats. Antimicrobial Agents and Chemotherapy 22: 1042–1050 (1982).
Bauemfeind, A.: An evaluation of the activity of cephalosporins against Pseudomonas aeruginosa. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 111–117 (1981).
Baumgärtner, M.: Cefoperazon, cefsulodin und ceftazidim —drei Pseudomonas-mrkssLme cephalosporinc in vergleich mit ihren monobaktam-analoga. Zentralblatt für Bakteriologie Mikrobiologie und Hygiene 254: 253–260 (1983).
Baumgärtner, M.; Müller, H.-P. and Wundt, W.: Antibiotische aktivität und Synergismus von ceftazidim und netilmicin bei gramnegativen problemkeimen. Arzneimittel-Forschung 33: 1615–1619 (1983).
Benoni, G.; Arosio, E.; Raimondi, M.G.; Apolloni, E.; Passarella, E; Lechi, A. and Velo, G.P.: Distribution of ceftazidime in ascitic fluid. Antimicrobial Agents and Chemotherapy 25: 760–763 (1984a).
Benoni, G.; Cuzzolin, L.; Puchetti, V. and Velo, G.P.: Penetration of ceftazidime in human pericardial fluid and lung tissue. Pharmacological Research Communications 16: 295–301 (1984b).
Berkowitz, F.E.; Colsen, P. and Raw, K.: Serratia marcescens endocarditis treated with ceftazidime. South African Medical Journal 64: 105–106 (1983).
Bianchi, A. and de Carneri, I.: In vitro susceptibility of Neisseria gonorrhoeae to ceftizoxime and other new cephalosporins. Proceedings of the 13th International Congress of Chemotherapy. Vienna (Aug 1983).
Bint, A.J.; Constable, J.; Patton, K.R. and Constable, F.L.: Ceftazidime therapy for serious infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 115–121 (1983).
Bint, A.J.; Yeoman, P.; Anderson, R. and Stansfield E.: In vitro activity of ceftazidime compared with that of other cephalosporins. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 47–51 (1981).
Blanco, J.D.; Gibbs, R.S.; Castaneda, Y.S. and St Clair, PJ.: Randomized comparison of ceftazidime versus clindamycin-tobramycin in the treatment of obstetric and gynecological infections. Antimicrobial Agents and Chemotherapy 24: 500–504 (1983a).
Blanco, J.D.; Jorgensen, J.H.; Castaneda, Y.S. and Crawford, S.A.: Ceftazidime levels in human breast milk. Antimicrobial Agents and Chemotherapy 23: 479–480 (1983b).
Blanco, J.D. and Lipscomb, K.A.: Single-dose prophylaxis in vaginal hysterectomy: a double-blind, randomised comparison of ceftazidime versus cefotaxime. Current Therapueitc Research 36: 389–393 (1984).
Blaser, J.; Bauernfeind, A.; Vogt, M. and Lüthy, R.: Monotherapie von systemischen Pseudomonas aeruginosa infektionen mit ceftazidim. Deutsche Medizinische Wochenschrift 108: 1312–1317 (1983).
Bodey, G.P.; Fainstein, V.; Garcia, I.; Rosenbaum, B. and Wong, Y.: Effect of broad-spectrum cephalosporins on the microbial flora of recipients. Journal of Infectious Diseases 148: 892–897 (1983).
Bodey, G.P.; Fainstein, V. and Hinkle, A.M.: Comparative in vitro study of new cephalosporins. Antimicrobial Agents and Chemotherapy 20: 226–230 (1981).
Bolivar, R.; Weaver, S.S. and Bodey, G.P.: Activity of β-lactamase inhibitors in combination with new β-lactam antibiotics against resistant gram-negative organisms. Diagnostic Microbiology and Infectious Disease 2: 255–260 (1984).
Bonner, D.P.; Whitney, R.R.; Baughn, C.O.; Miller, B.H.; Olsen, S.J. and Sykes, R.B.: In vivo properties of SQ 26,776. Journal of Antimicrobial Chemotherapy 8 (Suppl. E): 123–130 (1981).
Bouza, E.; Hellin, T.; Rodriguez-Creixems, M.; Martinez-Beltran, J.; Loza, E. and Baquero, F.: Comparison of ceftazidime concentrations in bile and serum. Antimicrobial Agents and Chemotherapy 24: 104–106 (1983b).
Bouza, E.; Hellin, T.; Sanz-Hospital, J.; Rodriguez-Creixems, M.; Loza, E. and Martinez-Beltran, J.: Evaluation of ceftazidime in the treatment of severe bacterial infection. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 153–159 (1983a).
Breyer, S.; Graninger, W.; Pauser, G.; Pirich, K. and Pichler, H.: Bacteriological and clinical comparison of moxalactam with newer cephalosporins. Drugs Under Experimental and Clinical Research 9: 287–290 (1983).
Brooks, G.F. and Barriere, S.L.: Clinical use of the new beta-lactam antimicrobial drugs. Annals of Internal Medicine 98: 530–535 (1983).
Brumfltt, W. and Hamilton-Miller, J.M.T.: Susceptibility of nosocomial pathogens to ceftazidime. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 15–21 (1981).
Burghele, A. and Ritzerfeld, W.: Pseudomonas-aclive cephalosporins in animal experiments; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July, 1981) pp. 519–521 (American Society for Microbiology, Washington D.C. 1982).
Bush, K. and Sykes, R.B.: Interaction of new β-lactams with β-lactamases and β-lactamase-producing Gram-negative rods; in Neu, H.C. (Ed) New Beta-Lactam Antibiotics: A Review from Chemistry to Clinical Efficacy of the New Cephalosporins, pp. 47–60 (Francis Clark Wood Institute for the History of Medicine, College of Physicians of Philadelphia, Philadelphia 1982).
Butler, J.K.: Microbiology, pharmacology and clinical activity of ceftazidime. Drugs Under Experimental and Clinical Research 9: 9–17 (1983).
Carbon, C; Dromer, F.; Brion, N.; Crcmieux, A.C. and Contrepois, A.: Renal disposition of ceftazidime illustrated by interferences by probenecid, furosemide, and indomethacin in rabbits. Antimicrobial Agents and Chemotherapy 26: 373–377 (1984).
Carmine, A.A.; Brogden, R.N.; Heel, R.C.; Romankiewicz, J.A.; Speight, T.M. and Avery, G.S.: Moxalactam (latamoxef): A review of its antibacterial activity, pharmacokinetic properties and therapeutic use. Drugs 26: 279–333 (1983).
Chattopadhyay, B.; Hall, I. and Cumow, S.R.: Ceftazidime (GR 20263), a new cephalosporin derivative with excellent activity against Pseudomonas and Enterobacteriaceae. Journal of Antimicrobial Chemotherapy 8: 491–493 (1981).
Chattopadhyay, B.; Hall, I. and Cumow, S.R.: Comparison of in vitro activity of lactamoxef (moxalactam) and ceftazidime against Pseudomonas and Enterobacteriaceae resistant to first generation cephalosporins. (Correspondence.) Journal of Antimicrobial Chemotherapy 11: 200–201 (1982).
Childs, S.J.; Mirelman, S. and Wells, W.G.: Perioperative use of ceftazidime as a prophylactic agent in transurethral surgery. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 71–76 (1983). aiuChilds, S.J.; Wells, W.G. and Mirelman, S.: Comparative clinical evaluation of ceftazidime and tobramycin in the treatment of Pseudomonas urinary tract infections. (Abstract no. 18.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Chow, A.W. and Bartlett, K.H.: Comparative in vitro activity of ceftazidime (GR 20263) and other β-lactamase stable cephalosporins against Pseudomonas. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 345–348 (1981a).
Chow, A.W. and Bartlett, K.H.: Comparative in vitro activity of ceftazidime (GR 20263) and other β-lactamase stable cephalosporins against anaerobic bacteria. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 91–95 (1981b).
Chow, A.W. and Finegold, S.M.: In vitro activity of ceftizoxime against anaerobic bacteria and comparison with other cephalosporins. Journal of Antimicrobial Chemotherapy 10 (Suppl. C): 45–50 (1982).
Clumeck, N.; Gordts, B.; Dab, I.; Jaspar, N.; Van Laethem, Y. and Butzler, J.-P.: Ceftazidime as a single agent in the treatment of severe Pseudomonas aeruginosa infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 207–211 (1983a).
Clumeck, N.; Van Laethem, Y.; Gordts, B.; Jaspar, N. and Butzler, J.-P.: Use of ceftazidime in the therapy of serious infections, incuding those due to multircsistant organisms. Antimicrobial Agents and Chemotherapy 24: 176–180 (1983b).
Cluzel, M.; Chanal, M.; Sirot, J. and Cluzel, R.: Activité bactericide comparée des associations beta-lactamines-aminosides sur Pseudomonas aeruginosa. Nouvelle Presse Medicale II: 3400–3404 (1982).
