Abstract
Background
Relatively little is known about the true aggressive potential of pleomorphic dermal sarcoma (PDS) or optimal management strategies.
Objective
To describe the outcomes of 16 cases of PDS treated at our hospital (14 with modified Mohs micrographic surgery [M-MMS] and two with conventional surgery) and establish an adequate plan for management.
Materials & Methods
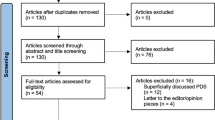
We reviewed 16 PDS cases treated at our hospital between October 2007 and June 2019 and compared our results with the available evidence.
Results
In total, 69% of cases had recurred after initial conventional surgery, M-MMS led to local disease control in 83% of cases, and 19% of patients developed metastasis. Combining all published PDS cases with ours, we calculated an overall metastasis rate of 12%, and an overall recurrence rate of 35% after conventional surgery and 17% after M-MMS.
Conclusion
PDS is more aggressive than previously estimated, with an overall metastatic rate of 12%. Despite high recurrence rates with previous conventional surgery (69%), M-MMS achieved a good rate of local disease control (83%). Given the potential aggressivity of PDS and the importance of clear surgical margins, M-MMS appears to be more adequate than conventional excision. Staging studies and close monitoring are warranted in PDS patients, for which we propose a management algorithm.
Similar content being viewed by others
References
Miller K, Goodlad JR, Brenn T. Pleomorphic dermal sarcoma: adverse histologic features predict aggressive behavior and allow distinction from atypical fibroxanthoma. Am J Surg Pathol 2012; 36: 1317–26.
Tardio JC, Pinedo F, Aramburu JA, et al. Pleomorphic dermal sarcoma: a more aggressive neoplasm than previously estimated. J Cutan Pathol 2016; 43: 101–12.
Brenn T. Pleomorphic dermal neoplasms: a review. Adv Anat Pathol 2014; 21: 108–30.
Mentzel T, Requena L, Brenn T. Atypical fibroxanthoma revisited. Surg Pathol Clin 2017; 10: 319–35.
Fletcher CD. Pleomorphic malignant fibrous histiocytoma: fact or fiction? A critical reappraisal based on 159 tumors diagnosed as pleomorphic sarcoma. Am J Sur Pathol 1992; 16: 213–28.
Soleymani T, Aasi SZ, Novoa R, Hollmig ST. Atypical fibroxanthoma and pleomorphic dermal sarcoma: updates on classification and management. Dermatol Clin 2019; 37: 253–9.
McCalmont TH. Correction and clarification regarding AFX and pleomorphic dermal sarcoma. J Cutan Pathol 2012; 39: 8.
Helwig EB, May D. Atypical fibroxanthoma of the skin with metastasis. Cancer 1986; 57: 368–76.
Iglesias-Pena N, Ló pez-Solache L, Martínez-Campayo N, et al. Incidence rate and clinicopathological features of 62 atypical fibroxanthomas in a North-Western Spanish population. Australas J Dermatol 2020; 61: e22–7.
Ziemer M, Jäger IM, Dippel E. Atypisches fibroxanthom und pleomorphes dermales sarkomatypical fibroxanthoma and pleomorphic dermal sarcoma. Der Hautarzt 2019; 70: 661–9.
Jacobs DS, Edwards WD, Ye RC. Metastatic atypical fibroxanthoma of skin. Cancer 1975; 35: 457–63.
Kemp JD, Stenn KS, Arons M, Fischer J. Metastasizing atypical fibroxanthoma. Coexistence with chronic lymphocytic leukemia. Arch Dermatol 1978; 114: 1533–5.
Doyle LA. Sarcoma classification: an update based on the 2013 World Health Organization Classification of Tumors of Soft Tissue and Bone. Cancer 2014; 120: 1763–74.
Winchester D, Lehman J, Tello T, et al. Undifferentiated pleomorphic sarcoma: factors predictive of adverse outcomes. J Am Acad Dermatol 2018; 79: 853–9.
