Abstract
Objective
Drug-resistance and metastasis are major reasons for the high mortality of ovarian cancer (OC) patients. Cyclooxygenase-2 (COX-2) plays a critical role in OC development. This study was designed to evaluate the effects of COX-2 on migration and cisplatin (cis-dichloro diammine platinum, CDDP) resistance of OC cells and explore its related mechanisms.
Methods
Cell counting kit-8 (CCK-8) assay was used to detect the cytotoxicity effects of celecoxib (CXB) and CDDP on SKOV3 and ES2 cells. The effect of COX-2 on migration was evaluated via the healing test. Western blot and real-time quantitative polymerase chain reaction (qPCR) were used to analyze E-cadherin, vimentin, Snail, and Slug levels.
Results
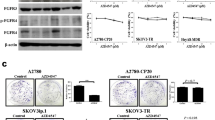
COX-2 promoted drug-resistance and cell migration. CXB inhibited these effects. The combination of CDDP and CXB increased tumor cell sensitivity, reduced the amount of CDDP required, and shortened treatment administration time. COX-2 upregulation increased the expression of Snail and Slug, resulting in E-cadherin expression downregulation and vimentin upregulation.
Conclusions
COX-2 promotes cancer cell migration and CDDP resistance and may serve as a potential target for curing OC.
摘要
目 的
研究环氧合酶-2 (COX-2) 对卵巢癌细胞迁移和耐药性的影响及其机制.
创新点
本研究发现COX-2对卵巢癌发生发展有一定的促进作用, 可以通过上皮间质转化 (EMT) 途径促进卵巢癌细胞的迁移和顺铂 (CDDP) 耐药. 其抑制剂塞来昔布 (CXB) 能起到协同抗癌的效果.
方 法
CCK-8 检测 CXB 和 CDDP 对 SKOV3 和 ES2 细胞的毒性作用. 划痕实验评估 COX-2 对卵巢癌细胞迁移的作用. 蛋白质免疫印迹 (western blot) 和聚合酶链式反应 (PCR) 检测 EMT 相关基因和蛋白的表达水平.
结 论
COX-2 可以通过 EMT 促进卵巢癌细胞迁移和 CDDP 耐药; CXB 可以起到抑制作用, 与 CDDP 协同抗癌. COX-2 可以作为卵巢癌治疗的一个潜在靶点.
Similar content being viewed by others
References
JNCI (Journal of the National Cancer Institute), 2011. Ovarian cancer, five-year stage-specific relative survival rates (2004–2008). J Natl Cancer Inst, 103(17):1287. https://doi.org/10.1093/jnci/djr342
al-Wadei HAN, al-Wadei MH, Ullah MF, et al., 2012. Celecoxib and GABA cooperatively prevent the progression of pancreatic cancer in vitro and in xenograft models of stress-free and stress-exposed mice. PLoS ONE, 7(8):e43376. https://doi.org/10.1371/journal.pone.0043376
Barnes AP, Miller BE, Kucera GL, 2007. Cyclooxygenase inhibition and hyperthermia for the potentiation of the cytotoxic response in ovarian cancer cells. Gynecol Oncol, 104(2):443–450. https://doi.org/10.1016/j.ygyno.2006.08.008
Bertagnolli MM, Eagle CJ, Zauber AG, et al., 2009. Five-year efficacy and safety analysis of the adenoma prevention with celecoxib trial. Cancer Prev Res, 2(4):310–321. https://doi.org/10.1158/1940-6207.Capr-08-0206
Buys SS, Partridge E, Black A, et al., 2011. Effect of screening on ovarian cancer mortality: the prostate, lung, colorectal and ovarian (PLCO) cancer screening randomized controlled trial. JAMA, 305(22):2295–2303. https://doi.org/10.1001/jama.2011.766
Cervello M, Bachvarov D, Lampiasi N, et al., 2013. Novel combination of sorafenib and celecoxib provides synergistic anti-proliferative and pro-apoptotic effects in human liver cancer cells. PLoS ONE, 8(6):e65569. https://doi.org/10.1371/journal.pone.0065569
Chen FL, Wen X, Lin PF, et al., 2019. HERP depletion inhibits zearalenone-induced apoptosis through autophagy activation in mouse ovarian granulosa cells. Toxicol Lett, 1: 1–10. https://doi.org/10.1016/j.toxlet.2018.10.026
Chen MHS, Yip GWC, Tse GMK, et al., 2008. Expression of basal keratins and vimentin in breast cancers of young women correlates with adverse pathologic parameters. Mod Pathol, 21(10):1183–1191. https://doi.org/10.1038/modpathol.2008.90
Chen XL, Lingala S, Khoobyari S, et al., 2011. Epithelial mesenchymal transition and hedgehog signaling activation are associated with chemoresistance and invasion of hepatoma subpopulations. J Hepatol, 55(4):838–845. https://doi.org/10.1016/j.jhep.2010.12.043
Cho EY, Choi Y, Chae SW, et al., 2006. Immunohistochemical study of the expression of adhesion molecules in ovarian serous neoplasms. Pathol Int, 56(2):62–70. https://doi.org/10.1111/j.1440-1827.2006.01925.x
Chung LY, Tang SJ, Sun GH, et al., 2012. Galectin-1 promotes lung cancer progression and chemoresistance by upregulating p38 MAPK, ERK, and cyclooxygenase-2. Clin Cancer Res, 18(15):4037–4047. https://doi.org/10.1158/1078-0432.Ccr-11-3348