Commers, J.R.; Gress, J.G.; Robichaud, K.J.; Longo, D.L. and Pizzo, P.A.: Keflin gentamicin, carbenicillin vs ceftazidime as empiric therapy for febrile granulocytopenic cancer patients. (Abstract no.2.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Fiona (Oct 1982).
Cone, L.A.; Woodward, D.R.; Stolzman, D.H. and Byrd, R.G.: A prospective randomised study comparing ceftazidime or ticarcillin/tobramycin in the treatment of pneumonia and sepsis. (Abstract no. 836.) 23rd Interscience Conference on Antimicrobial Agents and Chemotherapy, Las Vegas (Oct 1983).
Coombes, J.D.; Metabolism and pharmacokinetics of the newer cephalosporins: cefotaxime, ceftizoxime, ceftriaxone, ceftazidime, cefoperazone, moxalactam and cefsulodin; in Neu (Ed.) New Beta-Lactam Antibiotics, Proceedings of the Cephalosporins Symposium of the American College of Clinical Pharmacology, pp. 177–214 (1982).
Corbett, C.R.R.; McFarland, R.J.; Spencer, G.R. and Ryan, D.M.: Peritoneal penetration of ceftazidime in elective surgery. (Abstract no. 809.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Corser, C.A.; Day, C.J. and Humble, M.W.: Third-generation cephalosporins: Comparative antibacterial activity against routine clinical isolates. New Zealand Medical Journal 95: 414–416 (1982).
Cox, C.E.: Comparison of ceflazidime and tobramycin in the treatment of complicated urinary tract infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 47–52 (1983).
Crokaert, F.; Potvliege, C; Vanderlinden, M.P.; Glupczynski, Y. and Yourassowsky, E.: Clinical and bacteriological evaluation of ceftazidime (GR 20263) as an antipseudomonal drug. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 325–329 (1981).
Cullen, R.T.; McCrae, W.M.; Govar, J.; Raeburn, J.A. and Ingram, T.M.: Ceftazidime in cystic fibrosis: Clinical, microbiological and immunological studies. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 369–375 (1983).
Daikos, G.K.; Kosmidis, J.; Stathakis, C; Giamarellou, H.; Douzinas, E. and Kastanakis, S.; Ceftazidime, a new broad-spectrum antipseudomonal cephalosporin: In vitro activity, human pharmacokinetics, and therapeutic efficacy; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July, 1981) pp. 499–501 (American Society for Microbiology, Washington D.C. 1982).
Daikos, G.K.; Kosmidis, J.; Stathakis, G; Giamarellou, H.; Douzinas, E.; Kastanakis, S. and Papathanassiou, B.: Ceftazidime: Therapeutic results in various infections and kinetic studies. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 331–337 (1981).
Darbyshire, P.J.; Williamson, P.J.; Pedler, S.J.; Speller, D.C.E.; Mott, M.G. and Oakhill, A.: Ceftazidime in the treatment of febrile immunosuppressed children. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 357–360 (1983).
Daschner, F.D.; Goerke, G; Bassler, M. and Just, M.: In vitro comparison of cefotcian with five other cephalosporins against nosocomial pathogens. Chemotherapy 28: 351–354 (1982).
Daschner, F.D.; Petersen, E.E.; Just, M. and Hillemanns, H.G.: Penetration of ceftazidime into serum, myometrium, endometrium, salpinges and subcutaneous tissue. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 247–249 (1983).
David, T.J.; Phillips, B.M.; Connor, P.J.: Ceftazidime —A significant advance in the treatment of cystic fibrosis. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 337–340 (1983).
Davies, B.I.; Maesen, F.P.V. and van Noord, J.A.: Treatment of chronic and recurrent respiratory infections with intramuscular ceftazidime. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 1–8 (1983).
Denys, G.A.; Jerris, R.C.; Swenson, J.M. and Thornsberry, C: Susceptibility of Propionibacterium acnes clinical isolates to 22 antimicrobial agents. Antimicrobial Agents and Chemotherapy 23: 335–337 (1983)
de Pauw, B.E.; Kauw, F.; Muytjens, H.; Williams, KJ. and Bothof, Th.: Randomized study of ceftazidime versus gentamicin plus cefotaxime for infections in severe granulocytopenic patients. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 93–99 (1983).
Digranes, A.; Dibb, W.L. and Benonisen, E.: Ceftazidime: In vitro comparison with cephalothin, cefuroxime, and netilmicin. Chemotherapy (Basel) 30: 113–118 (1984).
Dodge, J.; Zamiri, I.; Goodchild, M. and Ingram, P.: Experience with ceftazidime in cystic fibrosis. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 325–329 (1983).
Doko, F.: Basic and clinical studies on ceftazidime in the field of obstetrics and gynecology. Japanese Journal of Antibiotics 37: 57–62 (1984).
Dornbusch, K; Adamsson, L; Gezelius, L. and Hallander, H.: In vitro activity and β-lactamase stability of Sch 29482, a penem antibiotic. Comparisons with ceftazidime and moxalactam. Journal of Antimicrobial Chemotherapy 9 (Supp!. C): 113–121 (1982).
Dunn, G.L.: Ceftizoxime and other third-generation cephalosporins: Structure-activity relationships. Journal of Antimicrobial Chemotherapy 10 (Suppl. C): 1–10 (1982).
Dürckheimer, W.; Blumbach, J.; Heymes, R.; Schrinner, E. and Seeger, K.: Chemistry and structure-activity relationships in the aminothiazole cephalosporin series; in Neu, H.C. (Ed) New Beta-Lactam Antibiotics: A Review from Chemistry to Clinical Efficacy of the New Cephalosporins. Proceedings of the Cephalosporin Symposium of the American College of Clinical Pharmacology, 1981 (College of Physicians of Philadelphia, Philadelphia 1982).
Dutoy, J.P. and Wauters, G.: Treatment of bone infections with ceflazidime. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 229–233 (1983).
Elliott, T.S.J. and Ispahani, P.: Successful treatment of Pseudomonas aeruginosa meningitis with ceftazidime (correspondence). Journal of Infection 7: 275–283 (1983).
Eng, R.H.K.; Smith, S.M. and Cherubin, G: Inoculum effect of new β-lactam antibiotics on Pseudomonas aeruginosa. Antimicrobial Agents and Chemotherapy 26: 42–47 (1984).
Eron, L.J.; Goldenberg, R.I.; Park, C.H. and Poretz, D.M.: Ceftazidime therapy of serious bacterial infections. Antimicrobial Agents and Chemotherapy 23: 236–241 (1983b).
Eron, L.J.; Park, C.H.; Hixon, D.L.; Goldenberg, R.I. and Poretz, D.M.: Ceftazidime in patients with Pseudomonas infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 161–169 (1983a).
Fainstein, V.; Bodey, G.P.; Elting, L.; Bolivar, R.; Keating, M.J.; McCredie, K.B. and Valdivieso, M.: Randomized study of ceftazidime compared to ceftazidime and tobramycin for the treatment of infections in cancer patients. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 101–110 (1983a).
Fainstein, V.; Le Blanc, B. and Bodey, G.P.: In vitro activity of new cephalosporins against multi-resistant Gram-negative bacteria. Infection 11 (Suppl. 1): 20–22 (1983b).
Fainstein, V.; Weaver, S. and Bodey, G.P.: Comparative in vitro study of SQ 26,776. Antimicrobial Agents and Chemotherapy 21: 294–298 (1982).
Fass, R.J.: Comparative in vitro activities of third-generation cephalosporins. Archives of Internal Medicine 143: 1743–1745 (1983).
Fillastre, J.P.; Humbert, G.; Olier, B.; Le Guy, F.; Borsa, F. and Spencer, G.R.: Pharmacokinetics of ceftazidime in renal failure. (Correspondence.) Journal of Antimicrobial Chemotherapy 5: 487–488 (1983).
Fleming, P.C. and Knie, B.: Activity of ceftazidime against Pseudomonas aeruginosa from bacteraemic and fibrocystic patients. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 169–173 (1981).
Foord, R.D.: Ceftazidime: A summary of the early clinical results; in Neu (Ed.) New Beta-Laclam Antibiotics: A Review from Chemistry to Clinical Efficacy of the New Cephalosporins. Proceedings of the Cephalosporins Symposium of the American College of Clinical Pharmacology (1981) pp. 276–284 (College of Physicians of Philadelphia, Philadelphia 1982).
Foord, R.D.: Ceftazidime: Aspects of efficacy and tolerance. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 339–403 (1983).
Forsgren, A.: Comparative in vitro activity of first, second and third generation cephalosporins. Acta Pathologica Microbiologica et Immunologica Scandinavica Section B 89: 221–22S (1981).
Fourtillan, J.B.; Brisson, A.M.; Lefebvre, M.A. and Becq-Giraudon, B.: Pharmacokinetics of ceftazidime (GR 20263) in healthy subjects after single 2g dose IV bolus. (Abstract no. 174.) Paper presented at the 21st Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago (Nov 1981).