Soleymani T, Tyler Hollmig S. Conception and management of a poorly understood spectrum of dermatologic neoplasms: atypical fibroxanthoma, pleomorphic dermal sarcoma, and undifferentiated pleomorphic sarcoma. Curr Treat Options Oncol 2017; 18: 50.
Fretzin DF, Helwig EB. Atypical fibroxanthoma of the skin. A clinicopathologic study of 140 cases. Cancer 1973; 31: 1541–52.
Iorizzo LJ 3rd, Brown MD. Atypical fibroxanthoma: a review of the literature. Dermatol Surg 2011; 37: 146–57.
Fletcher CD, Gustafson P, Rydholm A, Willen H, Akerman M. Clinicopathologic re-evaluation of 100 malignant fibrous histiocytomas: prognostic relevance of subclassification. J Clin Oncol 2001; 19: 3045–50.
Anderson ME, Rodic N, Subtil A, et al. Multifocal pleomorphic dermal sarcoma and the role of inflammation and immunosuppression in a lung transplant patient: a case report. J Med Case Rep 2019; 13: 169.
Llombart B, et al. Leiomiosarcoma y sarcoma pleomó rfico dérmico: directrices para el diagnó stico y tratamiento. Actas Dermosifiliogr 2019; 110: 4–11.
Hanlon A, Stasko T, Christiansen D, Cyrus N, Galan A. LN2, CD10, and ezrin do not distinguish between atypical fibroxanthoma and undifferentiated pleomorphic sarcoma or predict clinical outcome. Dermatol Surg 2017; 43: 431–6.
McCalmont TH. AFX: what we now know. J Cut Pathol 2011; 38: 853–6.
Koelsche C, Stichel D, Griewank KG, et al. Genome-wide methylation profiling and copy number analysis in atypical fibroxanthomas and pleomorphic dermal sarcomas indicate a similar molecular phenotype. Clin Sarcoma Res 2019; 9: 1–8.
Griewank KG, Schilling B, Murali R, et al. TERT promoter mutations are frequent in atypical fibroxanthomas and pleomorphic dermal sarcomas. Mod Pathol 2014; 27: 502–8.
Klein S, Mauch C, Wagener-Ryczek S, et al. Immune-phenotyping of pleomorphic dermal sarcomas suggests this entity as a potential candidate for immunotherapy. Cancer Immunol Immunother 2019; 68: 973–82.
Persa OD, Loquai C, Wobser M, et al. Extended surgical safety margins and ulceration are associated with an improved prognosis in pleomorphic dermal sarcomas. J Eur Acad Dermatol Venereol 2019; 33: 1577–80.
Tolkachjov SN, Kelley BF, Alahdab F, Erwin PJ, Brewer JD. Atypical fibroxanthoma: systematic review and meta-analysis of treatment with Mohs micrographic surgery or excision. J Am Acad Dermatol 2018; 79: 929–34.e6.
Ang GC, Roenigk RK, Otley CC, Kim Phillips P, Weaver AL. More than 2 decades of treating atypical fibroxanthoma at mayo clinic: what have we learned from 91 patients? Dermatol Surg 2009; 35: 765–72.
Serra-Guillen C, Llombart B, Nagore E, et al. Mohs micrographic surgery in dermatofibrosarcoma protuberans allows tumour clearance with smaller margins and greater preservation of healthy tissue compared with conventional surgery: a study of 74 primary cases. Br J Dermatol 2015; 172: 1303–7.
Llombart B, Monteagudo C, Sanmartín O, et al. Dermatofibrosarcoma protuberans: a clinicopathological, immunohistochemical, genetic (COL1A1-PDGFB), and therapeutic study of low-grade versus high-grade (fibrosarcomatous) tumors. J Am Acad Dermatol 2011; 65: 564–75.
