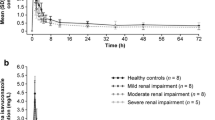
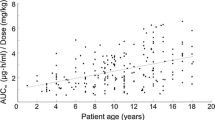
Summary
Many drugs are eliminated via the renal route and the usual dose must be modified in patients with severe renal impairment. This review is an attempt to supply physicians with the more recent data on pharmacokinetic studies of new drugs administered in uraemic patients. The review is in 2 parts: the first indicates the results of studies on the pharmacokinetics of antibiotic agents, antifungal, antiviral and antiulcer drugs, and nonsteroidal anti-inflammatory drugs. Special mention is made of epoetin (recombinant human erythropoietin). It was not possible to give all the information collected from the recent literature: since mild renal failure has little effect on the fate of a drug, pharmacokinetic data obtained in patients with a creatinine clearance (CLCR) of more than 50 ml/min has been omitted. Both the text and tables give recommendations for treating patients with moderate renal insufficiency (CLCR of about 50 ml/min), more severe renal impairment (CLCR between 10 and 50 ml/min) and end-stage renal failure with a very low creatinine clearance (below 10 ml/min). It was not possible to give uniform recommendations (i.e. reducing the dose while maintaining the same interval, or giving the same dose and prolonging the interval). This article follows the recommendations of the authors, which may vary for drugs in similar classes.
Similar content being viewed by others
References
Aronoff GR, Bergstrom RF, Bopp RJ, Sloan RS, Callaghan JT. Nizatidine disposition in subjects with normal and impaired renal fucntion. Clinical Pharmacology and Therapeutics 43: 688–695, 1988
Ashby JP, Nye KJ, Andrews JM, Wise R, Shi YG. The pharmacokinetics and tissue penetration of cefepime following a 2g IV infusion. 16th International Congress of Chemotherapy, Jerusalem, Israel, June 11–16, (Abstract, p. 308), 1989
Audet PR, Knowles JA, Troy SM, Waker BR, Morrisson G. Effect of chronic renal failure on oxaprozin multiple-dose pharmacokinetics. Clinical Pharmacology and Therapeutics 44: 303–309, 1988
Barbhaiya RH, Knupp CA, Forgue ST, Matzke GR, Guay DRP, et al. Pharmacokinetics of cefepime in subjects with renal insufficiency. Clinical Pharmacology and Therapeutics 48: 268–276, 1990
Bennett WM. Guide to drug dosage in renal failure. In Mammen GJ (Ed.) Clinical Pharmacokinetics drug data, handbook, pp. 39–90, Adis Press, Auckland, 1989
Bjaeldager PRL, Jensen JB, Larsen NE, Hvidberg EF. Elimination of oral cimetidine in chronic renal failure and during haemodialysis. British Journal of Clinical Pharmacology 7: 585–592, 1980
Blouin RA, Kneer J, Stoechel K. Pharmacokinetics of intravenous cefetamet (RO 15-8074) and oral cefetamet pivoxil (RO 15-8075) in young and elderly subjects. Antimicrobial Agents and Chemotherapy 33: 291–296, 1989
Blum RA, Kohli RK, Harrison NJ, Schentag JJ. Pharmacokinetics of ampicillin (2.0 grams) and sulbactam (1.0 gram) coadministered to subjects with normal and abnormal renal function and with end stage renal disease on hemodialysis. Antimicrobial Agents and Chemotherapy 33: 1470–1476, 1989
Boelaert JR, Jonnaert HA, Daneels RG, Schurgers ML, Thawley AR, et al. Nabumetone pharmacokinetics in patients with varying degrees of renal impairment. American Journal of Medicine 83 (Suppl. 48): 107–109, 1987
Boelaert JR, Schurgers ML, Matthys EV, Belpaire FM, Daneels RF, et al. Comparative pharmacokinetics of recombinant erythropoietin administered by the intravenous, subcutaneous and intraperitoneal routes in continuous ambulatory peritoneal dialysis patients. Peritoneal Dialysis International 9: 95–98, 1989
Boelaert J, Schurgers M, Matthys E, Daneels R, Van Peer A, et al. Itraconazole pharmacokinetics in patients with renal dysfunction. Antimicrobial Agents and Chemotherapy 32: 1595–1597, 1988
Bonati M, Traina GL, Villa G, Salvadeo A, Gentile MG, et al. Teicoplanin pharmacokinetics in patients with chronic renal failure. Clinical Pharmacokinetics 12: 292–301, 1987