Francioli, P.; Clément, M.; Geroulanos, S.; von Graevenitz, A.; Lüthy, R.; Regamey, C; Stalder, H.; Vogt, M. and Waldvogel, F.A.: Ceftazidime in serious infections: A Swiss multicentre study. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 139–146 (1983).
Frimodt-Møller, P.C. and Madsen, P.O.: Ceftazidime, a new cephalosporin in the treatment of complicated urinary tract infections: A comparative study with tobramycin. Journal of Urology 130: 796–797 (1983).
Fujita, K.; Sakata, H.; Murono, K.; Yoshioka, H.; Maruyama, S.; Sanae, N. and Takimoto, M.: Qinical and pharmacokinetic evaluation of ceftazidime in children. Japanese Journal of Antibiotics 37: 329–337 (1984).
Garcia, I.; Fainstein, V.; Le Blanc, B. and Bodey, G.P.: In vitro activities of new β-lactam antibiotics against Acinetobactet spp. Antimicrobial Agents and Chemotherapy 24: 297–299 (1983).
Garzone, P.; Lyon, J. and Yu, V.L.: Third generation and investigational cephalosporins: II. Microbiological review and clinical summaries. Drug Intelligence and Clinical Pharmacy 17: 615–622 (1983).
Gasparetto, A. and Delogu, G.: Ceftazidime, as a single antibiotic in the treatment of multi-resistant Gram-negative infections in intensive care. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 171–175 (1983).
Gentry, L.O.; Douthit, M.B.; Childs, SJ. and Madsen, P.O.: Random comparative trial of 0.25, 0.5 and 1.0g ceftazidime twice daily in urinary tract infection. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 53–57 (1983).
George, W.L.: In vitro antibacterial activity of the combination of clindamycin and ceftazidime. Antimicrobial Agents and Chemotherapy 25: 657–658 (1984).
Giamarellou, H.; Avlami, A.; Matsakas, V.; Kosmidis, J. and Daikos, G.K.: In vitro studies with ceftazidime. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 73–77 (1981a).
Giamarellou, H.; Gazis, J.; Petrikkos, G.; Antsaklis, A.; Aravantinos, D. and Daikos, G.K.: Study of cefoxitin, moxalactam and ceftazidime kinetics in pregnancy. American Journal of Obstetrics and Gynecology 147: 914–919 (1983).
Giamarellou, H.; Koumaditis, A. and Daikos, G.K..: Cefsulodin kinetics in renal failure including the effects of dialysis. Drugs Under Experimental and Clinical Research 7: 425–429 (1981b).
Giamarellou, H.; Zissis, N.P.; Tagari, G. and Bouzos, J.: In vitro synergistic activities of aminoglycosides and new β-lactams against m ultiresistant Pseudomonas aeruginosa. Antimicrobial Agents and Chemotherapy 25: 534–536 (1984).
Gold, R.; Jin, E.; Levison, H.; Isles, A. and Fleming, P.C.: Ceftazidime alone and in combination in patients with cystic fibrosis: Lack of effect in treatment of severe respiratory infections caused by Pseudomonas cepacia. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 331–336 (1983).
Gonzenbach, H.R. and Sonnabend, W.: Ceftazidime versus the combination of tobramycin and clindamycin in the treatment of intra-abdominal infections. (Abstract no. 305.) 3rd Mediterranean Congress of Chemotherapy, Dubrovnik (Sept 1982).
Gooch, Wm. HI; Bosso, J.A.; Smith, L.H. and Rakhsha, G.: Accurate, simple microbiological assay for ceftazidime in body fluids. (Abstract no. 113.) 23rd Interscience Conference on Antimicrobial Agents and Chemotherapy, Las Vegas (Oct 1983).
Gordts, B.; Dab, I. and Butzler, J.P.: Ceftazidime in cystic fibrosis. (Correspondence.) Lancet 1: 1355 (1982).
Gower, P.E.; Hobbs, P.M. and Harding, S.M.: Kinetics of ceftazidime in renal impairment; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981) pp. 498–499 (American Society for Microbiology, Washington D.C. 1982).
Gozzard, D.I.; Geddes, A.M.; Eykyn, S.; Leyland, M.J.; Phillips, I. and Wise, R.: Clinical experience with ceftazidime in immunologically compromised patients. (Abstract no. 26) 2nd International Symposium on Infections in the lmmunocom-promised Host, Stirling, Scotland (June 1982).
Graninger, W.; Schmidbauer, C.P.; Ganzinger, U.; Lenzhofer, R.; Ho, I. and Spitzy, K.H.: Treatment of urinary tract infections due to Pseudomonas aeruginosa: Ceftazidime versus netilmicin; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981) pp. 493–495 (American Society for Microbiology, Washington D.C. 1982).
Grasso, S. and Licci, S.: In vitro activity of ceftizoxime and other beta-lactam antibiotics against Haemophilus influenzae. Proceedings of the 13th International Congress of Chemotherapy, Vienna (August 1983).
Gray, H.H. and Eykyn, S.J.: CAPD peritonitis. (Correspondence.) Lancet 1: 349 (1983).
Gwynn, M.N. and Rolinson, G.N.: Selection of variants of Gram-negative bacteria with elevated production of type 1-β-lactamase. Journal of Antimicrobial Chemotherapy 11: 577–581 (1983).
Hallander, H.O.; Dornbusch, K.; Gezelius, L.; Jacobson, K. and Karlsson, I.: Synergism between aminoglycosides and cephalosporins with antipseudomonal activity: Interaction index and killing curve method. Antimicrobial Agents and Chemotherapy 22: 743–752 (1982).
Hanatani, Y.; Fukutomi, T.; Yokoyama, I.; Arai, T.; Yamada, Y. and Saito, T.: Qinical research on penetration of antibiotics into bile. Chemotherapy (Tokyo) 31: 1042–1046 (1983).
Harding, S.M.: Clinical Pharmacology of ceftazidime; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981) pp.495–498 (American Society for Microbiology, Washington, D.C. 1982).
Harding, S.M. and Harper, P.B.: Pharmacokinetic behaviour of ceftazidime in man and the relationship between serum levels and the in vitro susceptibility of clinical isolates. Infection 11 (Suppl. 1): 49–53 (1983).
Harding, S.M.; Monro, A.J.; Thornton, J.E.; Ayrton, J.and Hogg, M.I.J.: Comparative pharmacokinetics of ceftazidime and cefotaxime in healthy volunteers. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 263–272 (1981a).
Harding, S.M.; Spencer, G.R. and Ryan, D.M.: A volunteer study to investigate the penetration of ceftazidime into inflammatory and non-inflammatory tissue fluids. (Abstract no. 175.) 21st Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago (Nov 1981b).
Harper, P.B.: In vitro properties of ceftazidime. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 5–13 (1981).
Harper, P.B. and Kirby, S.M.: In vitro properties of GR 20263. A highly active broad-spectrum cephalosporin with anti-pseudomonal activity. Drugs Under Experimental and Clinical Research 7: 179–187 (1981).
Harris, A.M.; Harper, P.B.; Plested, SJ. and Beniacova, E: Ceftazidime: In vitro activity against isolates resistant to amino-glycoside or β-lactam antibiotics and activity in combination with antibiotics in clinical use; in Pcriti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981) pp.478–480 (American Society for Microbiology, Washington D.C. 1982).
Hart, CA.; Barr, K.; Makin, T.; Brown, P. and Cooke, R.W.I.: Characteristics of a β-lactamase produced by Clostridium butyricum. Journal of Antimicrobial Chemotherapy 10: 31–35 (1982).
Hart, C.A. and Percival, A.: Resistance to cephalosporins among gentamicin-resistant Klebsiellae. Journal of Antimicrobial Chemotherapy 9: 275–286 (1982).
Haruta, T.; Ohkura, K.; Kuroki, S. and Kobayashi, Y.: Clinical evaluation of ceftazidime in paediatrics. Japanese Journal of Antibiotics 37: 524–536 (1984).
Hayes, M.V. and Orr, D.C: Mode of action of ceftazidime: affinity for the penicillin-binding proteins of Escherichia coli K12, Pseudomonas aeruginosa and Staphylococcus aureus. Journal of Antimicrobial Chemotherapy 12: 119–126 (1983).
Hernandez, N.; Rodriguez, J. and Ramirez-Ronda, C.H.: Effect of mezlocillin and third generation cephalosporins against nosocomial bacterial strains. (Abstract no. 936.) 22nd Interscience Conference of Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Höffler, P.; Koeppe, P. and Williams, K.J.: Pharmacokinetics of ceftazidime in normal and impaired renal function. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 241–245 (1983).
Hoffstedt, B.; Haidl, S. and Wälder, M.: Influence of probenecid on serum and subcutaneous tissue fluid concentrations of benzylpenicillin and ceftazidime in human volunteers. (Correspondence.) European Journal of Clinical Microbiology 2: 604–606 (1983).
