Abstract
A menaquinone-7 (MK-7) high-producing strain needs to be isolated to increase MK-7 production, in order to meet a requirement of MK-7 given the low MK-7 content in food products. This article focuses on developing MK-7 high-producing strains via screening and mutagenesis by an atmospheric and room temperature plasma (ARTP) mutation breeding system. We isolated an MK-7-producing strain Y-2 and identified it as Bacillus amyloliquefaciens, which produced (7.1±0.5) mg/L of MK-7 with maize meal hydrolysate as carbon source. Then, an MK-7 highproducing strain B. amyloliquefaciens H.β.D.R.-5 with resistance to 1-hydroxy-2-naphthoic acid, β-2-thienylalanine, and diphenylamine was obtained from the mutation of the strain Y-2 using an ARTP mutation breeding system. Using strain H.β.D.R.-5, efficient production of MK-7 was achieved ((30.2±2.7) mg/L). In addition, the effects of nitrogen sources, prenyl alcohols, and MgSO4 on MK-7 production were investigated, suggesting that soymeal extract combined with yeast extract, isopentenol, and MgSO4 was beneficial. Under the optimized condition, the MK-7 production and biomass-specific yield reached (61.3±5.2) mg/L and 2.59 mg/L per OD600 unit respectively in a 7-L fermenter. These results demonstrated that strain H.β.D.R.-5 has the capacity to produce MK-7 from maize meal hydrolysate, which could reduce the substrate cost.
摘要
目的
通过从自然界中筛选和传统诱变育种相结合的方 法,获得一株以玉米水解液为底物且能高效合成 甲萘醌-7(MK-7)的芽孢杆菌突变菌株。
创新点
首次在中国的发酵豆制品——豆豉中分离得到一 株能以玉米水解液为底物合成MK-7 的解淀粉芽 孢杆菌Y-2(Bacillus amyloliquefaciens Y-2),并 通过传统诱变育种获得一株带有二苯胺和结构 类似物抗性的、以玉米水解液为底物的、高产 MK-7 的菌株B. amyloliquefaciens H.β.D.R.-5。
方法
以来自中国不同省市地区的豆豉为分离样品,筛 选高产纳豆激酶的菌株,再从中挑选出高产 MK-7 的菌株,并通过16S rDNA 分析对其种属进 行鉴定。采用常压室温等离子体(ARTP)系统, 对分离到的高产MK-7 菌株进行诱变处理,获得 解除3-脱氧-D-阿拉伯庚酮糖-7-磷酸合成酶(即 结构类似物抗性)和聚丙烯焦磷酸合成酶(即二 苯胺抗性)反馈调节的菌株。最后,考察不同氮 源、乙戊烯醇和镁离子(Mg2+)对突变菌株合成 MK-7 的影响,并分析在7 L 发酵罐中合成MK-7 的区别。
结论
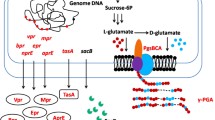
从中国豆豉中分离到了一株以玉米水解液为底物 合成MK-7 的菌株,经16S rDNA 分析比对,鉴 定为Bacillus amyloliquefaciens(图1)。通过比 较MK-7 产量,发现利用ARTP 可以有效获得解 除反馈调节作用的且高产MK-7 的突变菌株 H.β.D.R.-5(表1)。以大豆水解液和酵母水解液 为氮源, 异戊醇和MgSO4 有利于突变菌株 H.β.D.R.-5 合成MK-7(图2、表2 和表3)。综 上所述,利用ARTP 处理从中国豆豉中分离到的 以玉米水解液为底物的合成MK-7 的菌株,可获 得高产的MK-7 菌株,该方法对选育工业化合成 MK-7 的菌株有重要参考价值。
Similar content being viewed by others
References
Armougom, F., Bittar, F., Stremler, N., et al., 2009. Microbial diversity in the sputum of a cystic fibrosis patient studied with 16S rDNA pyrosequencing. Eur. J. Clin. Microbiol. Infect. Dis., 28(9):1151–1154. http://dx.doi.org/10.1007/s10096-009-0749-x
Berenjian, A., Mahanama, R., Talbot, A., et al., 2011. Efficient media for high menaquinone-7 production: response surface methodology approach. New Biotechnol., 28(6): 665–672. http://dx.doi.org/10.1016/j.nbt.2011.07.007
Chen, J.N., Yang, W.S., Dick, K., et al., 2008. Tip-enhanced Raman scattering of p-thiocresol molecules on individual gold nanoparticles. Appl. Phys. Lett., 92:093110. http://dx.doi.org/10.1063/1.2837543
Chen, Z.M., Li, Q., Liu, H.M., et al., 2010. Greater enhancement of Bacillus subtilis spore yields in submerged cultures by optimization of medium composition through statistical experimental designs. Appl. Microbiol. Biotechnol., 85(5):1353–1360. http://dx.doi.org/10.1007/s00253-009-2162-x
Fernandez, F., Collins, M.D., 1987. Vitamin K composition of anaerobic gut bacteria. FEMS Microbiol. Lett., 41(2): 175–180. http://dx.doi.org/10.1111/j.1574-6968.1987.tb02191.x
Fujii, H., Sagami, H., Koyama, T., et al., 1980. Variable product specificity of solanesyl pyrophosphate synthetase. Biochem. Biophys. Res. Commun., 96(4):1648–1653. http://dx.doi.org/10.1016/0006-291X(80)91363-7
AQSIQ (General Administration of Quality Supervision, Inspection and Quarantine of the People’s Republic of China), SAC (Standardization Administration of the People’s Republic of China), 2007. GB/T 20885-2007: Glucose Syrup.