Borsa F, Leroy A, Fillastre JP, Godin M, Moulin B. Comparative pharmacokinetics of tromethamine fosfomycin and calcium fosfomycin in young and elderly adults. Antimicrobial Agents and Chemotherapy 32: 938–941, 1988
Brammer KW, Tarbit MH. A review of the pharmacokinetics of fluconazole in laboratory animals and man. In Fromtiling (Ed.) Recent trends in the discovery development and evaluation of antifungal agents, pp. 141–149, Prous, Barcelona, 1987
Brittain DC, Scully BE, Hirose T, Neu HC. The pharmacokinetic and bactericidal characteristics of oral cefixime. Clinical Pharmacology and Therapeutics 38: 590–594, 1985
Brouard RJ, Kapusknik JE, Gambertoglio JG, Schoenfeld PY, Sachdeva M, et al. Teicoplanin pharmacokinetics and bioavailability during peritoneal dialysis. Clinical Pharmacology and Therapeutics 45: 674–681, 1989
Campoli-Richards DM. Clissold SP. Famotidine: pharmacodynamic and pharmacokinetic properties and a preliminary review of its therapeutic use in peptic ulcer disease and Zollinger-Ellison syndrome. Drugs 32: 197–221, 1986
Capparelli EV, Stevens RC, Chow MSS, Izard M, Wills RJ. Rimantadine pharmacokinetics in healthy subjects and patients with end stage renal failure. Clinical Pharmacology and Therapeutics 43: 536–541, 1988
Carmine AA, Brogden RN. Pirenzepine: a review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy in peptic ulcer disease and other allied diseases. Drugs 30: 85–126, 1985
Chu DTW, Fernandes PB. Structure-activity relationships of the fluoroquinolones. Antimicrobial Agents and Chemotherapy 33: 131–135, 1989
Clissold SP, Campoli-Richards DM. Omeprazole: a preliminary review of its pharmacodynamic and pharmacokinetic properties and therapeutic potential in peptic ulcer disease and Zollinger-Ellison syndrome. Drugs 32: 15–47, 1986
Collins JD, Pidgen AW. Pharmacokinetics of roxatidine in healthy volunteers. Drugs 35 (Suppl. 3): 41–47, 1988
Collins JM, Unadkat JD. Clinical pharmacokinetics of zidovudine: an overview of current data. Clinical Pharmacokinetics 17: 1–9, 1989
Conte JE. Pharmacokinetics of cefpiramide in volunteers with normal or impaired renal function. Interscience Conference on Antimicrobial Agents and Chemotherapy, New York, Abstract no. 1073, 1987
Daneshmend TK, Warnock DW. Clinical pharmacokinetics of systemic antifungal drugs. Clinical Pharmacokinetics 8: 17–42, 1983
Darragh A, Gordon AJ, O’Byrne H, Hobbs DC, Casey E. Single dose and steady state pharmacokinetics of piroxicam in elderly vs young adults. European Journal of Clinical Pharmacology 28: 305–309, 1985
Davies BE, Boon R, Horton R, Reubi FC, Descoeudres CE. Pharmacokinetics of amoxycillin and clavulanic acid in haemodialysis patients following intravenous administration of augmentin. British Journal of Clinical Pharmacology 26: 385–390, 1988
Deray G, Diquet B, Martinez F, Vidal AM, Petitclerc T, et al. Pharmacokinetics of zidovudine in a patient on maintenance hemodialysis. New England Journal of Medicine 24: 1606–1607, 1989
Derbyshire N, Webb DB, Roberts D, Glew D, Williams JD. Pharmacokinetics of teicoplanin in subjects with varying degrees of renal function. Journal of Antimicrobial Chemotherapy 23: 869–876, 1989
Dubois D, Becart J, Leroy A, Humbert G, Fillastre JP. Cefpiramide pharmacokinetics in subjects with normal and impaired renal function. Interscience Conference on Antimicrobial Agents and Chemotherapy, New York. Abstract no. 1072, 1987
Fillastre JP. Quinolones and renal failure. Quinolones Bulletin 4: 1–7, 1988
Fillastre JP, Leroy A, Josse S, Moulin B. Étude pharmacocinétique du trométamol-fosfomycine chez les malades insuffisants rénaux. Pathologie Biologie 36: 728–730, 1988
Forgue ST, Guay DRP, Morgenthein EA, Matzke GR, Barbhaiya RH. Pharmacokinetics of cefepime in patients with renal dysfunction. International Conference on Antimicrobial Agents, Los Angeles. Abstract no. 129, 1988
