Abstract
Objectives
In many jurisdictions, routine medical care was reduced in response to the COVID-19 pandemic. The objective of this study was to determine whether the frequency of on-time routine childhood vaccinations among children age 0–2 years was lower following the COVID-19 declaration of emergency in Ontario, Canada, on March 17, 2020, compared to prior to the pandemic.
Methods
We conducted a longitudinal cohort study of healthy children aged 0–2 years participating in the TARGet Kids! primary care research network in Toronto, Canada. A logistic mixed effects regression model was used to determine odds ratios (ORs) for delayed vaccination (> 30 days vs. ≤ 30 days from the recommended date) before and after the COVID-19 declaration of emergency, adjusted for confounding variables. A Cox proportional hazards model was used to explore the relationship between the declaration of emergency and time to vaccination.
Results
Among 1277 children, the proportion of on-time vaccinations was 81.8% prior to the COVID-19 declaration of emergency and 62.1% after (p < 0.001). The odds of delayed vaccination increased (odds ratio = 3.77, 95% CI: 2.86–4.96), and the hazard of administration of recommended vaccinations decreased after the declaration of emergency (hazard ratio = 0.75, 95% CI: 0.60–0.92). The median vaccination delay time was 5 days (95% CI: 4–5 days) prior to the declaration of emergency and 17 days (95% CI: 12–22 days) after.
Conclusion
The frequency of on-time routine childhood vaccinations was lower during the first wave of the COVID-19 pandemic. Sustained delays in routine vaccinations may lead to an increase in rates of vaccine-preventable diseases.
Résumé
Objectifs
Dans plusieurs juridictions, les soins médicaux systématiques étaient réduits à cause de la pandémie de COVID-19. L’objectif de cette étude était de déterminer si la fréquence de donner les vaccinations systématiques aux enfants de l’âge de 0 à 2 ans était réduite en conséquence de la déclaration d’urgence de COVID-19 en Ontario, Canada dès le 17 mars 2020, comparer avec la fréquence avant la pandémie.
Méthodes
Nous avons mené une étude de cohorte longitudinale des enfants en bonne santé âgés de 0 à 2 ans qui participent dans le réseau de recherche en soins primaires TARGet Kids! à Toronto, Canada. Un modèle de régression logistique à effets mixtes était utilisé pour déterminer le rapport de cotes (RC) pour les vaccinations retardées (> 30 jours c. ≤ 30 jours de la date recommandée) et était équilibré pour les variables confondantes. Le modèle à risques proportionnels de Cox était utilisé pour examiner le lien entre la déclaration d’urgence et le temps jusqu’à la vaccination.
Résultats
Parmi 1 277 enfants, la proportion de vaccination à l’heure était 81,8 % avant la déclaration d’urgence de COVID-19 et 62,1 % après (p < 0,001). La possibilité de vaccination retardée était augmentée (RC = 3,77; IC95% : 2,86–4,96), et le taux d’administration recommandé pour les vaccinations était réduit après la déclaration d’urgence (ratio de hasard = 0,75; IC95% : 0,60–0,92). Le médian temps de retard pour les vaccinations était 5 jours (IC95% : 4–5 jours) avant la déclaration d’urgence et 17 jours (IC95% : 12–22 jours) après.
Conclusion
La fréquence de vaccinations systématiques aux enfants à l’heure était inférieure pendant la première vague de la pandémie COVID-19. Des retards soutenus pour recevoir les vaccinations systématiques peuvent entrainer une augmentation des taux de maladies évitables par la vaccination.
Similar content being viewed by others
Introduction
Routine childhood vaccinations are among the most important public health interventions for preventing vaccine-preventable diseases (VPD). The National Advisory Committee on Immunization (NACI) in Canada recommends that 13 vaccinations be administered in the first 24 months of life, including the diphtheria, tetanus, pertussis, polio, Haemophilus influenzae type b vaccine (DTaP-IPV-Hib), pneumococcal conjugate vaccine (Pneu-C-13), rotavirus (Rot-5) vaccine, meningococcal conjugate (Men-C–C) vaccine, measles, mumps, and rubella (MMR) vaccine, and varicella vaccine (Canada, 2017). Timely vaccination occurs when vaccines are administered within 30 days of the recommended date and are considered delayed when administered more than 30 days after the recommended date (Kiely et al., 2018; O’Donnell et al., 2017; Sood et al., 2015). According to the 2017 National Immunization Coverage Survey, the proportion of children in Ontario vaccinated against all doses of recommended vaccinations range from 72.9% to 91.0% (Canada, 2020).