Llombart B, Serra-Guillén C, Monteagudo C, Ló pez Guerrero JA, Sanmartín O. Dermatofibrosarcoma protuberans: a comprehensive review and update on diagnosis and management. Semin Diagn Pathol 2013; 30: 13–28.
Hornick JL. Subclassification of pleomorphic sarcomas: how and why should we care? Ann Diagn Pathol 2018; 37: 118–24.
Lum DJ, King AR. Peritoneal metastases from an atypical fibroxanthoma. Am J Surg Pathol 2006; 30: 1041–6.
de Feraudy S, Mar N, McCalmont TH. Evaluation of CD10 and procollagen 1 expression in atypical fibroxanthoma and dermatofibroma. Am J Surg Pathol 2008; 32: 1111–22.
Helbig D, Angelika Ihle M, Pütz K, et al. Oncogene and therapeutic target analyses in atypical fibroxanthomas and pleomorphic dermal sarcomas. Oncotarget 2016; 7: 21763–74.
Nergard J, Glener J, Reimer D, Greenwald JS. Atypical fibrox-anthoma of the scalp with recurrent and multiple regional cutaneous metastases. JAAD Case Reports 2016; 2: 491–3.
Garcia del Muro X, de Alava E, Artigas V, et al. Clinical practice guidelines for the diagnosis and treatment of patients with soft tissue sarcoma by the Spanish group for research in sarcomas (GEIS). Cancer Chemother Pharmacol 2016; 77: 133–46.
Vieira AC, Baccili Cury Megid T, Melo R, et al. Response to anti-PD1 immunotherapy in patients with metastatic cutaneous sarcoma: case reports and literature review. Oxf Med Case Reports 2020; 2020: 214.
Klein S, Persa O-D, Mauch C, et al. First report on two cases of pleomorphic dermal sarcoma successfully treated with immune checkpoint inhibitors. Oncoimmunology 2019; 8: e1665977.
Casali PG, Abecassis N, Aro HT, et al. Soft tissue and visceral sarcomas: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2018; 29: iv51–67.
von Mehren M, Randall RL, Benjamin RS, et al. Soft Tissue Sarcoma, Version 2.2018. NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Cancer Netw 2016; 14: 758–86.
Kravvas G, Veitch D, Logan IT, Perrett CM. Metastatic pleomorphic dermal sarcoma: an uncommon skin tumour. An Bras Dermatol 2018; 93: 307–8.
Ardakani NM, Pearce R, Wood BA. Pleomorphic dermal sarcoma with osteosarcoma-like and chondrosarcoma-like elements. Pathology 2016; 48: 86–9.
Sharma SR, Meligonis G, Todd P. Uncommon skin cancer: pleomorphic dermal sarcoma. BMJ Case Rep 2018; 2018: bcr2018224483.
Crimini E, Roberto M, Degli Effetti V, et al. Electrochemotherapy as promising treatment option in rare recurrent cutaneous neoplasm of the scalp: case report of an elderly patient. Case Rep Oncol Med 2019; 2019: 1–5.
Klebanov N, Hoang MP, Reddy BY. Pleomorphic dermal sarcoma of the scalp. Cureus 2018; 10: 1–5.
Kim J-I, Choi Y-J, Seo H-M, et al. Case of pleomorphic dermal sarcoma of the eyelid treated with micrographic surgery and secondary intention healing. Annal Dermatol 2016; 28: 632–6.
Müller CSL, Jungmann J, Pföhler C, Mohammad F, Rübe C, Vogt T. Bericht über die sofortige Bestrahlung eines schnell wachsenden Sarkoms der Kopfhaut vor dem Wundverschluss. J Ger Soc Dermatol 2016; 14: 539–42.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ríos-Viñuela, E., Serra-Guillén, C., Llombart, B. et al. Pleomorphic dermal sarcoma: a retrospective study of 16 cases in a dermato-oncology centre and a review of the literature. Eur J Dermatol 30, 545–553 (2020). https://doi.org/10.1684/ejd.2020.3875
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2020.3875