Abstract
Background
Surgical margins of melanoma vary from 5 mm to 1 or 2 cm depending on histology thickness (Breslow). This approach usually requires two surgical steps: excisional biopsy and further reexcision according to histology thickness. A previous systematic review showed that measuring melanoma thickness with high-resolution ultrasound imaging equipment correlates well with histological measurement of melanoma thickness. Therefore, we routinely determined tumour sonographic thickness in order to perform surgery as a single step.
Objectives
To determine the proportion of patients who receive onestep surgery with adequate margins based on sonographic measurement of melanoma thickness and identify the reasons for differences between these two measurements.
Materials & Methods
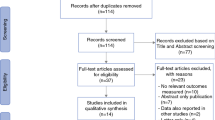
A retrospective series of patients with melanoma, in which thickness was measured by ultrasound (20 MHz) from April 2007 to December 2015 prior to surgery.
Results
Ninety-nine melanomas were treated, of which 78 were removed in a single step with surgical margins based on sonometric thickness measurements; 71 of these (91%, 95% CI: 82-96) did not require reexcision, five had excessive margins, and two had insufficient margins. The correlation between the histometric and sonometric measurements was good; r=0.88. Significant absolute difference between sonometric and histometric measurementswas associated with thickness, ulceration, and size of tumours, based on bivariate analysis. Thickness remained the only significant factor based on multivariate analysis.
Conclusions
Measuring the thickness of melanoma with high-resolution ultrasound imaging equipment makes it possible to remove the melanoma in a single step with adequate margins in at least 82% of the cases in routine care.
Similar content being viewed by others
References
Bichakjian CK, Halpern AC, Johnson TM, et al. Guidelines of care for the management of primary cutaneous melanoma. American Academy of Dermatology. J Am Acad Dermatol 2011; 65: 1032–47.
Garbe C, Peris K, Hauschild A, et al. Diagnosis and treatment of melanoma. European consensus-based interdisciplinary guideline -Update 2016. Eur J Cancer 2016; 63: 201–17.
Négrier S, Saiag P, Guillot B, et al. Guidelines for clinical practice: Standards, Options and Recommendations 2005 for the management of adult patients exhibiting an M0 cutaneous melanoma, full report. National Federation of Cancer Campaign Centers. French Dermatology Society. Update of the 1995 Consensus Conference and the 1998 Standards, Options, and Recommendations. Ann Dermatol Venereol 2005; 132(10): S3–85.
Guillot B, Dalac S, Denis MG, et al. Update to the recommendations for management of melanoma stages I to III. Ann Dermatol Venereol 2016; 143: 629–52.
Machet L, Belot V, Naouri M, et al. Preoperative measurement of thickness of cutaneous melanoma using high-resolution 20 MHz ultrasound imaging: a monocenter prospective study and systematic review of the literature. Ultrasound Med Biol 2009; 35: 1411–20.
Meyer N, Lauwers-Cances V, Lourari S, et al. High-frequency ultrasonography but not 930-nm optical coherence tomography reliably evaluates melanoma thickness in vivo: a prospective validation study. Br J Dermatol 2014; 171: 799–805.
Botar-Jid CM, Cosgarea R, Bolboacă SD, et al. Assessment of cutaneous melanoma by use of very high-frequency ultrasound and real-time elastography. Am J Roentgenol 2016; 206: 699–704.
Maj M, Warszawik-Hendzel O, Szymanska E, et al. High frequency ultrasonography: a complementary diagnostic method in evaluation of primary cutaneous melanoma. G Ital Dermatol Venereol 2015; 150: 595–601.
Crisan M, Crisan D, Sannino G, et al. Ultrasonographic staging of cutaneous malignant tumors: an ultrasonographic depth index. Arch Dermatol Res 2013; 305: 305–13.
Fernández Canedo I, de Troya Martín M, Fúnez Liébana R, Rivas Ruiz F, Blanco Eguren G, Blázquez Sánchez N. Preoperative 15-MHz ultrasound assessment of tumor thickness in malignant melanoma. Actas Dermosifilogr 2013; 104: 227–31.
Hinz T, Ehler L-K, Voth H, et al. Assessment of tumor thickness in melanocytic skin lesions: comparison of optical coherence tomography, 20-MHz ultrasound and histopathology. Dermatol Basel Switz 2011; 223: 161–8.
Kaikaris V, Samsanavičius D, Maslauskas K, et al. Measurement of melanoma thickness-comparison of two methods: ultrasound versus morphology. J Plast Reconstr Aesthetic Surg 2011; 64: 796–802.
Music MM, Hertl K, Kadivec M, Pavlovi´c MD, Hocevar M. Pre-operative ultrasound with a 12-15 MHz linear probe reliably differentiates between melanoma thicker and thinner than 1 mm. J Eur Acad Dermatol Venereol 2010; 24: 1105–8.
Vilana R, Puig S, Sanchez M, et al. Preoperative assessment of cutaneous melanoma thickness using 10-MHz sonography. Am J Roentgenol 2009; 193: 639–43.
Guitera P, Li LX, Crotty K, et al. Melanoma histological Breslow thickness predicted by 75-MHz ultrasonography. Br J Dermatol 2008; 159: 364–9.
Hayashi K, Koga H, Uhara H, Saida T. High-frequency 30-MHz sonography in preoperative assessment of tumor thickness of primary melanoma: usefulness in determination of surgical margin and indication for sentinel lymph node biopsy. Int J Clin Oncol 2009; 14: 426–30.
Andrekute K, Valiukeviciene S, Raisutis R, et al. Automated estimation of melanocytic skin tumor thickness by ultrasonic radiofrequency data. J Ultrasound Med 2016; 35: 857–65.
Varkentin A, Mazurenka M, Blumenröther E, et al. Comparative study of presurgical skin infiltration depth measurements of melanocytic lesions with OCT and high frequency ultrasound. J Biophotonics 2017; 10: 854–61.
Semple JL, Gupta AK, From L, et al. Does high-frequency (40-60 MHz) ultrasound imaging play a role in the clinical management of cutaneous melanoma? Ann Plast Surg 1995; 34: 599–605.
Schmid-Wendtner M-H, Burgdorf W. Ultrasound scanning in dermatology. Arch Dermatol 2005; 141: 217–24.
Gassenmaier G, Kiesewetter F, Schell H, Zinner M. Value of high resolution ultrasound in determination of vertical tumor thickness in malignant melanoma of the skin. Hautarzt 1990; 41: 360–4.
Hoffmann K, Jung J, el Gammal S, Altmeyer P. Malignant melanoma in 20-MHz B scan sonography. Dermatol Basel Switz 1992; 185: 49–55.
Sondak VK, Zager JS. Who is to blame for false-negative sentinel node biopsies in melanoma? Ann Surg Oncol 2010; 17: 670–3.
Kelemen PR, Essner R, Foshag LJ, Morton DL. Lymphatic mapping and sentinel lymphadenectomy after wide local excision of primary melanoma. J Am Coll Surg 1999; 189: 247–52.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Chaput, L., Laurent, E., Pare, A. et al. One-step surgical removal of cutaneous melanoma with surgical margins based on preoperative ultrasound measurement of the thickness of the melanoma. Eur J Dermatol 28, 202–208 (2018). https://doi.org/10.1684/ejd.2018.3298
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1684/ejd.2018.3298