Hoogkamp-Korstanje, J.A.A. and Meijer, C.A.: In vitro activity of ceftazidime, cefotaxime and piperacillin against multi-resistant Gram-negative bacteria tested with a modified agar dilution method. European Journal of Clinical Microbiology 1: 166–170 (1982).
Horowitz, M.A.; Youngworth, L.A.; Axelrod, J.L. and Kochman, R.S.: Ceftazidime concentrations in human aqueous humour. (Abstract no. 807.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Hoshina, H.; Mikuni, K. and Ichihashi, H.: Clinical studies of ceftazidime in pediatric field. Japanese Journal of Antibiotics 37: 460–468 (1984).
Hwang, P.T.R.; Drexler, P.G. and Meyer, M.C.: High-performance liquid Chromatographie determination of ceftazidime in serum, urine, CSF and peritoneal dialysis fluid. Journal of Liquid Chromatography 7: 979–987 (1984).
Ishibiki, K.: Clinical evaluation of ceftazidime in Japan. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 123–130 (1983).
Ito, K.; Kondo, H.; Hayasaki, M. and Noda, K.: Pharmacokinctic studies on ceftazidime in obstetrical and gynecological field. Japanese Journal of Antibiotics 37: 18–22 (1984).
Iwai, N.; Sasaki, A.; Taneda, Y.; Shibata, M.; Mizoguchi, F. and Nakamura, H.: Fundamental and clinical studies on ceftazidime in the field of pediatrics. Japanese Journal of Antibiotics 37: 490–511 (1984).
Jackson, M.A.; Kusmiesz, H. and Nelson, J.D.: Ceftazidime pharmacokinetics in pediatric patients. (Abstract no. 805.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Jones, R.N.; Barry, A.L. and Thomsberry, C: Disk agar diffusion susceptibility testing with 30μg ceftazidime disks: confirmation of interpretive breakpoints and quality control guidelines. Journal of Clinical Microbiology 18: 211–214 (1983).
Jones, R.N.; Barry, A.L.; Thornsberry, C; Gerlach, E.H.; Fuchs, P.C.; Gaven, T.L. and Sommers, H.M.: Ceftazidime, a Pseudomonas-active cephalosporin: in vitro antimicrobial activity evaluation including recommendations for disc diffusion susceptibility tests. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 187–211 (1981).
Jones, R.N. and Packer, R.R.: Antimicrobial activity of amikacin combinations against Enterobacteriaceae moderately susceptible to third-generation cephalosporins. Antimicrobial Agents and Chemotherapy 22: 985–989 (1982).
Kawamura, N.: Clinical experience with ceftazidime in urology in Japan. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 65–70 (1983).
Kayser, F.H.; Hornberger, F. and Morenzoni, G.: Influence of in vitro methods on phenotypic expression of bacterial susceptibility of cephalosporins; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th Internationa] Congress of Chemotherapy, Florence (July 1981) pp.368–370 (American Society for Microbiology, Washington D.C 1982b).
Kayser, F.H.; Morenzoni, G. and Hornberger, F.: Activity of cefoperazone against ampicillin-resistant bacteria in agar and broth dilution tests. Antimicrobial Agents and Chemotherapy 22: 15–22 (1982a).
Keeton, G.R.; Kehoe, B.; Phillips, S.W. and Daya, H.: Ceftazidime and cefamandole in the treatment of pneumonia. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 27–30 (1983).
Kemmcrich, B.; Warns, H.; Lode, H.; Borner, K.: Koeppe, P. and Knothe, H.: Multiple-dose pharmacokinetics of ceftazidime and its influence on fecal flora. Antimicrobial Agents and Chemotherapy 24: 333–338 (1983).
Kercsmar, CM.; Stern, R.C; Reed, M.D.; Myers, CM.; Murdell, D. and Blumer, J.L.: Ceftazidime in cystic fibrosis: Pharmacokinetics and therapeutic response. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 289–295 (1983).
Khan, M.Y.; Gruninger, R.P.; Nelson, S.M. and Obaid, S.R.: Comparative in vitro activity of cefodizime, ceftazidime, aztreonam, and other selected antimicrobial agents against Neisseria gonorrhoeae. Antimicrobial Agents and Chemotherapy 23: 477–478 (1983).
Kim, B.; Meguro, H.; Arimasu, O.; Mashiko, J.; Ohnari, S.; Tagaya, M.; Sakamoto, M. and Fujii, R.: Clinical evaluation of ceftazidime in the treatment of pediatric infection. Japanese Journal of Antibiotics 37: 363–376 (1984).
King, A.; Shannon, K.; Eykyn, S. and Phillips, I.: Reduced sensitivity to β-lactam antibiotics arising during ceftazidime treatment of Pseudomonas aeruginosa infections. Journal of Antimicrobial Chemotherapy 12: 363–370 (1983).
Klastersky, J.; Daneau, D.; Swings, G. and Weerts, P.: Antibacterial activity in serum and urine as a therapeutic guide in bacterial infections. Journal of Infectious Diseases 129: 187–193 (1974).
Knothe, H. and Dette, G.A.: In vitro activity of ceftazidime against clinically important pathogens. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 33–41 (1981).
Knothe, H. and Dette, G.A.: Current state of cephalosporin antibiotics: Microbiological aspects. Infection 11: 12–15 (1983).
Kohara, T.; Matsui, Y.; Mizutani, T. and Noda, M.: Experience with ceftazidime in the field of obstetrics and gynecology. Japanese Journal of Antibiotics 37: 6–13 (1984).
Koshinami, M. and Watanabe, A.: Clinical evaluation of ceftazidime in the field of pediatrics. Japanese Journal of Antibiotics 37: 344–348 (1984).
Kosmidis, J.; Stathakis, C; Giamardlou, H. and Daikos, G.K.: Ceftazidime in the treatment of severe infections due to organisms resistant to second generation cephalosporins. (Abstract no. 15.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Krasinski, K. and Nelson, J.D.: Pharmacokinetics and efficacy of ceftazidime in experimental Haemophilia influenzae b meningitis. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 339–343 (1981).
Kroll, M.; Koch, T.; Drusano, G.L. and Warren, J.W.: Moxalaclam, cefotaxime, cefoperazone and ceftazidime do not interfere with creatinine determinations in serum or urine. (Abstract no. 525.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Kuno, K.; Yafuso, M; Miyachi, Y.; Nakashima, T.; Ichihara, K. and Ueda, S.: Laboratory and clinical studies on ceftazidime in the field of pediatrics. Japanese Journal of Antibiotics 37: 469–484 (1984).
Lagast, H. and Klastersky, J.: Scrum bactericidal activity (SBA) of ceftazidime and amikacin alone or in combination against E. coli, K. pneumoniae and P. aeruginosa. (Abstract no. 172.) 21st Interscience Conference of Antimicrobial Agents and Chemotherapy, Chicago (Nov 1981).
Lamothe, F.; Auger, F. and Lacroix, J.-M.: Effect of clavulanic acid on the activities of ten β-lactam agents against members of the Bacteroides fragilis group. Antimicrobial Agents and Chemotherapy 25: 662–665 (1984).
Lautala, P.; Vare, M; Vuorinen, O. and Kaar, M.-L.: Ceftazidime in the treatment of chronic suppurative otitis media in children. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 365–367 (1983).
Le Bel, M.; Paone, R.P. and Lewis, G.P.: Lack of interference of five new beta-Iactam antibiotics with serum creatinine determination. Drug Intelligence and Clinical Pharmacy 17: 908–910 (1983).
Lechi, A.; Ghidini, O.; Luzzani, A.; Montesi, G.; Arosio, E.; Mengoni, M. and Passarella, E.: Clinical evaluation of ceftazidime in the treatment of infections caused by Pseudomonas and other Gram-negative bacteria. Drugs Under Experimental and Clinical Research 9: 665–669 (1983).
Leeder, J.S.; Spino, M; Isles, A.F.; Tcsoro, A.M.; Gold, R. and MacLeod, S.M.: Ceftrazidime disposition in acute and stable cystic fibrosis. Clinical Pharmacology and Therapeutics 6: 355–361 (1984).
Leeder, J.S.; Spino, M.; Tesoro, A.M. and MacLeod, S.M.: High pressure liquid Chromatographie analysis of ceftazidime in serum and urine. Antimicrobial Agents and Chemotherapy 24: 720–724 (1983).
Le Noc, P.; Robert, J. and Le Noc, D.: Activité comparée in vitro de cinq betalactamines (cefoperazone, cefsulodine, ceftrazidime, azthreonam, N-Formimidoyl-thiénamycine) sur 90 souches de Pseudomonas aeruginosa présentant différents niveaux de résistance à la ticarcilline. Pathologie Biologie 32: 404–407 (1984).
Lesage, D.; Delisle-Mizon, F.; Denis, G; Vergez, P. and Daguet, G.L.: Comparaison de l’effet bactériostatique in vitro de Pseudomonas aeruginosa. Pathologie Biologie 30: 440–443 (1982).
