Abstract
Background
R-spondin (RSPO)1 is a fibroblast-secreted protein that belongs to the R-spondin protein family which is essential for reproductive organ development, epithelial stem cell renewal and cancer induction or suppression. RSPO1 gene mutations cause palmoplantar hyperkeratosis with squamous cell carcinoma (SCC) of the skin, 46XX sex reversal and true hermaphroditism.
Objectives
To characterize RSPO1-deficient skin fibroblasts derived from two patients with mutations in RSPO1, with palmoplantar hyperkeratosis, recurrent SCC and 46XX sex reversal, to provide further insight into disease-related skin tumourigenesis.
Materials & Methods
Fibroblast cultures from non-tumoural palmoplantar skin biopsies were established to evaluate features and properties that may be altered at cancer onset, i.e. proliferation, extracellular matrix contraction and invasion, as well as TGF-β and matrix metalloproteinase (MMP) secretion.
Results
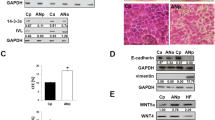
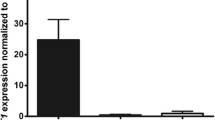
Fibroblasts demonstrated increased proliferative potential in vitro, a high level of collagen contraction and invasion by SCC cells, release of high levels of pro-inflammatory and pro-fibrotic TGF-β, and increased expression of MMP1 and MMP3. Analysis of the expression of selected proteins associated with RSPO1-activated pathways confirmed sustained activation of the TGF-β signalling pathway and indicated a loss of TGF-β inhibitory feedback. Also, treatment of fibroblasts with a recombinant RSPO1 protein aggravated this proinflammatory phenotype, suggesting caution in designing therapeutic strategies based on restoration of protein function.
Conclusion
Our findings indicate that fibroblasts from RSPO1 -mutated patients behave similarly to cancer-associated fibroblasts. Chronic inflammation and fibrotic changes in palmoplantar skin may play a role in SCC development and recurrence, possibly by irreversibly activating the tumourigenic phenotype of fibroblasts.
Similar content being viewed by others
References
Park S, Cui J, Yu W, Wu L, Carmon KS, Liu QJ. Differential activities and mechanisms of the four R-spondins in potentiating Wnt/β-catenin signaling. J Biol Chem 2018; 293: 9759–69, http://www.ncbi.nlm.nih.gov/pubmed/6016459.
Parma P, Radi O, Vidal V, et al. R-spondin1 is essential in sex determination, skin differentiation and malignancy. Nat Genet 2006; 38: 1304–9.
Tomaselli S, Megiorni F, De Bernardo C, et al. Syndromic true hermaphroditism due to an R-spondin1 (RSPO1) homozygous mutation. Hum Mutat 2008; 29: 220–6, http://www.ncbi.nlm.nih.gov/pubmed/18085567.
Naasse Y, Bakhchane A, Charoute H, et al. A novel homozygous missense mutation in the FU-CRD2 domain of the R-spondin1 gene associated with familial 46, XX DSD. Sex Dev 2017; 11: 269–74, http://www.ncbi.nlm.nih.gov/pubmed/29262419.
Tallapaka K, Venugopal V, Dalal A, Aggarwal S. Novel RSPO1 mutation causing 46, XX testicular disorder of sex development with palmoplantar keratoderma: a review of literature and expansion of clinical phenotype. Am J Med Genet A 2018; 176: 1006–10, http://www.ncbi.nlm.nih.gov/pubmed/29575617.
Guerriero C, Albanesi C, Girolomoni G, et al. Huriez syndrome: case report with a detailed analysis of skin dendritic cells. Br J Dermatol 2000; 143: 1091–6, http://www.ncbi.nlm.nih.gov/pubmed/11069529.
Micali G, Nasca MR, Innocenzi D, et al. Association of palmoplantar keratoderma, cutaneous squamous cell carcinoma, dental anomalies, and hypogenitalism in four siblings with 46, XX karyotype: a new syndrome. J Am Acad Dermatol 2005; 53: S234–9, http://www.ncbi.nlm.nih.gov/pubmed/16227098.
Radi O, Parma P, Imbeaud S, et al. XX sex reversal, palmoplantar keratoderma, and predisposition to squamous cell carcinoma: genetic analysis in one family. Am J Med Genet A 2005; 138A: 241–6, http://www.ncbi.nlm.nih.gov/pubmed/16158431.
Vernole P, Terrinoni A, Didona B, et al. An SRY-negative XX male with Huriez syndrome. Clin Genet 2000; 57: 61–6, http://www.ncbi.nlm.nih.gov/pubmed/10733237.
Riggio E, Spano A, Bonomi S, Nava M. Huriez syndrome: association with squamous cell carcinoma and a surgical approach. Plast Reconstr Surg 2005; 116: 689–91, http://www.ncbi.nlm.nih.gov/pubmed/16079734.
Zhou X, Geng L, Wang D, Yi H, Talmon G, Wang J. R-Spondin1/LGR5 activates TGFβ signaling and suppresses colon cancer metastasis. Cancer Res 2017; 77: 6589–602, http://www.ncbi.nlm.nih.gov/pubmed/28939678.
Chartier C, Raval J, Axelrod F, et al. Therapeutic targeting of tumor-derived R-spondin attenuates β-catenin signaling and tumorigenesis in multiple cancer types. Cancer Res 2016; 76: 713–23, http://www.ncbi.nlm.nih.gov/pubmed/26719531.
