Abstract
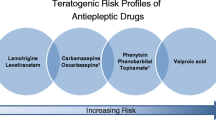
The treatment of epilepsy in pregnancy is particularly challenging in that the fetal and maternal risks associated with maternal seizures need to be balanced against the potential teratogenic effects of antiepileptic drugs (AEDs). Pregnancy is known to affect the pharmacokinetics of older-generation AEDs. Understanding such alterations is important in the effort to optimise drug therapy since they may affect seizure control as well as fetal drug exposure. Therapeutic drug monitoring has therefore been recommended to control for changes in the disposition of the older-generation AEDs during pregnancy. Much less is known about gestation-induced alterations in the pharmacokinetics of the newer AEDs that have been introduced in the last 15 years. Lamotrigine is by far the most extensively studied of the newer AEDs. Pronounced alterations have been reported in the apparent clearance of lamotrigine, with an increase of >300% from baseline in late pregnancy in some patients on monotherapy, most likely due to enhanced metabolism. The available data suggest that the corresponding decline in plasma concentrations can be associated with loss of seizure control. More limited data indicate that a similar decline in plasma concentrations of the active monohydroxy derivative of oxcarbazepine may occur in late pregnancy. Preliminary experience also suggests that a significant fall in plasma concentrations of levetiracetam may occur during pregnancy. No systematic information is available on the pharmacokinetics during pregnancy of other newer AEDs (e.g. gabapentin, pregabalin, tiagabine, topiramate or zonisamide). Given the importance of maintaining optimal treatment of epilepsy during pregnancy, therapeutic drug monitoring appears to be justified for lamotrigine and oxcarbazepine in particular. Systematic studies of the effects of pregnancy on the pharmacokinetics of the other newer-generation AEDs are urgently needed.



Similar content being viewed by others
References
Gaily E. Development and growth in children of epileptic mothers: a prospective controlled study [thesis]. Helsinki: University of Helsinki, 1990
Perucca E. Birth defects after prenatal exposure to antiepileptic drugs. Lancet Neurol 2005 Nov; 4(11): 781–6
Tomson T, Perucca E, Battino D. Navigating toward fetal and maternal health: the challenge of treating epilepsy in pregnancy. Epilepsia 2004 Oct; 45(10): 1171–5
Adab N, Kini U, Vinten J, et al. The longer term outcome of children born to mothers with epilepsy. J Neurol Neurosurg Psychiatry 2004 Nov; 75(11): 1575–83
Teramo K, Hiilesmaa V, Bardy A, et al. Fetal heart rate during a maternal grand mal epileptic seizure. J Perinat Med 1979; 7(1): 3–6
Teramo K, Hiilesmaa V. Pregnancy and fetal complications in epileptic pregnancies. In: Janz D, Dam M, Bossi L, et al., editors. Epilepsy, pregnancy, and the child. New York: Raven Press, 1982: 53–9
EURAP Study Group. Seizure control and treatment in pregnancy: observations from the EURAP epilepsy pregnancy registry. Neurology 2006 Feb 14; 66(3): 354–60
Guidelines for the care of epileptic women of childbearing age. Commission on Genetics, Pregnancy, and the Child, International League Against Epilepsy. Epilepsia 1989 Jul–Aug; 30(4): 409–10
Delgado-Escueta AV, Janz D. Consensus guidelines: preconception counseling, management, and care of the pregnant woman with epilepsy. Neurology 1992 Apr; 42(4 Suppl. 5): 149–60
Yerby MS, Friel PN, McCormick K. Antiepileptic drug disposition during pregnancy. Neurology 1992 Apr; 42(4 Suppl. 5): 12–6
Guidelines for the care of women of childbearing age with epilepsy. Commission on Genetics, Pregnancy, and the Child, International League Against Epilepsy. Epilepsia 1993 Jul–Aug; 34(4): 588–9
Committee on Educational Bulletins of the American College of Obstetricians and Gynecologists. Seizure disorders in pregnancy. ACOG educational bulletin number 231, December 1996. Int J Gynaecol Obstet 1997 Mar; 56(3): 279–86
Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter: management issues for women with epilepsy (summary statement). Epilepsia 1998 Nov; 39(11): 1226–31
Crawford P, Appleton R, Betts T, et al. Best practice guidelines for the management of women with epilepsy. Seizure 1999 Jun; 8(4): 201–17