Linares, J.; Perez, J.L.; Garau, J.; Murgui, L. and Martin, R.: Comparative susceptibilities of penicillin-resistant pneumococci to co-trimoxazole, vancomycin, nifampicin and fourteen β-lactam antibiotics. Journal of Antimicrobial Chemotherapy 13: 393–399 (1984).
Livermore, D.M.; Williams, R.J. and Lindridge, M.A.: Pseudomonas aeruginosa isolates with modified beta-lactamase inducibility: Effects on beta-lactamase sensitivity. Lancet 1: 1466–1467 (1982).
Livermore, D.M.; Williams, R.J. and Williams, J.D.: In vitro activity of ceftazidime against Pseudomonas aeruginosa and its stability to pscudomonal β-lactamases. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 163–167 (1981a).
Livermore, D.M.; Williams, RJ. and Williams, J.D.: Comparison of the β-lactamase stability and the in vitro activity of cefoperazone, cefotaxime, cefsulodin, ceftazidime, moxalactam and ceftriaxone against Pseudomonas aeruginosa. Journal of Antimicrobial Chemotherapy 8: 323–331 (1981b).
Livermore, D.M. and Williams, R.J.: Role of plasmid-mediated beta-lactamases in the resistance of Pseudomonas aeruginosa to new bcta-lactams; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981) pp. 754–756 (American Society for Microbiology, Washington D.C. 1982).
Loebis, L.H. and Williams, K.J.: Ceftazidime in the treatment of complicated respiratory and urinary tract infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 59–64 (1983).
Lundbergh, P.; Jarstrand, C; Morfeldt-Mänson, L. and Weiland, O.: Ceftazidime in septicaemia. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 199–205 (1983).
Lüthy, R.; Blaser, J.; Bonetti, A.; Simmen, H.; Wise, R. and Siegenthaler, W.: Comparative multiple-dose pharmacokinetics of cefotaxime, moxalactam, and ceftazidime. Antimicrobial Agents and Chemotherapy 20: 567–575 (1981).
Mandell, L.A.; Nicolle, L.E.; Ronald, A.R.; Duperval, R.; Robson, H.G.; Feld, R.; Vincelette, J. and Fong, I.: Multicentre prospective randomized trial comparing ceftazidime with cefazolin/tobramycin in the treatment of hospitalized patients with non-pneumococcal pneumonia. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 9–20 (1983).
Marre, R.; Herhahn, D.; Schulz, E.; Freiesleben, H. and Sack, K.: Ceftazidime, ceftizoxime, cefotaxime and HR 221 in experimental chronic Escherichia coli pyelonephritis in rats. Infection 11 (Suppl. 1): 54–56 (1983).
Maskell, J.P.; Nasu, M. and Williams, J.D.: Susceptibility of Bacteroides to eight beta-lactam compounds including N-Formimidoyl Thienamycin (MK 0787): Study of resistant strains; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp.716–718 (American Society for Microbiology, Washington D.C. 1982).
Maskell, J.P.; Nasu, M. and Williams, J.D.: Cephalosporin-resistance in the Bacteroides fragilis group and the effect of clavulanic acid. Journal of Antimicrobial Chemotherapy 13: 23–30 (1984).
Maslow, M J.; Rosenberg, A.; Pollock, A.A.; Press, R.A.; Silverman, D.; El-Sadr, W.; Richmond, A.S.; Simberkoff, M.S. and Rahal Jr, J.J.: Ceftazidime therapy of infections caused by Enterobacteriaceae and Pseudomonas aeruginosa. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 213–217 (1983).
Mastella, G.; Agostini, M.; Barlocco, G.; Bonomi, U.; Borgo, G.; Bozzino, L.; Cabrini, G.; Cappelletti, L.M.; Castellani, L.; Conforti, M.; Marazzani, S.; Martini, N.; Montemezzi, P.; Paulon, G.; Pederzini, F.; Sancassani, L. and Scroccaro, G.: Alternative antibiotics for the treatment of Pseudomonas infections in cystic fibrosis. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 297–311 (1983).
McCracken, G.H.Jr.; Threlkeld, N. and Thomas, M.L.: Pharmacokinetics of ceftazidime in newborn infants. Antimicrobial Agents and Chemotherapy 26: 583–584 (1984).
Meyer, R.D. and Pasiecznik, K.: In vitro activity of newer β-lactam agents in combination with amikacin against Pseudomonas aeruginosa, Klebsiella pneumoniae and Serratia marcescens. Diagnostic Microbiology and Infectious Disease 1: 287–293 (1983).
Minamitani, M.; Hachimori, K. and Kaneda, K.: Clinical study on ceftazidime in the field of pediatrics. Japanese Journal of Antibiotics 37: 377–388 (1984).
Modai, J.; Vittecoq, D.; Decazes, J.M.; Wolff, M. and Meulemans, A.: Penetration of ceftazidime into cerebrospinal fluid of patients with bacterial meningitis. Antimicrobial Agents and Chemotherapy 24: 126–128 (1983).
Møller Heilesen, A.; Permin, H.; Koch, C. and Hoiby, N.: Treatment of chronic Pseudomonas aeruginosa infection in cystic fibrosis patients with ceftazidime and tobramycin. Scandinavian Journal of Infectious Diseases 15: 271–276 (1983).
Morgan, G.; Duerden, B.I. and Lilleyman, J.S.: Ceftazidime as a single agent in the management of children with fever and neutropenia. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 347–351 (1983).
Motohiro, T.; Tanaka, K.; Koga, T.; Shimada, Y.; Tomita, N.; Sakata, Y.; Fujimoto, T.; Nishiyama, T.; Ishimoto, K.; Tominaga, K. et al.: Fundamental and clinical studies of ceftazidime in the pediatric field. Japanese Journal of Antibiotics 37: 537–553 (1984).
Mouton, R.P.; Bongaerts, G.P.A. and van Gestel, M.H.: Susceptibility of ceftazidime and other cephalosporins to a range of β-lactamases and their potential as inducing agents. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 147–152 (1981).
Muytjens, H.L. and van der Rosvan de Repe, J.: Comparative activities of 13 β-lactam antibiotics. Antimicrobial Agents and Chemotherapy 21: 925–934 (1982).
Myers, C.M. and Blumer, J.L.: Determination of ceftazidime in biological fluids using high-pressure liquid chromatography. Antimicrobial Agents and Chemotherapy 24: 343–346 (1983).
Naber, K.G.; Kees, F. and Grobecker, H.: Ceftazidime: Pharmacokinetics in healthy volunteers versus elderly patients and therapeutic efficacy with complicated urinary tract infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 41–45 (1983).
Nagamatsu, I. and Horiguchi, S.: Clinical experience with ceftazidime in the infectious disease of children. Japanese Journal of Antibiotics 37: 338–343 (1984).
Nakamura, T.; Seto, S.; Kimura, K.; Sato, K.; Sato, H.; Fukada, F. and Nishikawa, S.: Fundamental and clinical studies on ceftazidime in the field of paediatrics. Japanese Journal of Antibiotics 37: 485–489 (1984).
Nakayama, I.: Clinical studies on ceftazidime in surgery in Japan. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 377–381 (1983).
Nakazawa, S.; Sato, H.; Niino, K.; Hirama, Y.; Narita, A.; Suzuki, H. and Nakazawa, S.: Fundamental and clinical studies of ceftazidime, a new cephem antibiotic in the field of pediatrics. Japanese Journal of Antibiotics 37: 407–422 (1984).
Nasu, M.; Maskell, J.P.; Williams, R.J. and Williams, J.D.: In vitro activity of MK 0787 (N-Formimidoyl Thienamycin) and other beta-lactam compounds against Bacieroides spp. Antimicrobial Agents and Chemotherapy 20: 433–436 (1981).
National Committee for Clinical Laboratory Standards: Performance Standards for Amimicrobic Disc Susceptibility Tests M2-T3, Vol. 3 (NCCLS, Villanova PA, October 1983).
Neu, H.C.: The new β-lactamase stable cephalosporins. Annals of Internal Medicine 97: 408–419 (1982a).
Neu, H.C.: Clinical uses of cephalosporins. Lancet 2: 252–255 (1982b).
Neu, H.C. and Labthavikul, P.: Antibacterial activity and β-laclamase stability of ceftazidime, an aminothiazolyl cephalosporin potentially active against Pseudomonas aeruginosa. Antimicrobial Agents and Chemotherapy 21: 11–18 (1982a).
Neu, H.C. and Labthavikul, P.: In vitro activity and β-lactamase stability of cefmenoxime. Antimicrobial Agents and Chemotherapy 22: 316–322 (1982b).
Neu, H.C. and Labthavikul, P.: In vitro activity and β-lactamase stability of a monobactam, SQ 26,917, compared with those of aztreonam and other agents. Antimicrobial Agents and Chemotherapy 24: 227–232 (1983).