Denkert C, Fürstenberg A, Daniel PT, et al., 2003. Induction of G0/G1 cell cycle arrest in ovarian carcinoma cells by the anti-inflammatory drug NS-398, but not by COX-2-specific RNA interference. Oncogene, 22(54):8653–8661. https://doi.org/10.1038/sj.onc.1206920
Denkert C, Weichert W, Pest S, et al., 2004. Overexpression of the embryonic-lethal abnormal vision-like protein HuR in ovarian carcinoma is a prognostic factor and is associated with increased cyclooxygenase 2 expression. Cancer Res, 64(1):189–195. https://doi.org/10.1158/0008-5472.CAN-03-1987
Ferrandina G, Lauriola L, Distefano MG, et al., 2002. Increased cyclooxygenase-2 expression is associated with chemotherapy resistance and poor survival in cervical cancer patients. J Clin Oncol, 20(4):973–981. https://doi.org/10.1200/jco.2002.20.4.973
Ferrandina G, Zannoni GF, Ranelletti FO, et al., 2004. Cyclooxygenase-2 expression in borderline ovarian tumors. Gynecol Oncol, 95(1):46–51. https://doi.org/10.1016/j.ygyno.2004.07.005
Gartung A, Yang J, Sukhatme VP, et al., 2019. Suppression of chemotherapy-induced cytokine/lipid mediator surge and ovarian cancer by a dual COX-2/sEH inhibitor. Proc Natl Acad Sci USA, 116(5):1698–1703. https://doi.org/10.1073/pnas.1803999116
Giaginis C, Alexandrou P, Tsoukalas N, et al., 2015. Huantigen receptor (HuR) and cyclooxygenase-2 (COX-2) expression in human non-small-cell lung carcinoma: associations with clinicopathological parameters, tumor proliferative capacity and patients’ survival. Tumour Biol, 36(1):315–327. https://doi.org/10.1007/s13277-014-2637-y
Harris RE, Casto BC, Harris ZM, 2014. Cyclooxygenase-2 and the inflammogenesis of breast cancer. World J Clin Oncol, 5(4):677–692. https://doi.org/10.5306/wjco.v5.i4.677
Hu Z, Liu XJ, Tang ZF, et al., 2013. Possible regulatory role of Snail in NF-κB-mediated changes in E-cadherin in gastric cancer. Oncol Rep, 29(3):993–1000. https://doi.org/10.3892/or.2012.2200
Jemal A, Siegel R, Xu JQ, et al., 2010. Cancer statistics, 2010. CA Cancer J Clin, 60(5):277–300. https://doi.org/10.3322/caac.20073
Kim HJ, Yim GW, Nam EJ, et al., 2014. Synergistic effect of COX-2 inhibitor on paclitaxel-induced apoptosis in the human ovarian cancer cell line OVCAR-3. Cancer Res Treat, 46(1):81–92. https://doi.org/10.4143/crt.2014.46.L81
Kismet K, Akay MT, Abbasoglu O, et al., 2004. Celecoxib: a potent cyclooxygenase-2 inhibitor in cancer prevention. Cancer Detect Prev, 28(2):127–142. https://doi.org/10.1016/j.cdp.2003.12.005
Konstan MW, Byard PJ, Hoppel CL, et al., 1995. Effect of high-dose ibuprofen in patients with cystic fibrosis. N Engl J Med, 332(13):848–854. https://doi.org/10.1056/nejm199503303321303
Koti M, Siu A, Clément I, et al., 2015. A distinct pre-existing inflammatory tumour microenvironment is associated with chemotherapy resistance in high-grade serous epithelial ovarian cancer. Br J Cancer, 112(7):1215é1222. https://doi.org/10.1038/bjc.2015.81
Landen CN Jr., Mathur SP, Richardson MS, et al., 2003. Expression of cyclooxygenase-2 in cervical, endometrial, and ovarian malignancies. Am J Obstet Gynecol, 188(5): 1174–1176. https://doi.org/10.1067/mob.2003.284
Legge F, Paglia A, D’Asta M, et al., 2011. Phase II study of the combination carboplatin plus celecoxib in heavily pretreated recurrent ovarian cancer patients. BMC Cancer, 11:214. https://doi.org/10.1186/1471-2407-11-214
Li SF, Miner K, Fannin R, et al., 2004. Cyclooxygenase-1 and 2 in normal and malignant human ovarian epithelium. Gynecol Oncol, 92(2):622–627. https://doi.org/10.1016/j.ygyno.2003.10.053
Li SS, Ma J, Wong AST, 2018. Chemoresistance in ovarian cancer: exploiting cancer stem cell metabolism. J Gynecol Oncol, 29(2):e32. https://doi.org/10.3802/jgo.2018.29.e32
Madan RA, Xia Q, Chang VT, et al., 2007. A retrospective analysis of cardiovascular morbidity in metastatic hormonerefractory prostate cancer patients on high doses of the selective COX-2 inhibitor celecoxib. Expert Opin Pharmacother, 8(10):1425–1431. https://doi.org/10.1517/14656566.8.10.1425
Neumann W, Crews BC, Sárosi MB, et al., 2015. Conjugation of cisplatin analogues and cyclooxygenase inhibitors to overcome cisplatin resistance. ChemMedChem, 10(1):183–192. https://doi.org/10.1002/cmdc.201402353
Okamura H, Fujiwara H, Umehara S, et al., 2013. COX-2 overexpression induced by gene transfer reduces sensitivity of TE13 esophageal carcinoma cells to 5-fluorouracil and cisplatin. Anticancer Res, 33(2):537–542.