Garg DC, Baltodano N, Jallad N. Pharmacokinetics of ranitidine in patients with renal failure. Journal of Clinical Pharmacology 26: 286–291, 1986
Geyer Y, Hoffler D, Koeppe P. Pharmaockinetics of aspoxicillin in subjects with normal and impaired renal function. Arzneimittel-Forschung 38: 1635–1639, 1988
Gibson TP, Demetriades JL, Bland JA. Imipenem/cilastatin pharmacokinetic profile in renal insufficiency. American Journal of Medicine 78 (Suppl. 6a): 54–61, 1985
Gladziwa U, Klotz U, Krishna DK, Schmitt H, Glockner WN, et al. Pharmacokinetics and dynamics of famotidine in patients with renal failure. British Journal of Clinical Pharmacology 26: 315–321, 1988
Guay DRP, Morgenthein EA, Matzke GR, Barbhaiva RH. Pharmacokinetics of cefepime in patients with renal dysfunction. International Conference on Antimicrobial Agents, Los Angeles. Abstract no. 129, 1986
Halstenson CE, Abraham PA, Opsahl JA, Chremos AN, Keane WF. et al. Disposition of famotidine in renal insufficiency. Journal of Clinical Pharmacology 27: 782–787, 1987
Halstenson CE, Guay DRP, Opsahl JA, Hirata CAI, Olanoff LS, et al. Disposition of cefmetazole in healthy volunteers and patients with impaired renal function. Antimicrobial Agents and Chemotherapy 34: 519–523, 1990a
Halstenson CE, Kelloway J, Shapiro B, Lin C, Krane W, et al. Disposition of isepamicin in patients with various degrees of renal function. International Conference on Antimicrobial Agents and Chemotherapy, Atlanta, 21–24 October. Abstract no. 763, p. 213, 1990c
Halstenson CE, Opsahl JA, Schwenke MH, Kovarik JM, Puri SK, et al. Disposition of roxithromycin in patients with normal and severely impaired renal function. Antimicrobial Agents and Chemotherapy 34: 385–389, 1990b
Hammer R, Bozler G, Zimmer A, Koss FW. Pharmacokinetics and metabolism of gastrozepin in man. Therapiewoche 27: 1575–1593, 1977
Hayden FG, Minocha A, Spyker DA, Hoffman HE. Comparative single dose pharmacokinetics of amantadine-hydrochloride and rimantadine-hydrochloride in young and elderly adults. Antimicrobial Agents and Chemotherapy 28: 216–221, 1985
Hobbs DC. Piroxicam pharmacokinetics: recent clinical results relating kinetics and plasma levels to age, sex and adverse effects. American Journal of Medicine 81 (Suppl. 5B): 22–28, 1986
Horber FF, Frey FJ, Descoeudres CE, Murray AT, Reubi FC. Differential effects of impaired renal function on the kinetics of clavulanic acid and amoxycillin. Antimicrobial Agents and Chemotherapy 29: 614–619, 1986
Horber FF, Guentert TW, Weidekamm E, Heizmann P, Descoeudres C, et al. Pharmacokinetics of tenoxicam in patients with impaired renal function. European Journal of Clinical Pharmacology 29: 697–701, 1986b
Howden CW, Payton CD, Meredith PM, Hughes DMA, Macdougall AI, et al. Antisecretory effect and oral pharmacokinetics of omeprazole in patients with chronic renal failure. European Journal of Clinical Pharmacology 28: 637–640, 1985
Humphrey MJ, Jevons-Tarbitt MH. Pharmacokinetic evaluation of UK-49-858, a metabolically stable triazole antifungal drug in animals and humans. Antimicrobial Agents and Chemotherapy 28: 648–653, 1985
Jensen JC. Clinical pharmacokinetics of terbinafine (Lamisil). Clinical and Experimental Dermatology 14: 110–113, 1989
Kindler J, Eckardt KU, Ehmer B, Jandeleit K, Kurtz A, et al. Single-dose pharmacokinetics of recombinant human erythropoietin in patients with various degrees of renal failure. Nephrology Dialysis and Transplantation 4: 345–349, 1989
Kirst HA, Sides GD. New directions for macrolide antibiotics: structural modifications and in vitro activity. Antimicrobial Agents and Chemotherapy 33: 1413–1418, 1989
Klecker RW, Collins JM, Yarchoan R, Thomas R, Jenkins JF, et al. Plasma and cerebrospinal fluid pharmacokinetics of 3′ azido-3′ deoxy-thymidine, a novel pyrimidine with potential application for the treatment of patients with AIDS and related disease. Clinical Pharmacology and Therapeutics 41: 407–412, 1987