Nishimura, T.; Tabuki, K; Takashima, T. and Takagi, M.: Laboratory and clinical studies of ceftazidime in the pediatric field. Japanese Journal of Antibiotics 37: 512–523 (1984).
Nolen, T.; Phillips, H.; Finn, A.; Pinkston, V. and Chubb, J.M.: Comparative evaluation of ceftazidime and cefamandole in the treatment of lower respiratory tract infections. (Abstract no. 17.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Norrby, S.R.: Ceftazidime in clinical practice —a summary. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 405–408 (1983).
Norrby, S.R.; Burman, L.A.; Linderholm, H. and Trollfors, B.: Ceftazidime: Pharmacokinetics in patients and effects on the renal function. Journal of Antimicrobial Chemotherapy 10: 199–206 (1982).
O’Callaghan, C.H.; Acred, P.; Harper, P.B.; Ryan, D.M.; Kirby, S.M. and Harding, S.M.: GR 20263, a new broad spectrum cephalosporin with antipseudomonal activity. Antimicrobial Agents and Chemotherapy 17: 876–883 (1980).
Ogawa, E.; Ota, H. and Maki, M.: Concentration of ceftazidime in the gynecological organs and clinical effects. Japanese Journal of Antibiotics 36: 3476–3482 (1983).
Padoan, R.; Brienza, A.; Crossignani, R.M.; Lodi, G.; Giunta, A.; Assael, B.M.; Granata, F.; Passarella, E.; Vallaperta, P.A. and Xerri, L.: Ceftazidime in treatment of acute pulmonary exacerbations in patients with cystic fibrosis. Journal of Pediatrics 103: 320–324 (1983).
Papathanassiou, B.; Kosmidis, J. and Daikos, G.K.: Comparative study of the concentration of cefoperazone and ceftazidime in bone; in Pcriti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 503–504 (American Society for Microbiology, Washington D.C. 1982).
Parish, L.C.; Snow, R.; Eron, L. and Mogabgab, W.J.: Prospective comparison of two dosage regimens of ceftazidime for therapy of skin and skin structure infections. (Data on file, Glaxo Laboratories).
Pechère, J.-C. and Delisle, R.: Open study of ceftazidime in serious infections due to multiply-resistant bacteria. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 181–188 (1983).
Pedler, S.J.: Activity in vitro of ceftazidime (GR 20263) and moxalactam (LY 127935) against Gram-negative bacteria from hospital patients. Journal of Antimicrobial Chemotherapy 8: 417–420 (1981).
Peirce, T.H.; Vig, S.J. and Ingram, P.M.: Ceftazidime in the treatment of lower respiratory tract infection. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 21–25 (1983).
Pennington, J.E.; Johnson, CE. and Platt, R.: Third generation cephalosporins in the treatment of pneumonia due to Pseudomonas aeruginosa in guinea pigs. Journal of Infectious Diseases 146: 567 (1982).
Permin, H.; Koch, G; Hoiby, N.; Christensen, H.O.; Moller, A.F. and Møller, S.: Ceftazidime treatment of chronic Pseudomonas aeruginosa respiratory tract infection in cystic fibrosis. Journal of Antimicrobial Chemotherapy 12 (Suppl. A) 313–323 (1983).
Petit, J.C.; Richard, G.; Burghoffer, B. and Daguet, G.L.: Activité synergique des associations ticarcilline, azlocilline, cefsulodine, ceftazidime et tobramycine ou amikacine vis-a-vis de Pseudomonas aeruginosa. Pathologie Biologie 30: 426–431 (1982).
Pettersson, T.; Storgards, E. and Ahvonen, P.: Treatment of lower respiratory tract infections with ceftazidime. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 31–34 (1983).
Philippon, A.; Paul, G. and Nevot, P.: Comparative in vitro activity of cefsulodin and ceftazidime against ticarcillin-resislant Pseudomonas aeruginosa. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 119–122 (1981).
Phillips, I.; King, A.; Shannon, K.; Warren, C. and Hanslo, D.: In vitro activity of ceftazidime compared with that of other β-lactam antibiotics and gentamicin against pseudomonas and enterobacteria; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 480–481 (American Society for Microbiology, Washington D.C 1982).
Phillips, I.; Warren, C; Shannon, K.; King, A. and Hanslo, D.: Ceftazidime: Invitroantibacterial activity and susceptibility to β-lactamases compared with that of cefotaxirne, moxalactam and other β-lactam antibiotics. Journal of Antimicrobial Chemotherapy 8 (Suppl. B) 23–31 (1981b).
Phillips, I.; Warren, C; Taylor, E.; Timewell, R. and Eykyn, S.: Antimicrobial susceptibility of anaerobic bacteria in a London teaching hospital. Journal of Antimicrobial Chemotherapy 8 (Suppl. D): 17–26 (1981a).
Piccolomini, R.; Cellini, L.; Arpini, A. and Ravagnan, G.: Comparative invitroactivity of ceftazidime and other beta-lactamase-stable cephalosporins against isolates from urinary tract infections. Chemioterapia 2: 45–50 (1983).
Piot, P.; Van Dyck, E. and Colaert, J.: Invitroactivity of ceftazidime (GR 20263) and other β-lactam antibiotics against Haemophilus influenzae. Infection 11 (Suppl. 1): 32–34 (1983).
PottageJr, J.C.; Karakusis, P.H.; Fliegelman, R.M.; Levin, S. and Trenholmc, G.M.: Ceftazidime in the therapy of serious Gram-negative bacillary infections. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 223–228 (1983).
Preblud, S.R.; Gill, CJ. and Campos, J.M.: Bactericidal activities of chloramphenicol and eleven other antibiotics against Salmonella spp. Antimicrobial Agents and Chemotherapy 25: 327–330 (1984).
Prinsloo, J.G.; Delport, S.D.; Moncrieff, J. and Paton, A.M.: Preliminary pharmacokinetic study of ceftazidime in premature, new bom and small infants. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 361–364 (1983).
Rabin, H.R. and Bryan, L.E.: Ceftazidime therapy of Pseudomonas aeruginosabronchopneumonitis in cystic fibrosis. Annals of the Royal College of Physicians and Surgeons of Canada 16: 350 (1983).
Ramphal, R.; Kramer, B.S.; Rand, K.H.; Werner, R.S. and Shands Jr, J.W.: Early results of a comparative trial of ceftazidime versuscephalothin, carbcnicillin and gentamicin in the treatment of febrile granulocytopenic patients. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 81–88 (1983).
Rapp, R.P.; Young, B.; Foster, T.S.; Tibbs, P.A. and O’Neal, W.: Ceftazidime versustobramycin/ticarcillin in treating hospital acquired pneumonia and bacteremia. Pharmacotherapy 4: 211–215 (1984).
Reed, M.; Murdell, D.; O’Brien, G; Aronoff, S. and Blumer, J.: Efficacy and safety of ceftazidime in serious pediatric infections. (Abstract no. 78.) 23rd Interscience Conference on Antimicrobial Agents and Chemotherapy, Las Vegas (Oct 1983).
Reeves, D.S.; Bywater, M.J. and Holt, H.A.: Comparative activity invitroof cefotetan, a new cephamycin antibiotic. Drugs Under Experimental and Clinical Research 9: 125–131 (1983b).
Reeves, D.S.; Holt, H.A. and Bywater, M.J.: Comparative activity invitroof ceftazidime and nine other antibacterial agents. Infection 11 (Suppl. 1): S3–S11 (1983a).
Reilly, J.T.; Brada, M.; Bellingham, A.J.; Hart, C.A. and Bennet, C: Ceftazidime compared to tobramycin and ticarcillin in immunocompromised haematological patients. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 89–92 (1983).
Ritzerfeld, W.: Ceftazidime activity in experimental Pseudomonas pyelonephritis in rats. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 257–259 (1981).
Rodriguez, J.; Hernandez, K; Fuxench, Z.; Nevarez, M. and Ramirez-Ronda, Ch.: Effect of combination antibiotics against gentamicin-resistant nosocomial strains from compromised hosts. (Abstract no. 111.) 2nd International Symposium on Infections in the Immunocompromised Host, Stirling, Scotland (June 1982).
Rolfe, R.D. and Finegold, S.M.: Comparative invitroactivity of new beta-lactam antibiotics against anaerobic bacteria. Antimicrobial Agents and Chemotherapy 20: 600–609 (1981).
Rolston, K.V.I.; Chandrasekar, P.H.; LeFrock, J.L. and Schell, R.F.: Activity of ceftazidime, other β-lactams, and aminoglycosides against Pseudomonasaeruginosa. Chemotherapy 30: 31–34 (1984).
Rusconi, F.; Assael, B.M.; Florioli, A. and Zaffaroni, G.: Ceftazidime in the treatment of pediatric patients with severe urinary tract infections due to Pseudomonasspp. Antimicrobial Agents and Chemotherapy 25: 395–397 (1984).