Howard, L.M., Payne, A.C., 2006. Health Benefits of Vitamin K2: A Revolutionary Natural Treatment for Heart Disease and Bone Loss. Basic Health Publications, California.
Kim, Y.K., Kim, S.M., Kim, J.Y., et al., 2011. The culture filtrates from Bacillus subtilis natto lowers blood pressure via renin-angiotensin system in spontaneously hypertensive rats fed with a high-cholesterol diet. J. Korean Soc. Appl. Biol. Chem., 54(6):959–965. http://dx.doi.org/10.1007/BF03253186
Li, H.G., Ofosu, F.K., Li, K.T., et al., 2014. Acetone, butanol, and ethanol production from gelatinized cassava flour by a new isolates with high butanol tolerance. Bioresour. Technol., 172:276–282. http://dx.doi.org/10.1016/j.biortech.2014.09.058
Li, H.P., Wang, L.Y., Li, G., et al., 2011. Manipulation of lipase activity by the helium radio-frequency, atmosphericpressure glow discharge plasma jet. Plasma Proc. Polym., 8(3):224–229. http://dx.doi.org/10.1002/ppap.201000035
Liu, Y., Zhang, Z.M., Qiu, H.W., et al., 2014. Surfactant supplementation to enhance the production of vitamin K2 metabolites in shSphingobacterium multivorumake flask cultures using Escherichia sp. mutant FM3-1709. Food Technol. Biotechnol., 52(3): 269–275.
Lorenzi, V., Muselli, A., Bernardini, A.F., et al., 2009. Geraniol restores antibiotic activities against multidrugresistant isolates from Gram-negative species. Antimicrob. Agents Chemother., 53(5):2209–2211. http://dx.doi.org/10.1128/AAC.00919-08
Patil, S.R., Dayanand, A., 2006. Optimization of process for the production of fungal pectinases from deseeded sunflower head in submerged and solid-state conditions. Bioresour. Technol., 97(18):2340–2344. http://dx.doi.org/10.1016/j.biortech.2005.10.025
Rosa-Putra, S., Hemmerlin, A., Epperson, J., et al., 2001. Zeaxanthin and menaquinone-7 biosynthesis in Sphingobacterium multivorum via the methylerythritol phosphate pathway. FEMS Microbiol. Lett., 204(2):347–353. http://dx.doi.org/10.1111/j.1574-6968.2001.tb10909.x
Sagami, H., Ogura, K., Seto, S., 1977. Solanesyl pyrophosphate synthetase from Micrococcus lysodeikticus. Biochemistry, 16(21):4616–4622. http://dx.doi.org/10.1021/bi00640a014
Saito, Y., Ogura, K., 1981. Biosynthesis of menaquinones. Enzymatic prenylation of 1,4-dihydroxy-2-naphthoate by Micrococcus luteus membrane fractions. J. Biochem., 89(5):1445–1452. http://dx.doi.org/10.1093/oxfordjournals.jbchem.a133337
Sato, T., Yamada, Y., Ohtani, Y., et al., 2001a. Efficient production of menaquinone (vitamin K2) by a menadioneresistant mutant of Bacillus subtilis. J. Ind. Microbiol. Biotech., 26(3):115–120. http://dx.doi.org/10.1038/sj.jim.7000089
Sato, T., Yamada, Y., Ohtani, Y., et al., 2001b. Production of menaquinone (vitamin K2)-7 by Bacillus subtilis. J. Biosci. Bioeng., 91(1):16–20. http://dx.doi.org/10.1016/S1389-1723(01)80104-3
Song, J.Y., Liu, H.X., Wang, L., et al., 2014. Enhanced production of vitamin K2 from Bacillus subtilis (natto) by mutation and optimization of the fermentation medium. Braz. Arch. Biol. Technol., 57(4):606–612. http://dx.doi.org/10.1590/S1982-88372014000100019
Takahashi, I., Ogura, K., Seto, S., 1980. Heptaprenyl pyrophosphate synthetase from Bacillus subtilis. J. Biol. Chem., 255:4539–4543.