On March 11, 2020, the World Health Organization (WHO) declared coronavirus disease 2019 (COVID-19) to be a pandemic. This was followed by a declaration of emergency in Ontario, Canada, on March 17, 2020 (A Timeline of the Novel Coronavirus in Ontario, n.d.). Non-essential medical care and elective services were reduced to minimal levels or ceased completely (Ontario Health, 2020). Although public health and government institutions communicated that routine vaccinations should not be delayed during the pandemic, the scale-back of non-essential medical services due to factors such as inadequate supply of personal protective equipment (PPE) may have contributed to a decrease in vaccine delivery (Jenco, 2020; WHO/UNICEF Joint Statement – Maintaining Routine Immunization Services Vital during the COVID-19 Pandemic, 2020).
The primary objective of this study was to determine whether the frequency of on-time routine childhood vaccinations among children age 0–2 years was lower following the COVID-19 declaration of emergency in Ontario, Canada, compared to prior to the pandemic. A secondary objective was to explore the relationship between the COVID-19 declaration of emergency and vaccination timeliness.
Methods
Subjects and design
A longitudinal cohort study involving healthy children aged 0–2 years (as of May 31, 2020) receiving primary healthcare through the TARGet Kids! primary care research network in Toronto, Canada, was conducted between November 1, 2018 and May 31, 2020, a period that included the first wave of the COVID-19 pandemic. Vaccinations for children are primarily administered during primary care physician visits in Ontario. TARGet Kids! is a collaboration between researchers and primary care providers affiliated with the University of Toronto, Unity Health Toronto, and The Hospital for Sick Children in Toronto, Canada (Carsley et al., 2015). Children were excluded from the TARGet Kids! cohort if they had health conditions affecting growth, all chronic medical conditions except asthma, or severe developmental delay.
Exposure, outcome, and confounding variables
The expected date of each recommended vaccination according to Ontario’s routine immunization schedule was determined based on the birth date of each child (Supplementary Table 1). These vaccination dates were converted from months to days, where 1 month is equivalent to 30.5 days. Time to vaccination was calculated as the number of days after the recommended date that the vaccine was administered according to the child’s electronic medical records (EMRs). Routine vaccinations among children in Ontario are administered by primary care physicians; thus, we expect EMRs to be reliable records of childhood vaccinations. On-time vaccination was defined as vaccinations received ≤ 30 days after the recommended date, including those received before the recommended date. Delayed vaccination was defined as vaccinations received > 30 days after the recommended date or that were incomplete. The median time to vaccination was calculated as the median number of days between the recommended vaccination date and the actual vaccination date.
The primary exposure was the Ontario COVID-19 declaration of emergency on March 17, 2020. This was a binary breakpoint, where vaccinations received on or after the declaration of emergency were considered to be provided during the pandemic while vaccinations provided prior to this date were considered to be provided before the pandemic.
The primary outcome was a binary variable indicating whether the vaccination was on-time (≤ 30 days after the recommended date) or delayed (> 30 days after the recommended date). The secondary outcome was time to vaccination in days after the recommended date of vaccination.
Potential confounding variables hypothesized a priori included child sex, maternal ethnicity, maternal education, single-parent household, birth order, vaccine type, primary care clinic, parent-reported health conditions, self-reported annual household income, and the distance between the patient’s residence and their primary care clinic in kilometres. Age of recommended vaccination was examined as a potential effect measure modifying variable as a post hoc analysis based on reviewer feedback. These variables were collected using a parent-completed questionnaire based on the Canadian Community Health Survey by a trained research assistant during the well-child visit (Carsley et al., 2015). Participant name, birth date, and primary care clinic were used to link survey and EMR data.