Ryan, D.M.; Hodges, B.; Spencer, G.R. and Harding, S.M.: Simultaneous comparison of three methods for assessing ceftazidime penetration into extravascular fluid. Antimicrobial Agents and Chemotherapy 22: 995–998 (1982).
Ryan, D.M.; Mason, U. and Harding, S.M.: Penetration of ceftazidime into extracellular fluid. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 283–288 (1981).
Saito, A.: Studies on absorption, distribution, metabolism and excretion of ceftazidime in Japan. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 255–262 (1983).
Sakata, Y.; Bocazzi, A. and McCracken, G.H. Jr. Pharmacokinetics and bacteriological effect of ceftazidime in experimental Streptococcuspneumoniae, Haemophilusinjluenzae, and Escherichia colimeningitis. Antimicrobial Agents and Chemotherapy 23: 213–217 (1983).
Sanders, C.C.: Novel resistance selected by the new expanded spectrum cephalosporins —a concern. Journal of Infectious Diseases 147: 585–589 (1983).
Sato, Y.; Iwata, S.; Iwasaki, Y.; Akita, H.; Murai, T.; Hayano, S.; Wakabayashi, R.; Sunakawa, K.; Oikawa, T. and Osano, M.: Basic and clinical studies on ceftazidime in pediatric field. Japanese Journal of Antibiotics 37: 389–406 (1984).
Schassan, H.H. and Fedder, J.: Activity of ceftazidime and newer antibiotics against Gram-negative bacilli. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 79–82 (1981).
Schassan, H.H.; Horning, R.; Malottke, R. and Potel, J.: Activity of thienamycin (MK 0787) in comparison with cefotaxime, moxalactam, ceftazidime, and other beta-lactams against clinically important Gram-negative nonfermenters and Enterobacteriaceae; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 735–737 (American Society for Microbiology, Washington D.C. 1982).
Schoengut, H. and Jelinek, R.: Comparative study of the effects of ceftazidime compared with tobramycin plus cefamandole in the treatment of gall bladder empyema. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 219–222 (1983).
Scribner, R.K.; Marks, MX; Weber, A.H. and Pai, C.H.: Yersinia enterocolitica:Comparative invitroactivities of seven new β-lactam antibiotics. Antimicrobial Agents and Chemotherapy 22: 140–141 (1982b).
Scribner, R.K.; Wedro, B.C.; Weber, A.H. and Marks, MX: Activities of eight new β-lactam antibiotics and seven antibiotic combinations against Neisseriameningitidis. Antimicrobial Agents and Chemotherapy 21: 678–680 (1982a).
Scully, B.E. and Neu, H.C.: Clinical efficacy of ceftazidime. Treatment of serious infection due to multiresistant Pseudomonas and other Gram-negative bacteria. Archives of Internal Medicine 144: 57–62 (1984).
Seibert, G.; Limbert, M.; Winkler, I. and Dick, T.: Antibacterial activity invitroand β-lactamase stability of the new cepha-losporin HR 810 in comparison with five other cephalosporins and two aminoglycosides. Infection 11: 275–279 (1983).
Selwyn, S. and Bakhtiar, M.: Practical significance of resistance to β-lactamases among new and established β-lactam antibiotics; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 756–758 (American Society for Microbiology, Washington D.C. 1982).
Shah, P.M.: Bactericidal activity of ceftazidime against Pseudomonas aeruginosaunder conditions simulating serum phar-macokinetics. Journal of Antimicrobial Chemotherapy 8 (Suppl. B) 135–140 (1981).
Shimizu, T.; Kanno, K.; Kawaguchi, H.; Yoshida, K. and Muto, T.: Influence of renal function on biliary excretion and clinical evaluation of a new antibiotic, ceftazidime, Chemotherapy (Tokyo) 31 (Suppl. 3): 697–702 (1983).
Shintani, M.; Takashima, K.; Kosakai, H.; Takayama, T.; Nakanishi, A.; Shimamoto, I. and Ichijo, M.: Fundamental and clinical studies of ceftazidime in the field of obstetrics and gynecology. Japanese Journal of Antibiotics 37: 28–32 (1984).
Simonian, S.; Meijer, J.H.; Allon, W.; McClaren, D.M.; Verhoef, J. and Oe, P.L.: Clearance of newly developed cephalosporins by artificial kidneys; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 595–596 (American Society for Microbiology, Washington D.C. 1982).
Simpson, I.N.; Plested, S.J. and Harper, P.B.: Investigation of the β-lactamase stability of ceftazidime and eight other new cepha-losporin antibiotics. Journal of Antimicrobial Chemotherapy 9: 357–368 (1982).
Singleton, B.L.: Killing kinetics of cefsulodin, ceftazidime, and gentamicin in Pseudomonasaeruginosa: in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 513–514 (American Society for Microbiology, Washington D.C. 1982).
Snelling, S.; Hart, C.A. and Cooke, R.W.I.: Ceftazidime or gentamicin plus benzylpenicillin in neonates less than 48 hours old. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 353–356 (1983).
So, S.Y.; Chau, P.Y.; Leung, Y.K., Lam, W.K. and Yu, D.Y.C.: Successful treatment of mclioidosis caused by a multiresistant strain in an immunocompromised host with third generation cephalosporins (case report). American Review of Respiratory Disease 127: 650–654 (1983).
Sommers, D.; Walters, L.; Van Wyk, M.; Harding, S.M.; Paton, A.M. and Ayrton, J.: Pharmacokinetics of ceftazidime in maie and female volunteers. Antimicrobial Agents and Chemotherapy 23: 892–896 (1983).
Spencer, G.R.; Hassani, H. and Harding, S.M.: Dosing recommendations for ceftazidime in patients with renal insufficiency, (abstract.) Clinical Research 31: 633A (1983).
Stobberingh, E.E.; Houben, A.W. and Van Boven, C.P.A.: Comparative activity of different anti-pseudomonas compounds. Drugs Under Experimental and Clinical Research 8: 633–635 (1982).
Strandberg, D.A.; Jorgensen, J.H. and Drutz, D.J.: Activities of aztreonam and new cephalosporins against infrequently isolated Gram-negative bacilli. Antimicrobial Agents and Chemotherapy 24: 282–286 (1983).
Strandvik, B.; Malmborg, A.-S.; Alfredson, H. and Ericsson, A.: Oinical results and pharmacokinetics of ceftazidime treatment in patients with cystic fibrosis. Journal ofAntimicrobial Chemotherapy 12 (Suppl. A): 283–287 (1983).
Straughn, A.B.; Meyer, M.C.; Acchiardo, S.; Chubb, J. and Corn-stock, T.J.: Pharmacokinetics of ceftazidime in normal subjects and end stage renal disease patients on hemodialysis. (Abstract no 801.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Takase, Z.: Experience with ceftazidime in obstretrics and gynaecology in Japan. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 383–387 (1983).
Terashima, I.; Nakamura, A.; Okimoto, Y.; Sugaya, N.; Kurosaki, T.; Himi, K.; Yamazaki, T. and Uehara, S.: Fundamental and clinical studies of ceftazidime in the field of pediatrics. Japanese Journal of Antibiotics 37: 355–362 (1984).
Thabaut, A.; Durosoir, J.L. and Meyran, M.: Activité des associations nouvelles bêtalactamines-aminoglycosides sur Pseudomonas aeruginosa. Pathologie Biologie 30: 549–554 (1982).
Thabaut, A.; Durosoir, J.L. and Saliou, P.: Activité comparée de six nouvelles céphalosporines sur les Pseudomonasaeruginosa sensibles et résistants à la carbénicilline et sur les entérobactéries résistantes à la céfazoline. Médicine et Maladies Infectieuses 11: 433–438 (1981a).
Thabaut, A.; Durosoir, J.L. and Saliou, P.: Comparison of the in vitroactivities of ceftazidime and new cephalosporins against 107 strains of Pseudomonasaeruginosaand 249 strains of cefazolin-resistant Enterobacteriaceae. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 123–125 (1981b).
Thornton, J.E.: Microbiological assay of ceftazidime. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 225–226 (1981).
Tjandramaga, T.B.; Van Hecken, A.; Mullie, A.; Verbesselt, R.; De Schepper, PJ. and Verbist, L.: Comparative pharmacokinetics of ceftazidime and moxalactam. Antimicrobial Agents and Chemotherapy 22: 237–241 (1982).
Tourkantonis, A. and Nicolaidis, P.: Pharmacokinetics of ceftazidime in patients undergoing peritoneal dialysis. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 263–267 (1983).
Toyonaga, Y.; Kurosu, Y.; Sugita, M.; Kawamura, G.; Matsunaga, K.; Kawahito, N.; Mochizuki, R; Ida, H.; Nakamura, H.; Kawamura, K. et al.: Fundamental and clinical studies on ceftazidime in the field of pediatrics. Japanese Journal of Antibiotics 37: 423–458 (1984).