Raspollini MR, Amunni G, Villanucci A, et al., 2005. Increased cyclooxygenase-2 (COX-2) and p-glycoprotein-170 (MDR1) expression is associated with chemotherapy resistance and poor prognosis. Analysis in ovarian carcinoma patients with low and high survival. Int J Gynecol Cancer, 15(2):255–260. https://doi.org/10.1136/ijgc-00009577-200503000-00011
Sangoi AR, Soslow RA, Teng NN, et al., 2008. Ovarian clear cell carcinoma with papillary features: a potential mimic of serous tumor of low malignant potential. Am J Surg Pathol, 32(2):269–274. https://doi.org/10.1097/PAS.0b013e31814fa9b0
Shigemasa K, Tian X, Gu L, et al., 2003. Expression of cyclooxygenase-2 and its relationship to p53 accumulation in ovarian adenocarcinomas. Int J Oncol, 22(1):99–105.
Soslow RA, Dannenberg AJ, Rush D, et al., 2000. COX-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer, 89(12):2637–2645. https://doi.org/10.1002/1097-0142(20001215)89:12<2637::aid-cncr17>3.0.co;2-b
Subbaramaiah K, Hart JC, Norton L, et al., 2000. Microtubule-interfering agents stimulate the transcription of cyclooxygenase-2. Evidence for involvement of ERK1/2 and p38 mitogen-activated protein kinase pathways. J Biol Chem, 275(20):14838–14845. https://doi.org/10.1074/jbc.275.20.14838
Wei J, Xu G, Wu M, et al., 2008. Overexpression of vimentin contributes to prostate cancer invasion and metastasis via SRC regulation. Anticancer Res, 28(1A):327–334.
Yim GW, Kim HJ, Kim LK, et al., 2017. Long non-coding RNA HOXA11 antisense promotes cell proliferation and invasion and predicts patient prognosis in serous ovarian cancer. Cancer Res Treat, 49(3):656–668. https://doi.org/10.4143/crt.2016.263
Zhai XL, Zhu HJ, Wang W, et al., 2014. Abnormal expression of EMT-related proteins, S100A4, vimentin and E-cadherin, is correlated with clinicopathological features and prognosis in HCC. Med Oncol, 31(6):970. https://doi.org/10.1007/s12032-014-0970-z
Zhang RR, Zhang P, Wang H, et al., 2015. Inhibitory effects of metformin at low concentration on epithelial-mesenchymal transition of CD44+CD117+ ovarian cancer stem cells. Stem Cell Res Ther, 6(1):262. https://doi.org/10.1186/s13287-015-0249-0
Zhao Y, Yan QM, Long X, et al., 2008. Vimentin affects the mobility and invasiveness of prostate cancer cells. Cell Biochem Funct, 26(5):571–577. https://doi.org/10.1002/cbf.1478
Acknowledgments
The authors would like to thank Prof. Zhong-jun DONG and Dr. Juan DU (Tsinghua University, Beijing, China) for technical support.
Author information
Authors and Affiliations
Contributions
Lin DENG and Bin LING designed experiments. Lin DENG carried out experiments, analyzed experimental results, and wrote the manuscript. Bin LING and Ding-qing FENG revised the manuscript. All authors have read and approved the final manuscript, have full access to all the data in the study, and take responsibility for the integrity and security of the data.
Corresponding author
Ethics declarations
Lin DENG, Ding-qing FENG, and Bin LING declare that they have no conflict of interest.
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Project supported by the National Natural Science Foundation of China (No. 81372777)
Rights and permissions
About this article
Cite this article
Deng, L., Feng, Dq. & Ling, B. Cyclooxygenase-2 promotes ovarian cancer cell migration and cisplatin resistance via regulating epithelial mesenchymal transition. J. Zhejiang Univ. Sci. B 21, 315–326 (2020). https://doi.org/10.1631/jzus.B1900445
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1900445
Key words
- Ovarian cancer (OC)
- Cyclooxygenase-2 (COX-2)
- Drug resistance
- Migration
- Epithelial-mesenchymal transition (EMT)