Dotto GP. Multifocal epithelial tumors and field cancerization: Stroma as a primary determinant. J Clin Invest 2014; 124: 1446–53.
Günther C, Lee-Kirsch MA, Eckhard J, et al. SMARCAD1 haploinsufficiency underlies Huriez syndrome and associated skin cancer susceptibility. J Invest Dermatol 2018; 138: 1428–31, http://www.ncbi.nlm.nih.gov/pubmed/29409814.
Panacchia L, Dellambra E, Bondanza S, et al. Nonirradiated human fibroblasts and irradiated 3T3-J2 murine fibroblasts as a feeder layer for keratinocyte growth and differentiation in vitro on a fibrin substrate. Cells Tissues Organs 2010; 191: 21–35, http://www.ncbi.nlm.nih.gov/pubmed/19546512.
Albrengues J, Bertero T, Grasset E, et al. Epigenetic switch drives the conversion of fibroblasts into proinvasive cancer-associated fibroblasts. Nat Commun 2015; 6: 1–15. doi: https://doi.org/10.1038/ncomms10204.
Rheinwald JG, Beckett MA. Tumorigenic keratinocyte lines requiring anchorage and fibroblast support cultured from human squamous cell carcinomas. Cancer Res 1981; 41: 1657–63, http://www.ncbi.nlm.nih.gov/pubmed/7214336.
Odorisio T, Di Salvio M, Orecchia A, et al. Monozygotic twins discordant for recessive dystrophic epidermolysis bullosa phenotype highlight the role of TGF-β signalling in modifying disease severity. Hum Mol Genet 2014; 23: 3907–22, http://www.ncbi.nlm.nih.gov/pubmed/24599399.
Alkasalias T, Moyano-Galceran L, Arsenian-Henriksson M, Lehti K. Fibroblasts in the tumor microenvironment: shield or spear? Int J Mol Sci 2018; 19: 1532, http://www.ncbi.nlm.nih.gov/pubmed/29883428.
Huschtscha LI. p16INK4a and the control of cellular proliferative life span. Carcinogenesis 1999; 20: 921–6, http://carcin.oxfordjournals.org/content/20/6/921.short.
Bates S, Phillips A, Clark P, Stott F. p14ARF links the tumour suppressors RB and p53. Nature 1998; 395: 3–4, http://www.nature.com/nature/journal/v395/n6698/abs/395124a0.html.
Guerra L, Odorisio T, Zambruno G, Castiglia D. Stromal microenvironment in type VII collagen-deficient skin: The ground for squamous cell carcinoma development. Matrix Biol 2017; 63: 1–10, http://www.ncbi.nlm.nih.gov/pubmed/28126522.
Luo K. Signaling cross talk between TGF-β/smad and other signaling pathways. Cold Spring Harb Perspect Biol 2017; 9: a022137, https://doi.org/10.1101/cshperspect.a022137.
Li WP, Anderson CJ. Imaging matrix metalloproteinase expression in tumors. Q J Nucl Med 2003; 47: 201–8, http://www.ncbi.nlm.nih.gov/pubmed/12897711.
Boudreau MW, Peh J, Hergenrother PJ. Procaspase-3 overexpression in cancer: a paradoxical observation with therapeutic potential. ACS Chem Biol 2019; 14: 2335–48, https://doi.org/10.1021/acschembio.9b00338.
Nakazono-Kusaba A, Takahashi-Yanaga F, Morimoto S, Furue M, Sasaguri T. Staurosporine-induced cleavage of α-smooth muscle actin during myofibroblast apoptosis. J Invest Dermatol 2002; 119: 1008–13. doi: https://doi.org/10.1046/j.1523-1747.2002.19525.x.
de Lau WBM, Snel B, Clevers HC. The R-spondin protein family. Genome Biol 2012; 13: 242, http://www.ncbi.nlm.nih.gov/pubmed/22439850.
Yoon JK, Lee J-S. Cellular signaling and biological functions of R-spondins. Cell Signal 2012; 24: 369–77, http://www.ncbi.nlm.nih.gov/pubmed/21982879.
Pickup MW, Mouw JK, Weaver VM. The extracellular matrix modulates the hallmarks of cancer. EMBO Rep 2014; 15: 1243–53, http://www.ncbi.nlm.nih.gov/pubmed/25381661.
Hinz B, Lagares D. Evasion of apoptosis by myofibroblasts: a hallmark of fibrotic diseases. Nat Rev Rheumatol 2020; 16: 11–31. doi: https://doi.org/10.1038/s41584-019-0324-5.
Acknowledgments and disclosures
Acknowledgments: SCC cells were kindly provided by Professor James G. Rheinwald (Department of Dermatology, Brigham and Women’s Hospital, Harvard Skin Disease Research Center, Boston, MA, USA). We thank Dr. Laura Panacchia for her collaboration with cell culture experiments. In memory of Dr. Liliana Guerra who died on August 23rd, 2020. Financial support: This work was supported by the Italian Ministry of Health (5 PER MILLE 2013 and RICERCA CORRENTE 2019 1.1) to LG. Conflicts of interest: none.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Dellambra, E., Cordisco, S., Proto, V. et al. RSPO1-mutated fibroblasts from non-tumoural areas of palmoplantar keratoderma display a cancer-associated phenotype. Eur J Dermatol 31, 342–350 (2021). https://doi.org/10.1684/ejd.2021.4066
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2021.4066