Statistical analysis
Descriptive statistics were performed on all variables. A Kaplan–Meier survival curve was used to visually compare the time to vaccination before and after the COVID-19 declaration of emergency. The proportion of on-time vaccinations was calculated before and after the declaration of emergency, where the numerator is the number of on-time vaccinations and the denominator is the total number of recommended vaccinations. The proportion of children with on-time vaccinations before and after the declaration of emergency was also calculated for each age group, where the numerator is the number of children with on-time vaccination and the denominator is the total number of children with vaccinations recommended at this age. These proportions were compared using a normal approximation test of proportions. For the primary analysis, a logistic generalized mixed effects regression model was used to estimate the odds of delayed vaccination before and after the Ontario COVID-19 declaration of emergency. Each recommended vaccination during the study period was assessed as a data point. Random effects were used to account for multiple vaccinations in the same subject. A likelihood ratio test was used to determine whether age of recommended vaccination should be included in the model (p < 0.3). The model was adjusted for the clinically relevant covariates described above.
For the secondary analysis, a Cox proportional hazards model was constructed to examine the association between the COVID-19 declaration of emergency and time to vaccination. Each recommended vaccination during the study period was assessed as a data point. Robust standard errors were used to account for multiple vaccinations in the same subject (Harrell Jr, 2020). This model was adjusted for the same variables as the logistic mixed effects regression model, and a likelihood ratio test was used to determine whether age of recommended vaccination should be included as an effect modifier (p < 0.3). Schoenfeld residuals were used to test the proportional hazards assumption indicating no concern. Effect sizes were reported as hazard ratios (HR) and their 95% confidence intervals. Longer time to vaccination and a hazard ratio less than one indicate that vaccinations occurred at a lower hazard rate following the declaration of emergency.
To handle missing data, all variables were considered missing at random, as missingness can be accounted for by other variables in the model with complete information. An imputed dataset was created using multivariate imputation by chained equations (MICE) (van Buuren & Groothuis-Oudshoorn, 2011).
This study was approved by the Research Ethics Boards at The Hospital for Sick Children, Unity Health Toronto, and the Children’s Hospital of Eastern Ontario. Consent was obtained from parents of all participating children in TARGet Kids!. All statistical analyses were performed using R version 4.0.3 (R: The R Project for Statistical Computing, n.d.).
Results
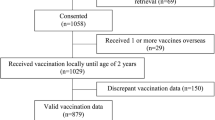
A total of 1277 children 0–2 years of age were included in the analysis (Fig. 1). During the period of observation, 6867 vaccinations were recommended for this population. 50.7% of children were female and 38.4% of children resided in two-parent households. The average distance from the patient’s home to their primary care clinic was 7.1 km. Children with at least one on-time vaccination vs. at least one delayed vaccination seemed clinically similar (Tables 1 and 2). 8.7% of vaccinations were incomplete in the EMR (n = 605/6937).
Proportion of on-time vaccinations
For the primary analysis, the proportion of on-time vaccinations (≤ 30 days) was 81.8% (n = 5177/6332) prior to the COVID-19 declaration of emergency and 62.1% (n = 376/605) after (p < 0.001). The proportion of children aged 6 months with on-time vaccinations decreased from 64.5% (n = 529/820) prior to the COVID-19 declaration of emergency to 43.8% (n = 21/48) after (p = 0.04). The proportion of children with on-time vaccinations at age 15 months decreased from 60.8% (n = 417/686) to 39.7% (n = 48/121) (p < 0.001). At 18 months, this decreased from 59.4% (n = 293/493) to 37.6% (n = 50/133) (p < 0.001).