Ullmann, U.: Distribution of Klebsiellapneumoniaeserotypes from different sources and their sensitivity to cephalosporins. Infection 11 (Suppl. 1): 28–31 (1983).
van Dalen, R.; Muytjens, H.L. and Gimbrére, J.S.F.: Ceftazidime treatment in intensive care patients. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 189–197 (1983).
Vanhoof, R.; Coignau, H.; Stas, G.; Gordts, B. and Butzler, J.P.: Bactericidal activity of ceftazidime against cephalothin-resistant, Gram-negative clinical isolates; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 485–486 (American Society for Microbiology, Washington D.C. 1982).
Van Klingeren, B.; Van Wijngaarden, L.J. and Rutgers, A.: Comparative susceptibility of Pseudomonasaeruginosato four cephalosporins. (Correspondence.) Journal of Antimicrobial Chemotherapy 6: 674–675 (1980).
Van Laethem, Y.; Gordts, B.; Clumeck, N.; Vanderauwera, P. and Butzler, J.P.: Clinical experience with third generation cephalosporins in multiresistant gram negative bacteremia. (Abstract no 20.) 22nd Interscience Conference on Antimicrobial Agents and Chemotherapy, Florida (Oct 1982).
Van Laethem, Y.; Lagast, H.; Husson, M. and Klastersky, J.: Serum bactericidal activity of cefoperazone and ceftazidime at increasing dosages against Pseudomonasaeruginosa. Journal of Antimicrobial Chemotherapy 12: 475–480 (1983b).
Van Laethem, Y.; Lagast, H. and Klastersky, J.: Serum bactericidal activity of ceftazidime and cefoperazone alone or in combination with amikacin against Pseudomonasaeruginosaand Klebsiellapneumoniae. Antimicrobial Agents and Chemotherapy 23: 435–439 (1983a).
Van Landuyt, H.W.; Denolf, B.L.L.; Pyckavet, M. and Lambert, A.M.: Invitroactivity of ceftazidime against resistant clinical isolates. Infection 11 (Suppl. 1): 23–27 (1983).
Van Landuyt, H.W.; Pyckavet, M.; Lambert, A.M. and Boelaert, J.: Invitroactivity of temocillin (BRL 17421), a novel β-lactam antibiotic. Antimicrobial Agents and Chemotherapy 22: 535–540 (1982)
Verbist, L.: Comparison of m vitroactivities of eight β-lactamase-stable cephalosporins against β-lactamase-producing Gram-negative bacteria. Antimicrobial Agents and Chemotherapy 19: 407–413 (1981).
Verbist, L. and Verhaegen, J.: GR-20263: A new aminothiazolyl cephalosporin with high activity against Pseudomonasand Enterobacteriaceae. Antimicrobial Agents and Chemotherapy 17: 807–812 (1980).
Verbist, L. and Verhaegen, J.: Ceftazidime: comparative invitro study. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 67–71 (1981a).
Verbist, L. and Verhaegen, J.: Invitroactivity of N-formimidoyl thienamycin in comparison with cefotaxime, moxalactam, and ceftazidime. Antimicrobial Agents and Chemotherapy 19: 402–406 (1981b).
Vetter, N.; Feist, H.; Muhar, F. and Williams, K.J.: Comparative study of the efficacy of ceftazidime versusccfazolin and tobramycin in patients with acute exacerbations of chronic bronchitis. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 35–39 (1983).
von Graevenitz, A. and Bucher, C: Effect of N-formimidoyl thienamycin, ceftazidime, cefotiam, ceftriaxone and cefotaxime on non-fermentative Gram-negative rods, Aeromonas, Plesiomonasand Enterobacteragglomerons. Infection 10: 293–298 (1982).
Walstad, R.A.; Helium, K.B.; Blika, S.; Dale, L.G.; Fredriksen, T.; Myhre, K.I. and Spencer, G.R.: Pharmacokinetics and tissue penetration of ceftazidime: Studies on lymph, aqueous humour, skin blister, cerebrospinal and pleural fluid. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 275–282 (1983a).
Walstad, R.A.; Helium, KB.; Svarva, P.L. and Ingram, P.M.: Evaluation of ceftazidime in the treatment of bacterial infections in eighty seriously ill patients. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 131–137 (1983b).
Wardle, J.K.; Selkon, J.B.; Ingham, H.R. and Freeman, R.: Ceftazidime in Gram-negative infections: Three case reports. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 313–316 (1981).
Warns, H.; Lode, H.; Harnoss, CM.; Kemmerich, B.; Koeppe, P. and Wagner, J.: Multiple dose pharmacokinetics and therapeutic results with ceftazidime. Journal of Antimicrobial Chemotherapy 12 (Suppl. A): 235–240 (1983).
Welage, L.S.; Schultz, R.W. and Schentag, J.J.: Pharmacokinetics of ceftazidime in patients with renal insufficiency. Antimicrobial Agents and Chemotherapy 25: 201–204 (1984).
Wise, R.; Andrews, J.M. and Bedford, K.A.: Comparison of in vitroactivity of GR 20263, a novel cephalosporin derivative, with activities of other beta-lactam compounds. Antimicrobial Agents and Chemotherapy 17: 884–889 (1980).
Wise, R.; Armstrong, G.C.; Brown, R.M. and Andrews, J.M.: Pharmacokinetics and tissue penetration of ceftazidime and cefamandole in healthy volunteers. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 277–282 (1981).
Wittmann, D.H. and Schassan, H.-H.: Bone concentrations of 6 new β-lactam antibiotics; in Periti, P. and Grassi, G.G. (Eds) Current Chemotherapy and Immunotherapy. Proceedings of the 12th International Congress of Chemotherapy, Florence (July 1981), pp. 628–630 (American Society for Microbiology, Washington D.C. 1982).
Wittmann, D.H. and Schassan, H.-H.: Penetration of eight β-lactam antibiotics into the peritoneal fluid. Archives of Surgery 118: 205–213 (1983).
Wittman, D.H.; Schassan, H.-H.; Kohler, F. and Seibert, W.: Pharmacokinetic studies of ceftazidime in serum, bone, bile, tissue fluid and peritoneal fluid. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 293–297 (1981).
Xerri, L.; Orsolini, P.; Caro, B.; Erani, E. and Coppi, G.: Invitro and invivoactivity of ceftazidime (GR-20263) and other β-lactam and aminoglycoside antibiotics against hospital strains. Chemioterapia 2: 333–336 (1983).
Yamamoto, T.; Yasuda, J.; Kanao, M. and Okada, H.: Fundamental and clinical studies on ceftazidime in the field of obstetrics and gynecology. Japanese Journal of Antibiotics 37: 23–27 (1984).
Yokota, T. and Sekiguchi, R.: Ceftazidime (SN 401): Penetrability through the outer membrane and the affinity for the penicillin-binding proteins of Gram-negative bacteria. Chemotherapy (Tokyo) 31 (Suppl. 3): 17–21 (1983).
Zuccoli, P.; Ho, I.; Mergoni, M. and Melley, L.: Evaluation of ceftazidime in the treatment of severe infection. Journal of Antimicrobial Chemotherapy 8 (Suppl. B): 311–312 (1981).
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: K. Alestig, Department of Infectious Diseases, University of Göteborg, Göteborg, Sweden; A.J. Bint, Department of Microbiology, Royal Victoria Infirmary, Newcastle upon Tyne, England; A.W. Chow, Division of Infectious Diseases, University of British Columbia, Vancouver, Canada; P.J. Darbyshire, Department of Haematology and Oncology, Hospitals for Sick Children, London, England; F. Daschner, Department of Hospital Epidemiology, Klinikum Der Albert-Ludwigs-Universitat, Freiburg, West Germany; V. Fainstein, Department of Internal Medicine, University of Texas System Cancer Center, Houston, Texas, USA; H. Giamarellou, Infectious Diseases, Athens University School of Medicine, Athens, Greece; K. Ishibiki, School of Medicine, Keio University, Tokyo, Japan; R.N. Jones, Kaiser Permanente Medical Care Program, Clackamas, Oregon, USA; R.W. Lacey, Department of Microbiology, University of Leeds, Leeds, England; D.M. Livermore, Department of Medical Microbiology, London Hospital Medical College, University of London, London, England; L.A. Mandell, Division of Infectious Diseases, McMaster University, Hamilton, Ontario, Canada; G. Mastella, Instituti Ospitalierei di Verona, Verona, Italy; H.C. Neu, Division of Infectious Diseases, College of Physicians and Surgeons of Columbia University, New York, New York, USA; R. Walstad, Department of Pharmacology and Toxicology, University of Trondheim, Trondheim, Norway; L. Weinstein, Brigham and Women’s Hospital, Boston, Massachusetts, USA
‘Fortam’, ‘Fortaz’, ‘Fortum’, ‘Glazidim’, ‘Modacin’ (Glaxo Laboratories)
Rights and permissions
About this article
Cite this article
Richards, D.M., Brogden, R.N. Ceftazidime. Drugs 29, 105–161 (1985). https://doi.org/10.2165/00003495-198529020-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-198529020-00002