Abstract
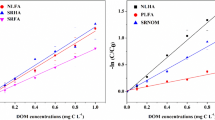
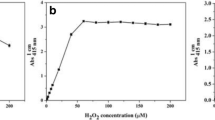
A non-enzymatic fluorescence method for the determination of hydrogen peroxide (H2O2) was investigated. This method is based on the hydroxylation reaction of terephthalate (TP) by hydroxyl radical formed from reaction between H2O2 and Fe(II), resulting in the formation of a strongly fluorescent 2-hydroxyterephthalate (HTP). Under optimized conditions, a 3 nM detection limit and 1.0% precision at 200 nM were obtained. This was sensitive enough to determine the concentrations of H2O2 in coastal marine environments. The slopes of the calibration curve in seawater were nearly the same as those in Milli-Q water, suggesting that the fluorescent intensity was not affected by coexisting sea salts. However, the presence of nitrite at more than 10 μM interfered with the formation of HTP. The developed method was successfully applied to determine the concentrations of H2O2 in Osaka Bay on the research vessel. The results obtained in Osaka Bay demonstrated that H2O2 was photochemically produced via the photolysis of dissolved organic matter supplied from the river with fresh water at the surface layer.
Similar content being viewed by others
References
C. Van Baalen and J. E. Marler, Nature, 1966, 211, 951.
R. J. Kieber and G. R. Helz, Anal. Chem., 1986, 58, 2312.
D. Abele-Oeschger, H. Tüg, and R. Röttgers, Limnol. Oceanogr., 1997, 42, 1406.
G. B. Avery Jr., W. J. Cooper, R. J. Kieber, and J. D. Willey, Mar. Chem., 2005, 97, 236.
K. Fujiwara, T. Ushiroda, K. Takeda, Y. Kumamoto, and H. Tsubota, Geochem. J., 1993, 27, 103.
B. H. Yocis, D. J. Kieber, and K. Mopper, Deep Sea Res., Part I, 2000, 47, 1077.
D. J. Kieber, G. W. Miller, P. J. Neale, and K. Mopper, Environ. Sci.: Processes Impacts, 2014, 16, 777.
G. W. Miller, C. A. Morgan, D. J. Kieber, D. W. King, J. A. Snow, B. G. Heikes, K. Mopper, and J. J. Kiddle, Mar. Chem., 2005, 97, 4.
W. J. Cooper, J. K. Moegling, R. J. Kieber, and J. J. Kiddle, Mar. Chem., 2000, 70, 191.
E. F. Olasehinde, S. Makino, H. Kondo, K. Takeda, and H. Sakugawa, Anal. Chim. Acta, 2008, 627, 270.
H. Sakugawa and I. R. Kaplan, Atmos. Environ., 1993, 27A, 1509.
H. Sakugawa, T. Yamashita, H. Kawai, N. Masuda, N. Hashimoto, S. Makino, N. Nakatani, and K. Takeda, Cikyukagaku (Geochemistry, in Japanese), 2006, 40, 47.
T. Yamamoto, J. Lewis, J. Wataha, D. Dickinson, B. Singh, W. B. Bollag, E. Ueta, T. Osaki, M. Athar, G. Schuster, and S. Hsu, J. Pharmacol. Exp. Ther., 2004, 308, 317.