In the unadjusted and adjusted logistic regression models, the odds of delayed vaccination (> 30 days) were higher after the declaration of emergency than before (unadjusted OR = 4.44, 95% CI: 4.44–4.44, p < 0.001 and adjusted OR = 3.77, 95% CI: 2.86–4.96, p < 0.001) (Supplementary Table 2). Age of recommended vaccination was included in the model as an effect modifier (p = 0.005). The age-stratified model indicated that the odds of delayed vaccination were higher after the declaration of emergency among vaccinations recommended at 6 months, 12 months, 15 months, and 18 months (adjusted OR = 5.36, 95% CI: 2.29–13.00; adjusted OR = 1.98, 95% CI: 1.20–3.00; adjusted OR = 6.09, 95% CI: 3.57–10.00; adjusted OR = 5.26, 95% CI: 3.14–9.00, respectively). The odds of delayed vaccination was not statistically different before and after the declaration of emergency for vaccinations recommended at 2 months and 4 months of age (Supplementary Table 4a).
Vaccination timeliness
For the secondary analysis, the median time to vaccination was 5 days (95% CI: 4–5) prior to the COVID-19 declaration of emergency and 17 days (95% CI: 12–22) after (Fig. 2). Unadjusted and adjusted Cox proportional hazards models revealed that the hazard rate at which vaccinations occurred was lower following the declaration of emergency (unadjusted HR = 0.60, 95% CI: 0.54–0.67, p < 0.001 and adjusted HR = 0.75, 95% CI: 0.60–0.92, p = 0.008) (Supplementary Table 3). On a given day following the declaration of emergency, each child was, on average, 25% less likely to be vaccinated than prior to the declaration of emergency. Age of recommended vaccination was included in the model as an effect modifier (p = 0.10). When stratified by age of recommended vaccination, the hazard rate at which vaccinations occurred was lower following the declaration of emergency for vaccinations recommended at 15 months and 18 months (adjusted HR = 0.60, 95% CI: 0.48–0.76; adjusted HR = 0.79, 95% CI: 0.64–0.98, respectively). The hazard rate at which vaccinations occurred was not statistically different before and after the declaration of emergency for vaccinations recommended at ages younger than 15 months (Supplementary Table 4b).
Discussion
In this study involving 1277 children aged 0–2 years who were participating in a primary care research network in Toronto, Canada, the frequency of on-time vaccinations was lower during the first wave of the COVID-19 pandemic. The odds of vaccination delay (> 30 days) were nearly fourfold higher in the 3 months following the COVID-19 declaration of emergency than before, particularly among older children (i.e., 6, 12, 15, or 18 months). The median time to vaccination increased from 5 days (95% CI: 4–5) before to 17 days (95% CI: 12–22) after the declaration of emergency.
Childhood vaccination has been a major contributor to reductions in morbidity and mortality from vaccine-preventable illness over the past century. Reductions in routine childhood vaccinations during the COVID-19 pandemic may increase the risk of outbreaks of vaccine-preventable diseases including measles and varicella zoster virus (Lee et al., 2004; Measles Outbreaks Reported in Five Districts Including in Kathmandu and Lalitpur in Last One Month, n.d.; Suk et al., 2016). Since 2016, the incidence of measles has been increasing in both low- and high-income countries, owing to factors including vaccine hesitancy (Mortier et al., 2019; Ortega-Sanchez et al., 2014; Roberts, 2020; Wadman, 2019). Reduction in vaccination during the COVID-19 pandemic may further increase the risk of measles outbreaks, as small decreases in measles vaccine coverage can lead to increased transmission.
Parental concerns about COVID-19, local lockdown measures, suspended supply chains, and reduced clinic staff capacity and PPE to administer vaccines may be contributing factors to delayed or missed vaccinations among children (Bell et al., 2020; Dinleyici et al., 2020). Strategies to ameliorate delayed vaccines among children due to COVID-19 are needed. Public health and governmental institutions must continue to stress the necessity of timely vaccinations. Clinics must continue training their staff and maintaining adequate supplies of PPE to ensure safe delivery of vaccines. Reminders and education about the importance of vaccination for both parents and healthcare providers, and electronic immunization records have been shown to be effective in encouraging families and healthcare providers to complete vaccinations on schedule (Chandir et al., 2020; Williams et al., 2011). Other strategies such as informing parents about the ways clinics are minimizing COVID-19 transmission risk and school vaccination programs or special clinics in community spaces to safely administer vaccines may help children catch up missed vaccinations (Chanchlani et al., 2020). In addition, at-home visits and drive-through vaccination clinics have emerged as creative solutions which rely heavily on healthcare providers willing to deliver vaccines in unconventional settings (Hoffman, 2020). Support for strategies such as these during the COVID-19 pandemic and afterwards is needed to help children catch up on delayed vaccinations and prevent the spread of vaccine-preventable diseases.