Scottish Intercollegiate Guidelines Network. Diagnosis and management of epilepsy in adults: a national clinical guideline. Guideline number 70 [online]. Edinburgh: Royal College of Physicians, 2003. Available from URL: http://www.sign.ac.uk/guidelines/fulltext/70/index.html [Accessed 2007 Jan 8]
UK National Institute for Clinical Excellence. Newer drugs for epilepsy in adults: technology appraisal 76 [online]. London: National Institute for Clinical Excellence, 2004. Available from URL: http://www.nice.org.uk/pdf/ta076fullguidance.pdf [Accessed 2007 Jan 8]
Perucca E. Drug metabolism in pregnancy, infancy and childhood. Pharmacol Ther 1987; 34(1): 129–43
Pennell PB. Antiepileptic drug pharmacokinetics during pregnancy and lactation. Neurology 2003 Sep 1;61 (6 Suppl. 2): S35–42
Commission on Genetics, Pregnancy, and the Child, International League Against Epilepsy. Guidelines for the care of epileptic women of childbearing age. Epilepsia 1989 Jul–Aug; 30(4): 409–10
Anderson GD. Pregnancy-induced changes in pharmacokinetics: a mechanistic-based approach. Clin Pharmacokinet 2005; 44(10): 989–1008
Perucca E, Crema A. Plasma protein binding of drugs in pregnancy. Clin Pharmacokinet 1982; 7(4): 336–52
Yerby MS, Friel PN, McCormick K, et al. Pharmacokinetics of anticonvulsants in pregnancy: alterations in plasma protein binding. Epilepsy Res 1990 Apr; 5(3): 223–8
Dansky L, Andermann E, Shervin A, et al. Plasma levels of pheytoin during pregnancy and the puerperium. In: Janz D, Dam M, Bossi L, et al., editors. Epilepsy, pregnancy, and the child. New York: Raven Press, 1982: 155–62
Bardy AH, Hiilesmaa VK, Teramo KA. Serum phenytoin during pregnancy, labor and puerperium. Acta Neurol Scand 1987 Jun; 75(6): 374–5
Battino D, Avanzini G, Bossi L, et al. Monitoring of antiepileptic drugs plasma levels during pregnancy and puerperium. In: Janz D, Dam M, Bossi L, et al., editors. Epilepsy, pregnancy and the child. New York: Raven Press, 1982: 147–54
Tomson T, Lindbom U, Ekqvist B, et al. Disposition of carbamazepine and phenytoin in pregnancy. Epilepsia 1994 Jan–Feb; 35(1): 131–5
Tomson T, Lindbom U, Ekqvist B, et al. Epilepsy and pregnancy: a prospective study of seizure control in relation to free and total plasma concentrations of carbamazepine and phenytoin. Epilepsia 1994 Jan–Feb; 35(1): 122–30
Battino D, Binelli S, Bossi L, et al. Plasma concentrations of carbamazepine and carbamazepine 10, 11-epoxide during pregnancy and after delivery. Clin Pharmacokinet 1985 May–Jun; 10(3): 279–84
Yerby MS, Friel PN, Miller DQ. Carbamazepine protein binding and disposition in pregnancy. Ther Drug Monit 1985; 7(3): 269–73
Bernus I, Hooper WD, Dickinson RG, et al. Metabolism of carbamazepine and co-administered anticonvulsants during pregnancy. Epilepsy Res 1995 May; 21(1): 65–75
Battino D, Binelli S, Bossi L, et al. Changes in primidone/phenobarbitone ratio during pregnancy and the puerperium. Clin Pharmacokinet 1984 May–Jun; 9(3): 252–60
Rating D, Nau H, Jager-Roman E, et al. Teratogenic and pharmacokinetic studies of primidone during pregnancy and in the offspring of epileptic women. Acta Paediatr Scand 1982 Mar; 71(2): 301–11
Omtzigt JG, Los FJ, Meijer JW, et al. The 10, 11-epoxide-10,11-diol pathway of carbamazepine in early pregnancy in maternal serum, urine, and amniotic fluid: effect of dose, comedication, and relation to outcome of pregnancy. Ther Drug Monit 1993 Feb; 15(1): 1–10
Philbert A, Pedersen B, Dam M. Concentration of valproate during pregnancy, in the newborn and in breast milk. Acta Neurol Scand 1985 Nov; 72(5): 460–3
Stewart BH, Kugler AR, Thompson PR, et al. A saturable transport mechanism in the intestinal absorption of gabapentin is the underlying cause of the lack of proportionality between increasing dose and drug levels in plasma. Pharm Res 1993 Feb; 10(2): 276–81
McLean MJ. Gabapentin. Epilepsia 1995; 36 Suppl. 2: S73–86
Vollmer KO, von Hodenberg A, Kölle EU. Pharmacokinetics and metabolism of gabapentin in rat, dog and man. Arzneim Forsch Drug Res 1988; 36: 830–9
Öhman I, Vitols S, Tomson T. Pharmacokinetics of gabapentin during delivery, in the neonatal period, and lactation: does a fetal accumulation occur during pregnancy? Epilepsia 2005 Oct; 46(10): 1621–4
Fitton A, Goa KL. Lamotrigine: an update of its pharmacology and therapeutic use in epilepsy. Drugs 1995 Oct; 50(4): 691–713
Hussein Z, Posner J. Population pharmacokinetics of lamotrigine monotherapy in patients with epilepsy: retrospective analysis of routine monitoring data. Br J Clin Pharmacol 1997 May; 43(5): 457–65