Knadler MP, Bergstrom RF, Callaghan JT, Rubin A. Nizatidine, an H2-blocker: its metabolism and disposition in man. Drug Metabolism and Disposition 14: 175–182, 1986
Ko H, Novak E, Peters GR, Bothwell WM, Hosley JD, et al. Pharmacokinetics of single dose cefmetazole following intramuscular administration of cefmetazole sodium to healthy male volunteers. Antimicrobial Agents and Chemotherapy 33: 508–512, 1989
Koup JR, Dubach UC, Brandt R, Wyss R, Stoeckel K. Pharmacokinetics of cefetamet (Ro 15-8074) and cefetamet pivoxil (Ro 15-8075) after intravenous and oral doses in humans. Antimicrobial Agents and Chemotherapy 32: 573–579, 1988
Krakamp B, Tanswell P, Vogel H, Bozler G. Steady state intravenous pharmacokinetics of pirenzepine in patients with differing degrees of renal dysfunction. European Journal of Clinical Pharmacology 36: 75–78, 1989
Lagerstrom PO, Persson BA. Determination of omeprazole and metabolites in plasma and urine by liquid chromatography. Journal of Chromatography 309: 347–356, 1984
Lake KD, Fletcher CV, Love KR, Brown DC, Joyce LD, et al. Ganciclovir pharmacokinetics during renal impairment. Antimicrobial Agents and Chemotherapy 32: 1899–1900, 1988
Lameire N, Rosenkranz B, Maas L, Brockmeier D. A pharmacokinetic study of roxatidine acetate in chronic renal failure. Drugs 35 (Suppl. 3): 48–52, 1988
Lameire NH, Rosenkranz B, Malerczyck V. Influence of renal functional impairment and hemodialysis on the pharmacokinetics of cefpirome. 29th Interscience Conference on Antimicrobial Agents and Chemotherapy, Houston. Abstract 1218, 1989
Langtry HD, Campoli-Richards DM. Zidovudine: a review of its pharmacodynamic and pharmacokinetic properties and therapeutic efficacy. Drugs 37: 408–450, 1989
Laskin OL. Clinical pharmacokinetics of acyclovir. Clinical Pharmacokinetics 8: 187–201, 1983
Lassman HB, Ho I, Puri SK, Sabo R, Scheffer MR. The pharmacokinetics and pharmacodynamics of multiple doses of the new H2-receptor antagonist, roxatidine acetate, in healthy men. Drugs 35 (Suppl. 3): 53–64, 1988
Leroy A, Fillastre JP, Humbert G. Lomefloxacin pharmacokinetics in subjects with normal and impaired renal function. Antimicrobial Agents and Chemotherapy 34: 17–20, 1990
Lin CJH, Chrenos AN, Yeh KC, Antonello J, Hessey GA. Effects of age and chronic renal failure on the urinary excretion kinetics of famotidine in man. European Journal of Clinical Pharmacology 34: 41–46, 1988
Lin C, Kramer W, Kim H, Perentesis G, Affrime M, et al. Pharmacokinetics of a new oral antifungal agent SCH 39304 in man. 16th International Congress of Chemotherapy, Jerusalem, Israel, June 11–16. Abstract, p. 270, 1989
Macdougall IC, Roberts DE, Neubert P, Dharmasena Ad, Coles GA, et al. Pharmacokinetics of recombinant human erythropoietin in patients on continuous ambulatory peritoneal dialysis. Lancet 1: 425–427, 1989
Meffin PJ, Orgurinovich N, Brooks PM, Miners JO, Cochran M. Ranitidine disposition in patients with renal impairment. British Journal of Clinical Pharmacology 16: 731–734, 1983
Naesdal J, Andersson T, Bodemar G, Larsson R, Regårdh CG, et al. Pharmacokinetics of 14C-omeprazole in patients with impaired renal function. Clinical Pharmacology and Therapeutics 40: 344–351, 1986
O’Brien JJ, Campoli-Richards M. Acyclovir: an updated review of its antiviral activity, pharmacokinetic properties and therapeutic efficacy. Drugs 37: 233–309, 1989
Periti P, Mazzei T. Pharmacokinetics of roxithromycin in renal and hepatic failure and drug interactions. Journal of Antimicrobial Chemotherapy 20 (Suppl. B): 107–112, 1987
Puri SK, Lassman HB. Roxithromycin: a pharmacokinetic review of a macrolide. Journal of Antimicrobial Chemotherapy 20 (Suppl. B): 89–100, 1989
Regårdh CG, Gabrielson M, Hoffman KJ, Lofberg I, Skanberg I. Pharmacokinetics and metabolism of omeprazole in animals and man: an overview. Scandinavian Journal of Gastroenterology 108: 79–94, 1985
