Abstract
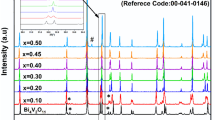
The preparation and characterization of Sb-doped Bi2UO6 solid solutions, in a limited composition range, is reported for the first time. The solid solutions were prepared by solid-state reactions of Bi2O3, Sb2O3 and U3O8 in the required stoichiometry. The reaction products were characterized by X-ray diffraction (XRD) and X-ray absorption spectroscopy (XAS) measurements at the Bi and U L3 edges. The XRD patterns indicate the precipitation of additional phases in the samples when Sb doping exceeds 4 at%. The chemical shifts of the Bi absorption edges in the samples, determined from the XANES spectra, show a systematic variation only up to 4 at% of Sb doping and support the results of XRD measurements. These observations are further supported by the local structure parameters obtained by analysis of the EXAFS spectra. The local structure of U is found to remain unchanged upon Sb doping indicating that Sb+3 ions replace Bi+3 during the doping of Bi2UO6 by Sb.
Similar content being viewed by others
References
S. Dash and Z. Singh, J. Nucl. Mater., 2010, 404, 9.
L. Smith, P. E. Raison, and R. J. M. Konings, J. Nucl. Mater., 2011, 413, 114.
A. Nakamura, Y. Doi, and Y. Hinatsu, J. Solid State Chem., 2011, 184, 531.
N. L. Misra, D. Lahiri, K. D. S. Mudher, L. Olivi, and S. M. Sharma, X-Ray Spectrom., 2008, 37, 215.
A. V. Soldatov, D. Lamoen, M. J. Konstantinovic, S. Van den Berghe, A. C. Scheinost, and M. Verwerft, J. Solid State Chem., 2007, 180, 53.
D. White and S. Ramdas, Ultramicroscopy, 1989, 31, 124.
H. Collette, V. Deremince-Mathieu, Z. Gabelica, J. B. Nagy, E. G. Derouane, and J. J. Verbist, J. Chem. Soc. Faraday Trans. 2, 1987, 83, 1263.
I. V. Dulera and R. K. Sinha, J. Nucl. Mater., 2008, 383, 183.
A. S. Koster, J. P. P. Renaud, and G. D. Rieck, Acta Crystallogr., Sect. B: Struct. Sci., 1975, 31, 127.
N. Bonanos, Mater. Res. Bull., 1989, 24, 1531.
O. Thery, R. N. Vannier, C. Dion, and F. Abraham, Solid State Ionics, 1996, 90, 105.
D. O. Charkin, D. N. Lebedev, S. Y. Stefanovich, and S. M. Kazakov, Solid State Sci., 2010, 12, 2079.
R. N. Vannier, O. Théry, C. Kinowski, M. Huvé, G. Van Tendeloo, E. Suard, and F. Abraham, J. Mater. Chem., 1999, 9, 435.
J. Rodriguez-Carjaval, Proceedings of the Satellite Meeting on Powder Diffraction, Toulouse, 1990, 127.
A. K. Sinha, A. Sagdeo, P. Gupta, A. Kumar, M. N. Singh, R. K. Gupta, S. R. Kane, and S. K. Deb, AIP Conf. Proc., 2011, 1349, 503.
D. Bhattacharyya, A. K. Poswal, S. N. Jha, Sangeeta, and S. C. Sabharwal, Nucl. Instrum. Methods phys. Res., Sect. A, 2009, 609, 286.
Neetika, A. Das, I. Dhiman, A. K. Nigam, A. K. Yadav, D. Bhattacharyya, and S. S. Meena, J. Appl. Phys., 2012, 112, 123913.
S. Basu, S. Varma, A. N. Shirsat, B. N. Wani, S. R. Bharadwaj, A. Chakrabarti, S. N. Jha, and D. Bhattacharyya, J. Appl. Phys., 2012, 111, 053532.
S. Basu, B. S. Naidu, M. Pandey, V. Sudarsan, S. N. Jha, D. Bhattacharyya, R. K. Vatsa, and R. J. Kshirsagar, Chem. Phys. Lett., 2012, 528, 21.
J. Mondal, A. Modak, A. Dutta, S. Basu, S. N. Jha, D. Bhattacharyya, and A. Bhaumik, Chem. Commun., 2012, 48, 8000.
S. K. Pandey, A. R. Chetal, and P. R. Sarode, J. Phys. Soc. Jpn., 1990, 59, 1848.
H. K. Hinge, S. K. Joshi, B. D. Shrivastava, J. Prasad, and K. Srivastava, Indian J. Pure Appl. Phys., 2011, 49, 168.
D. Joseph, S. Basu, S. N. Jha, and D. Bhattacharyya, Nucl. Instrum. Methods Phys. Res., Sect. B, 2012, 274, 126.
A. R. Chetal, P. Mahto, and P. R. Sarode, J. Phys. Chem. Solids, 1988, 49, 279.
C. Mande and M.Y. Apte, Bull. Mater. Sci., 1981, 3, 193.
J. P. Suchet, “Chemical Physics of Semiconductors”, 1965, Van Nostrand, London.
“X-Ray Absorption: Principles, Applications, Techniques of EXAFS, SEXAFS and XANES”, ed. D. C. Konigsberger and R. Prince, 1988, Wiley, New York.
B. Ravel and M. Newville, J. Synchrotron Radiat., 2005, 12, 537.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Misra, N.L., Yadav, A.K., Dhara, S. et al. Characterization of Sb-doped Bi2UO6 Solid Solutions by X-ray Diffraction and X-ray Absorption Spectroscopy. ANAL. SCI. 29, 579–584 (2013). https://doi.org/10.2116/analsci.29.579
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.29.579