Strengths of this study include data from a large, ongoing, prospective cohort study involving healthy young children receiving primary healthcare. Detailed data allowed for adjustment of numerous clinically relevant potentially confounding factors, including self-reported annual income and ethnicity. Access to participants’ electronic medical records to verify immunization status and timeliness was also a strength of this study. This study also supports the findings by Langdon-Embry et al. who identified lower childhood vaccination in New York City during a similar time period (Langdon-Embry et al., 2020). The increased odds of delayed vaccination among vaccinations recommended later in childhood also reflects findings suggesting that vaccination delays are more prevalent for later vaccination visits (Kiely et al., 2018).
Limitations of this study include a relatively short observation window of 3 months during the first wave of the COVID-19 pandemic. A longer observation window with data collected later in the COVID-19 pandemic would help address whether delayed vaccinations are eventually caught up, as other regions have seen a rebound in vaccinations several months into the pandemic (Langdon-Embry et al., 2020). Clinics may have improved capacity for routine vaccinations in subsequent months as PPE supply became adequate, which may limit generalizability of this study to subsequent waves or inter-wave periods of the pandemic. Different measures taken across the various primary care practices involved in this study may have contributed to heterogeneity. Children from clinics with less capacity to support routine vaccinations may have been transferred to other clinics of greater capacity, and not captured through the available EMRs. In addition, this study was conducted using a primary care research network in a large urban centre and thus, results may not be generalizable to rural settings. The study also did not account for clustering of vaccinations among children residing in the same household. Finally, parental perspectives on vaccines were not measured in this study and may have contributed to our understanding of delayed vaccinations.
Conclusion
The frequency of on-time routine childhood vaccinations was lower during the first wave of the COVID-19 pandemic in this population of healthy children receiving primary healthcare. Delay in childhood vaccinations may lead to an increased risk of vaccine-preventable diseases. Primary healthcare and public health strategies to reduce delays in routine childhood immunizations during the COVID-19 pandemic are needed.
Contributions to knowledge
What does this study add to existing knowledge?
-
Vaccines are among the most important public health interventions and have contributed immensely to the decreased rates of infectious diseases globally. However, the COVID-19 pandemic has led to scale-backs of non-essential medical services, and a decrease in the use of elective services has been observed.
-
Although routine childhood vaccinations are considered essential, clinics may not have the capacity or resources to provide these services during a strenuous time such as the pandemic.
-
This study helps understand how practices such as routine vaccination are affected during a global health crisis, and potential implications for vaccine-preventable disease rates in the future.
What are the key implications for public health interventions, practice, or policy?
-
Among the children from Toronto, Ontario included in the study, the proportion of on-time vaccination decreased, the odds of delayed vaccination increased, and the rate of vaccination decreased after the COVID-19 declaration of emergency compared to before this date.
-
This observed delay in vaccinations calls for a system to ensure that children catch up on their vaccinations. This could be through increasing education to parents and healthcare providers, ensuring that clinics are supplied with adequate personal protective equipment, and creating a central database that keeps up-to-date vaccinations for children across the province.
Data availability
Data used in this analysis will be made available to editors upon request either before or after publication for checking.
Code availability
Code used in this analysis will be made available to editors upon request either before or after publication for checking.