Reitberg DP, Marble DA, Schultz RW, Whall TJ, Schentag JJ. Pharmacokinetics of cefoperazone (2.0g) and sulbactam (1.0g) coadministered to subjects with normal renal function, patients with decreased renal function and patients with endstage renal disease on hemodialysis. Antimicrobial Agents and Chemotherapy 32: 503–509, 1988
Roberts C, Clinical pharmacokinetics of ranitidine. Clinical Pharmacokinetics 9: 211–217, 1983
Rogers JD, Meisinger MAP, Ferber G, Calandra GB, Demetriades JL, et al. Pharmacokinetics of imipenem and cilastatin in volunteers. Review of Infectious Diseases 7: S435–S446, 1885
Singlas E, Leroy A, Sultan E, Godin M, Moulin B, et al. Disposition of fleroxacin, a new trifluoroquinolone, and its metabolites: pharmacokinetics in renal failure and influence of hemodialysis. Clinical Pharmacokinetics 19: 67–79, 1990
Singlas E, Pioger JC, Taburet AM, Colaneri S, Fillastre JP. Comparative pharmacokinetics of zidovudine (AZT) and its metabolite (G.AZT) in healthy subjects and HIV seropositive patients. European Journal of Clinical Pharmacology 36: 639–640, 1989a
Singlas E, Pioger JC, Taburet AM, Colin JN, Fillastre JP. Zidovudine disposition in patients with severe renal impairment: influence of hemodialysis. Clinical Pharmacology and Therapeutics 46: 190–197, 1989b
Singlas E, Taburet AM. Pharmacocinétique clinique des nouvelles quinolones. In Les nouvelles quinolones: problèmes actuels de pathologie infectieuse et de réanimation, Vol. 1, pp. 83–102, Arnette, Paris, 1985
Sommadossi JP, Bevan R, Ling T, Lee F, Mastre B, et al. Clinical pharmacokinetics of ganciclovir in patients with normal and impaired renal function. Reviews of Infectious Diseases 10:(Suppl. 3): 507–514, 1988
Somogyi A, Gugler R. Clinical pharmacokinetics of cimetidine. Clinical Pharmacokinetics 8: 463–475, 1983
Stoeckel K, Tarn YK, Kneer J. Pharmacokinetics of oral cefetamet pivoxil (RO 15-8075) and intravenous cefetamet (RO 15-8074) in humans: a review. Current Medical Research and Opinion 11: 432–440, 1989
Takabatake T, Ohta M, Haekawa M, Yamamoto Y, Ishida Y, et al. Pharmacokinetics of famotidine, a new H2-receptor antagonist, in relation to renal function. European Journal of Clinical Pharmacology 28: 327–331, 1985
Takazawa T, Aoki N, Kouda Y. Pharmacokinetic study of sparfloxacin in patients with impaired renal function. Interscience Conference on Antimicrobial Agents and Chemotherapy, Atlanta, Abstract no. 1253, 1990
Thomas MG, Ellis-Pegler RB. Fluconazole treatment of Candida glabrata peritonitis. Journal of Antimicrobial Chemotherapy 24: 94–95, 1989
Toon S, Ross CE, Gokal R, Rowland M. Effects of impaired renal function on the oral pharmacokinetics of fluconazole. Abstracts of the 10th Meeting of the International Society for Human and Animal Mycology (ISHAM), Barcelona, Abstract no. 095, 1988
Van der Auwera P, Stolear JC, George B, Dudley MN. Pharmacokinetics of enoxacin and its oxo-metabolite following intravenous administration to patients with different degrees of renal impairment. Antimicrobial Agents and Chemotherapy 34: 1491–1497, 1990
Wills RJ, Farolino DA, Choma N, Keigher N. Rimantadine pharmacokinetics after single and multiple doses. Antimicrobial Agents and Chemotherapy 28: 216–221, 1987
Wise RD, Lister D, McNulty CAM, Griggs D, Andrews JM. The comparative pharmacokinetics and tissue penetration of four quinolones including intravenously administered enoxacin. Infection 14 (Suppl. 3): 196–202, 1986
Woolf AD, Rogers HJ, Bradbrook ID, Corless D. Pharmacokinetic observations on piroxicam in young adult, middle aged and elderly patients. British Journal of Clinical Pharmacology 16: 433–437, 1983
Wright N, Wise R. The elimination of sulbactam alone and combined with ampicillin in patients with renal dysfunction. Journal of Antimicrobial Chemotherapy 11: 583–587, 1983
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fillastre, JP., Singlas, E. Pharmacokinetics of Newer Drugs in Patients with Renal Impairment (Part I). Clin. Pharmacokinet. 20, 293–310 (1991). https://doi.org/10.2165/00003088-199120040-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-199120040-00004