Tomson T, Ohman I, Vitols S. Lamotrigine in pregnancy and lactation: a case report. Epilepsia 1997 Sep; 38(9): 1039–41
Ohman I, Vitols S, Tomson T. Lamotrigine in pregnancy: pharmacokinetics during delivery, in the neonate, and during lactation. Epilepsia 2000 Jun; 41(6): 709–13
Tran TA, Leppik IE, Blesi K, et al. Lamotrigine clearance during pregnancy. Neurology 2002 Jul 23; 59(2): 251–5
Pennell PB, Newport DJ, Stowe ZN, et al. The impact of pregnancy and childbirth on the metabolism of lamotrigine. Neurology 2004 Jan; 62(2): 292–5
de Haan GJ, Edelbroek P, Segers J, et al. Gestation-induced changes in lamotrigine pharmacokinetics: a monotherapy study. Neurology 2004 Aug; 63(3): 571–3
Petrenaite V, Sabers A, Hansen-Schwartz J. Individual changes in lamotrigine plasma concentrations during pregnancy. Epilepsy Res 2005 Jul; 65(3): 185–8
Pennell PB, Ritchie JC, Newport DJ, et al. Defining fetal exposure to lamotrigine [abstract]. Epilepsia 2004 Oct; 45 Suppl. 7: 233
Garnett WR. Lamotrigine: pharmacokinetics. J Child Neurol 1997 Nov; 12 Suppl. 1: S10–5
Tomson T, Luef G, Sabers A, et al. Valproate effects on kinetics of lamotrigine in pregnancy and treatment with oral contraceptives. Neurology 2006; 37: 1297–9
Patsalos PN, Perucca E. Clinically important drug interactions in epilepsy: general features and interactions between antiepileptic drugs. Lancet Neurol 2003 Jun; 2(6): 347–56
Patsalos PN. Pharmacokinetic profile of levetiracetam: toward ideal characteristics. Pharmacol Ther 2000 Feb; 85(2): 77–85
Radtke RA. Pharmacokinetics of levetiracetam. Epilepsia 2001 Aug; 42 Suppl. 4: 24–7
Johannessen SI, Heide G, Brodtkorb E. Levetiracetam concentrations in serum and in breast milk at birth and during lactation. Epilepsia 2005 May; 46(5): 775–7
Penneil PB, Roganti A, Helmers S, et al. The impact of pregnancy and childbirth on the elimination of levetiracetam [abstract]. Epilepsia 2005 Oct; 46 Suppl. 8: 89
May TW, Korn-Merker E, Rambeck B. Clinical pharmacokinetics of oxcarbazepine. Clin Pharmacokinet 2003; 42(12): 1023–42
Mazzucchelli I, Onat FY, Ozkara C, et al. Changes in the disposition of oxcarbazepine and its metabolites during pregnancy and the puerperium. Epilepsia 2006 Mar; 47(3): 504–9
Langtry HD, Gillis JC, Davis R. Topiramate: a review of its pharmacodynamic and pharmacokinetic properties and clinical efficacy in the management of epilepsy. Drugs 1997 Nov; 54(5): 752–73
Adin J, Gomez MC, Blanco Y, et al. Topiramate serum concentration-to-dose ratio: influence of age and concomitant antiepileptic drugs and monitoring implications. Ther Drug Monit 2004 Jun; 26(3): 251–7
Ohman I, Vitols S, Luef G, et al. Topiramate kinetics during delivery, lactation, and in the neonate: preliminary observations. Epilepsia 2002 Oct; 43(10): 1157–60
Perucca E, Bialer M. The clinical pharmacokinetics of the newer antiepileptic drugs: focus on topiramate, zonisamide and tiagabine. Clin Pharmacokinet 1996 Jul; 31(1): 29–46
Seino M, Naruto S, Ito T, et al. Zonisamide. In: Levy RH, Mattson RH, Meldrum BS, editors. Antiepileptic drugs, 4th ed. New York: Raven Press, 1995: 1001–24
Kawada K, Itoh S, Kusaka T, et al. Pharmacokinetics of zonisamide in perinatal period. Brain Dev 2002 Mar; 24(2): 95–7
Ben-Menachem E. Pregabalin pharmacology and its relevance to clinical practice. Epilepsia 2004; 45 Suppl. 6: 13–8
Adkins JC, Noble S. Tiagabine: a review of its pharmacodynamic and pharmacokinetic properties and therapeutic potential in the management of epilepsy. Drugs 1998 Mar; 55(3): 437–60
Johannessen SI, Battino D, Berry DJ, et al. Therapeutic drug monitoring of the newer antiepileptic drugs. Ther Drug Monit 2003 Jun; 25(3): 347–63
Adab N. Birth defects and epilepsy medication. Expert Rev Neurother 2006 Jun; 6(6): 833–45
Acknowledgements
No sources of funding was used in the preparation of this manuscript. Torbjörn Tomson has received speaker’s honoraria from GSK, UCB, Novartis, Pfizer and Sanofi-Aventis, and has received grants from GSK, UCB, Novartis, Pfizer, Sanofi-Aventis and Janssen-Cilag.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tomson, T., Buttino, D. Pharmacokinetics and Therapeutic Drug Monitoring of Newer Antiepileptic Drugs During Pregnancy and the Puerperium. Clin Pharmacokinet 46, 209–219 (2007). https://doi.org/10.2165/00003088-200746030-00002
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200746030-00002