P. C. Chai, L. H. Long, and B. Halliwel, Biochem. Biophys. Res. Commun., 2003, 304, 650.
J. L. Manzoori, M. Amjadi, and M. Orooji, Anal. Sci., 2006, 22, 1201.
J. Liu, L. Lu, A. Li, J. Tang, S. Wang, S. Xu, and L. Wang, Biosens. Bioelectron., 2015, 68, 204.
A. K. Dutta, S. Das, P. K. Samanta, S. Roy, B. Adhikary, and P. Biswas, Electrochim. Acta, 2014, 144, 282.
R. J. Kieber, W. J. Cooper, J. D. Willey, and G. B. Avery Jr., J. Atmos. Chem., 2001, 39, 1.
B. Palenik, O. C. Zafiriou, and F. M. M. Morel, Limnol. Oceanogr., 1987, 32, 1365.
R. G. Petasne and R. G. Zika, Mar. Chem., 1997, 56, 215.
M. Tarvin, B. McCord, K. Mount, K. Sherlach, and M. L. Miller, J. Chromatogr. A, 2010, 1217, 7564.
D. Price, R. F. C. Mantoura, and P. J. Worsfold, Anal. Chim. Acta, 1998, 377, 145.
M. J. Hopwood, I. Rapp, C. Schlosser, and E. P. Achterberg, Sci. Rep., 2017, 7, Article number: 43436.
J. Liu, S. M. Steinberg, and B. J. Johnson, Chemosphere, 2003, 52, 815.
J. H. Lee, I. N. Tang, J. B. Weinstein-Lloyd, and E. B. Halper, Environ. Sci. Technol., 1994, 28, 1180.
W. Luo, M. E. Abbas, L. Zhu, K. Deng, and H. Tang, Anal. Chim. Acta, 2008, 629, 1.
M. E. Abbas, W. Luo, L. Zhu, J. Zou. and H. Tang, Food Chem., 2010, 120, 327.
J. C. Barreto, G. S. Smith, N. H. P. Strobel, P. A. McQuillin, and T. A. Miller, Life Sci., 1995, 56, 89.
X. Fang, G. Mark, and C. von Sonntag, Ultrason. Sonochem., 1996, 3, 57.
L. Linxiang, Y. Abe, Y. Nagasawa, R. Kudo, N. Usui, K. Imai, T. Mashino, M. Mochizuki, and N. Miyata, Biomed. Chromatogr., 2004, 18, 470.
M. Saran and K. H. Summer, Free Radical Res., 1999, 31, 429.
T. Charbouillot, M. Brigante, G. Mailhot, P. R. Maddigapu, C. Minero, and D. Vione, J. Photochem. Photobiol., A, 2011, 222. 70.
S. E. Page, W. A. Arnold, and K. McNeill, J. Environ. Monit., 2010, 12, 1658.
W. L. Miller and D. R. Kester, Anal. Chem., 1988, 60, 2711.
Z. Stuglik and Z. PawełZagórski, Radiat. Phys. Chem., 1981, 17, 229.
L. L. Land and R. J. Hanrahan, Radiat. Phys. Chem., 2004, 69, 401.
A. A. Burbano, D. D. Dionysiou, M. T. Suidan, and T. L. Richardson, Water Res., 2005, 39, 107.
H. C. Brown, D. H. McDaniel, and O. Häfliger, “Determination of Organic Structures by Physical Methods”, ed. E. A. Baude and F. C. Nachod, 1955, Vol. 1, Academic Press, New York, 567.
G. G. Jayson, B. J. Parsons, and A. J. Swallow, J. Chem. Soc., Faraday Trans. 1, 1973, 69, 1597.
A. E. Grigor'ev, I. E. Makarov, and A. K. Pikaev, High Energy Chem., 1987, 21, 99.
D. Zehavi and J. Rabani, J. Phys. Chem., 1972, 76, 312.
K. Takeda, K. Fujisawa, H. Nojima, R. Kato, R. Ueki, and H. Sakugawa, J. Photochem. Photobiol., A, 2017, 340, 8.
T. Lpgager and K. Sehested, J. Phys. Chem., 1993, 97, 6664.
A. Treinin and E. Hayon, J. Am. Chem. Soc., 1970, 92, 5821.
E. F. Olasehinde, K. Takeda, and H. Sakugawa, Environ. Sci. Technol., 2010, 44, 8403.
K. Takeda, K. Yamane, Y. Horioka, and K. Ito, Aqua. Geochem., 2017, 23, 315.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takeda, K., Nojima, H., Kuwahara, K. et al. Nanomolar Determination of Hydrogen Peroxide in Coastal Seawater Based on the Fenton Reaction with Terephthalate. ANAL. SCI. 34, 459–464 (2018). https://doi.org/10.2116/analsci.17P536
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2116/analsci.17P536