Tani, Y., Asahi, S., Yamada, H., 1985. Production of menaquinone (vitamin K2)-5 by a hydroxynaphthoate-resistant mutant derived from Flavobacterium meningosepticum, a menaquinone-6 producer. Agric. Biol. Chem., 49(1): 111–115. http://dx.doi.org/10.1080/00021369.1985.10866688
Tsukamoto, Y., Kasai, M., Kakuda, H., 2001. Construction of a Bacillus subtilis (natto) with high productivity of vitamin K2 (menaquinone-7) by analog resistance. Biosci. Biotechnol. Biochem., 65(9):2007–2015. http://dx.doi.org/10.1271/bbb.65.2007
Unnanuntana, A., Bonsignore, L., Shirtliff, M.E., et al., 2009. The effects of farnesol on Staphylococcus aureus biofilms and osteoblasts. An in vitro study. J. Bone Joint. Surg. Am., 91(11):2683–2692. http://dx.doi.org/10.2106/JBJS.H.01699
Walther, B., Karl, J.P., Booth, S.L., et al., 2013. Menaquinones, bacteria, and the food supply: the relevance of dairy and fermented food products to vitamin K requirements. Adv. Nutr., 4:463–473. http://dx.doi.org/10.3945/an.113.003855
Wee, Y.J., Reddy, L.V.A., Ryu, W.H., 2008. Fermentative production of L(+)-lactic acid from starch hydrolyzate and corn steep liquor as inexpensive nutrients by batch culture of Enterococcus faecalis RKY1. J. Chem. Technol. Biotechnol., 83(10):1387–1393. http://dx.doi.org/10.1002/jctb.1953
Wu, W.J., Ahn, B.Y., 2011. Isolation and identification of Bacillus amyloliquefaciens BY01 with high productivity of menaquinone for cheonggukjang production. J. Korean Soc. Appl. Biol. Chem., 54(5):783–789. http://dx.doi.org/10.1007/BF03253160
Xu, J.Z., Han, M., Zhang, J.L., et al., 2014. Metabolic engineering Corynebacterium glutamicum for the L-lysine production by increasing the flux into L-lysine biosynthetic pathway. Amino Acids, 46(9):2165–2175. http://dx.doi.org/10.1007/s00726-014-1768-1
Yanagisawa, Y., Sumi, H., 2005. Natto bacillus contains a large amount of water-soluble vitamin K (menaquinone-7). J. Food Biochem., 29(3):267–277. http://dx.doi.org/10.1111/j.1745-4514.2005.00016.x
Zhang, X., Zhang, X.F., Li, H.P., et al., 2014. Atmospheric and room temperature plasma (ARTP) as a new powerful mutagenesis tool. Appl. Microbiol. Biotechnol., 98(12): 5387–5396. http://dx.doi.org/10.1007/s00253-014-5755-y
Zhang, X., Zhang, C., Zhou, Q.Q., et al., 2015. Quantitative evaluation of DNA damage and mutation rate by atmospheric and room-temperature plasma (ARTP) and conventional mutagenesis. Appl. Microbiol. Biotechnol., 99(13): 5639–5646. http://dx.doi.org/10.1007/s00253-015-6678-y
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Natural Science Foundation of Jiangsu Province (No. BK20150149), China
ORCID: Jian-zhong XU, http://orcid.org/0000-0001-6555-6059
Rights and permissions
About this article
Cite this article
Xu, Jz., Zhang, Wg. Menaquinone-7 production from maize meal hydrolysate by Bacillus isolates with diphenylamine and analogue resistance. J. Zhejiang Univ. Sci. B 18, 462–473 (2017). https://doi.org/10.1631/jzus.B1600127
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1600127
Key words
- Menaquinone-7
- Bacillus amyloliquefaciens
- Analog resistance
- Diphenylamine resistance
- Maize meal hydrolysate