Summary
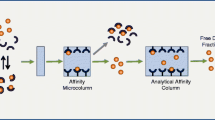
A rapid new ultrafiltmtion technique (EMIT® FreeLevel™ System I) for the routine measurement of unbound phenytoin concentrations was evaluated. The precision of this procedure was sufficient (between-days coefficient of variation, 11.7%). The results obtained by the ultrafiltration technique for the percentage of free phenytoin in samples from about 40 non-uraemic patients treated with this drug were in good agreement with those determined by ultracentrifugation and equilibrium dialysis (ultrafiltration vs ultracentrifugation, y = 0.94x + 0.60%; ultrafiltration vs equilibrium dialysis, y = 1.02x − 0.60%). The mean value of the results obtained by ultracentrifugation was significantly about 8% lower than that observed with equilibrium dialysis, apparently due to a sedimentation of free phenytoin during ultracentrifugation. With the methods used in our study, the mean values of percentage phenytoin bound to serum proteins obtained in samples from nonuraemic patients ranged from 91.8 to 92.8%.
All 3 methods yielded similar binding curves for phenytoin, with spiked human pool sera containing albumin concentrations between 19 and 45 g/L. A rise of the unbound phenytoin fraction was observed with increasing total concentrations of the drug and a decrease of the albumin concentration.
With samples from non-uraemic patients (n = 203), a rather good correlation was found between free and total phenytoin concentrations (r = 0.91). In a number of patients (n = 11), free phenytoin concentrations correlated better than total phenytoin concentrations with the clinical status. Patients with free phenytoin concentrations of ≥ 2.1 μg/ml and total phenytoin concentrations above the therapeutic range did not show signs of toxicity.
From the results of our study it is concluded that the EMIT FreeLevel ultrafiltration technique is very well-suited for a rapid and reliable separation of unbound phenytoin. In patients with altered protein binding or an unusual clinical response, free phenytoin determinations appear to be necessary for proper interpretation of total phenytoin levels and rational dosage adjustment.
Similar content being viewed by others
References
Booker, H.E. and Darcey, B.: Serum concentrations of free di-phenylhydantoin and their relationship to clinical intoxication. Epilepsia 14: 177–184 (1973).
Bowmer, J.C. and Lindup, W.E.: Binding of phenytoin, L-tryp-tophan and o-methy! red to albumin. Unexpected effect of albumin concentration on the binding of phenytoin and L-tryptophan. Biochemical Pharmacology 27: 937–942 (1978).
Büttner, H.: Die Messung der Eiweissbindung von Arzneimitteln mit Hilfe der Ultrazentrifuge. Antibiotica et Chemotherapia Fortschritte 12: 95–102 (1964).
Chanutin, A.S.; Ludewig, S. and Masket, A.V.: Studies on the calcium-protein-relationship with the aid of the ultracentri-fuge. I. Observations on calcium caseinate solutions. Journal of Biological Chemistry 143: 737–751 (1942).
Ehrnebo, M. and Odar-Cederlüf, J.: Binding of amobarbital, pentobarbital and diphenylhydantoin to blood cells and plasma proteins in healthy volunteers and uremic proteins. European Journal of Clinical Pharmacology 8: 445–453 (1975).
Feldmann, U.; Schneider, B.; Klinkers, H. and Haeckel, R.: A multivariate approach for the biométric comparison of analytical methods in clinical chemistry. Journal of Clinical Chemistry and Clinical Biochemistry 19: 121–137 (1981).
Greenblatt, D.J.; Sellers, E.M. and Koch-Weser, J.: Importance of protein binding for the interpretation of serum or plasma drug concentrations. Journal of Clinical Pharmacology 22: 259–263 (1982).
Gugler, R. and Azarnoff, D.L.: Drug protein binding and the ne-phrotic syndrome. Clinical Pharmacokinetics 1: 25–35 (1976).
Gugler, R.; Azarnoff, D.L. and Shoeman, D.W.: Diphenylhydantoin: Correlation between protein binding and albumin concentration. Klinische Wochenschrift 53: 445–446 (1975).
Haeckel, R. and Oellerich, M.: Evaluation of the 30°C-version of the ACA (Automatic Clinical Analyzer). Deutsche Gesellschaft für Klinische Chemie E.V. — Mitteilungen 11: 74–95 (1980).
Hoppel, C; Garle, M.; Rane, A. and Sjöqvist, F.: Plasma concentrations of 5- (4-hydroxyphenyl)-5-phenylhydantoin in phenytoin-treated patients. Clinical Pharmacology and Therapeutics 21: 294–300 (1977).
Koch-Weser, J. and Sellers, E.M.: Binding of drugs to serum albumin. New England Journal of Medicine 294: 311–316 (1976).
Kurz, H.; Trunk, H. and Weitz, B.: Evaluation of methods to determine protein-binding of drugs. Arzneimittel-Forschung 27: 1373–1380 (1977).
Levy, R.H. and Koch, K.M.: Drug interactions with valproic acid. Drugs 24: 543–556 (1982).
Lunde, P.K.M.; Rane, A.; Yaffe, S.J.; Lunde, L. and Sjöqvist, F.: Plasma protein binding of diphenylhydantoin in man. Clinical Pharmacology and Therapeutics 11: 846–855 (1970).
Oellerich, M.; Külpmann, W.R.; Haeckel, R. and Heyer, R.: Determination of phénobarbital and phenytoin in serum by a mechanized enzyme immunoassay (EMIT) in comparison with a gas-liquid Chromatographie method. Journal of Clinical Chemistry and Clinical Biochemistry 15: 353–358 (1977).
Porter, R.J. and Layzer, R.B.: Plasma albumin concentration and diphenylhydantoin binding in man. Archives of Neurology 32: 298–303 (1975).
Rowland, M.: Plasma protein binding and therapeutic drug monitoring. Therapeutic Drug Monitoring 2: 29–37 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oellerich, M., Müller-Vahl, H. The EMIT® FreeLevel™ Ultrafiltration Technique Compared with Equilibrium Dialysis and Ultracentrifugation to Determine Protein Binding of Phenytoin. Clin Pharmacokinet 9 (Suppl 1), 61–70 (1984). https://doi.org/10.2165/00003088-198400091-00008
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-198400091-00008