References
A timeline of the novel coronavirus in Ontario. (n.d.). Global News. Retrieved August 11, 2020, from https://globalnews.ca/news/6859636/ontario-coronavirus-timeline/
Bell, S. L., Clarke, R., Paterson, P., & Mounier-Jack, S. (2020). Parents’ and guardians’ views and experiences of accessing routine childhood vaccinations during the coronavirus (COVID-19) pandemic: A mixed methods study in England [Preprint]. Primary Care Research. https://doi.org/10.1101/2020.09.04.20186569
Carsley, S., Borkhoff, C. M., Maguire, J. L., Birken, C. S., Khovratovich, M., McCrindle, B., Macarthur, C., & Parkin, P. C. (2015). Cohort profile: The Applied Research Group for Kids (TARGet Kids!). International Journal of Epidemiology, 44(3), 776–788. https://doi.org/10.1093/ije/dyu123
Chanchlani, N., Buchanan, F., & Gill, P. J. (2020). Addressing the indirect effects of COVID-19 on the health of children and young people. CMAJ, 192(32), E921–E927. https://doi.org/10.1503/cmaj.201008
Chandir, S., Siddiqi, D. A., Mehmood, M., Setayesh, H., Siddique, M., Mirza, A., Soundardjee, R., Dharma, V. K., Shah, M. T., Abdullah, S., Akhter, M. A., Ali Khan, A., & Khan, A. J. (2020). Impact of COVID-19 pandemic response on uptake of routine immunizations in Sindh, Pakistan: An analysis of provincial electronic immunization registry data. Vaccine, 38(45), 7146–7155. https://doi.org/10.1016/j.vaccine.2020.08.019
Dinleyici, E. C., Borrow, R., Safadi, M. A. P., van Damme, P., & Munoz, F. M. (2020). Vaccines and routine immunization strategies during the COVID-19 pandemic. Human Vaccines & Immunotherapeutics, 1–8. https://doi.org/10.1080/21645515.2020.1804776
Harrell Jr, F. E. (2020). rms: Regression Modeling Strategies [R package version 6.1–0]. https://CRAN.R-project.org/package=rms
Hoffman, J. (2020, April 23). Vaccine rates drop dangerously as parents avoid doctor’s visits. The New York Times. https://www.nytimes.com/2020/04/23/health/coronavirus-measles-vaccines.html
Jenco, M. (2020). AAP urges vaccination as rates drop due to COVID-19. AAP News. https://www.aappublications.org/news/2020/05/08/covid19vaccinations050820
Kiely, M., Boulianne, N., Talbot, D., Ouakki, M., Guay, M., Landry, M., Sauvageau, C., & De Serres, G. (2018). Impact of vaccine delays at the 2, 4, 6 and 12 month visits on incomplete vaccination status by 24 months of age in Quebec, Canada. BMC Public Health, 18(1), 1364. https://doi.org/10.1186/s12889-018-6235-6
Langdon-Embry, M., Papadouka, V., Cheng, I., Almashhadani, M., Ternier, A., & Zucker, J. R. (2020). Notes from the field: Rebound in routine childhood vaccine administration following decline during the COVID-19 pandemic — New York City, March 1–June 27, 2020. Morbidity and Mortality Weekly Report, 69(30), 999–1001. https://doi.org/10.15585/mmwr.mm6930a3
Lee, B. R., Feaver, S. L., Miller, C. A., Hedberg, C. W., & Ehresmann, K. R. (2004). An elementary school outbreak of varicella attributed to vaccine failure: Policy implications. The Journal of Infectious Diseases, 190(3), 477–483. https://doi.org/10.1086/422041
Measles outbreaks reported in five districts including in Kathmandu and Lalitpur in last one month. (n.d.). Retrieved March 11, 2021, from https://kathmandupost.com/national/2020/04/29/measles-outbreaks-reported-in-five-districts-including-in-kathmandu-and-lalitpur-in-last-one-month
Mortier, C., Mailhe, M., Amrane, S., & Lagier, J.-C. (2019). Measles from Madagascar. Travel Medicine and Infectious Disease, 29, 58–59. https://doi.org/10.1016/j.tmaid.2019.04.003
O’Donnell, S., Dubé, E., Tapiero, B., Gagneur, A., Doll, M. K., & Quach, C. (2017). Determinants of under-immunization and cumulative time spent under-immunized in a Quebec cohort. Vaccine, 35(43), 5924–5931. https://doi.org/10.1016/j.vaccine.2017.08.072
Ontario Health. (2020). COVID-19 Directive #1 for Health Care Providers and Health Care Entities. https://www.cmo.on.ca/wp-content/uploads/2020/04/cmoh-directive-1-2020-03-12.pdf
Ortega-Sanchez, I. R., Vijayaraghavan, M., Barskey, A. E., & Wallace, G. S. (2014). The economic burden of sixteen measles outbreaks on United States public health departments in 2011. Vaccine, 32(11), 1311–1317. https://doi.org/10.1016/j.vaccine.2013.10.012
Public Health Agency of Canada. (2017, October 12). National Advisory Committee on Immunization (NACI): Statements and publications [Transparency - other; navigation page]. Aem. https://www.canada.ca/en/public-health/services/immunization/national-advisory-committee-on-immunization-naci.html
Public Health Agency of Canada. (2020, January 29). Vaccine coverage in Canadian children: Results from the 2017 Childhood National Immunization Coverage Survey (cNICS) [Research]. Aem. https://www.canada.ca/en/public-health/services/publications/healthy-living/2017-vaccine-uptake-canadian-children-survey.html#_Children_aged_two
R: The R Project for Statistical Computing. (n.d.). Retrieved March 17, 2021, from https://www.r-project.org/
Roberts, L. (2020). Why measles deaths are surging—And coronavirus could make it worse. Nature, 580(7804), 446–447. https://doi.org/10.1038/d41586-020-01011-6
Sood, R., Sood, A., Bharti, O., Ramachandran, V., & Phull, A. (2015). High immunization coverage but delayed immunization reflects gaps in Health Management Information System (HMIS) in District Kangra, Himachal Pradesh, India—An immunization evaluation. World Journal of Vaccines, 5, 69–78. https://doi.org/10.4236/wjv.2015.52009
Suk, J. E., Jimenez, A. P., Kourouma, M., Derrough, T., Baldé, M., Honomou, P., Kolie, N., Mamadi, O., Tamba, K., Lamah, K., Loua, A., Mollet, T., Lamah, M., Camara, A. N., & Prikazsky, V. (2016). Post-Ebola measles outbreak in Lola, Guinea, January–June 20151. Emerging Infectious Diseases, 22(6), 1106–1108. https://doi.org/10.3201/eid2206.151652
van Buuren, S., & Groothuis-Oudshoorn, K. (2011). mice: Multivariate Imputation by Chained Equations in R. 45(3), 1–67.
Wadman, M. (2019). Measles epidemic in Ukraine drove troubling European year. Science, 363(6428), 677–678. https://doi.org/10.1126/science.363.6428.677
WHO/UNICEF joint statement – Maintaining routine immunization services vital during the COVID-19 pandemic. (2020, May 20). World Health Organization. http://www.euro.who.int/en/media-centre/sections/statements/2020/whounicef-joint-statement-maintaining-routine-immunization-services-vital-during-the-covid-19-pandemic
Williams, N., Woodward, H., Majeed, A., & Saxena, S. (2011). Primary care strategies to improve childhood immunisation uptake in developed countries: Systematic review. JRSM Short Reports, 2(10), 1–21. https://doi.org/10.1258/shorts.2011.011112
Funding
The study was funded by the Canadian Institutes of Health Research COVID-19 Rapid Research Funding Opportunity and the Canadian Immunity Task Force.
Author information
Authors and Affiliations
Contributions
Da In (Diane) Lee conceptualized and designed the study, carried out data collection and initial analyses, drafted the initial manuscript, and reviewed and revised the manuscript. Dr. Shelley Vanderhout, Mary Aglipay, and Dr. Jonathon Maguire conceptualized and designed the study, supervised data collection and analysis, and reviewed and revised the manuscript. Drs. Catherine S. Birken, Shaun K. Morris, and Pierre-Philippe Piché-Renaud conceptualized and designed the study, critically reviewed the manuscript for important intellectual content, and reviewed and revised the manuscript. Dr. Charles D.G. Keown-Stoneman conceptualized and designed the study, carried out data analyses, and reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
N/A.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, D.I.D., Vanderhout, S., Aglipay, M. et al. Delay in childhood vaccinations during the COVID-19 pandemic. Can J Public Health 113, 126–134 (2022). https://doi.org/10.17269/s41997-021-00601-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.17269/s41997-021-00601-9