Abstract
The effects of crude protein (CP) levels in the diet on the mRNA expression of amino acid (AA) transporters were studied in a 45-d trial. Eighteen piglets with an initial body weight (BW) of 9.57 kg were assigned to three groups (14%, 17%, and 20% CP in the diet) in a completely randomized design (six replicates per treatment). Diets were supplemented with crystalline AA to achieve equal standardized ileal digestible contents of Lys, Met plus Cys, Thr, and Trp, and were provided ad libitum. After 45 d, all piglets were slaughtered to collect small intestine samples. Compared with the values in the 14% CP group, the expressions of ASCT2, 4F2hc, and ATB0 mRNA in the jejunum were increased by 23.00%, 12.00%, 6.00% and 48.00%, 47.00%, 56.00% in the 17% and 20% CP groups, respectively. These results indicate that a 14% CP diet supplemented with crystalline AA may not transport enough AA into the body and maintain growth performance of piglets. However, a reduction of dietary 17% CP may reduce the excretion of nitrogen into the environment while supporting the development of piglets. Therefore, the 17% CP level is more suitable than 14% CP level.
概要
目的
验证日粮中粗蛋白质浓度对断奶仔猪氨基酸转运载体的影响效应。
创新点
从营养物质感应体的角度分析采用低氮日粮的可 行性, 并探索营养限制对营养转运体的影响及它 们间的相互作用。
方法
十八头初始体重9.57 公斤的断奶仔猪被随机分为三组 (每组6 个重复), 分别饲喂含有14%、17%和20%的粗蛋白日粮45 天。按照理想蛋白质模型, 日粮分别添加赖氨酸、蛋氨酸+半胱氨酸、苏氨酸和色氨酸来满足断奶仔猪的需要, 整个试验期自由采食。试验结束后, 屠宰仔猪并采集小肠样品。与14%粗蛋白组相比, 空肠中ASCT2、4F2hc 和ATB0 mRNA 表达在17%和20%粗蛋白水平组分别上升23.00% 、12.00% 、6.00%和48.00%、47.00%、56.00%。
结论
结果表明, 14%粗蛋白水平组外源添加合成氨基酸并不能促进氨基酸转运载体增加氨基酸的吸收来满足断奶仔猪的生长性能。然而, 17%粗蛋白水平组可以减少氮排放到环境中,同时又能满足此阶段断奶仔猪的生长发育。因此, 17%粗蛋白水平组对这个阶段断奶仔猪最合适。
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
It has been well documented that low crude protein (CP), amino acid (AA)-supplemented diets may reduce feed costs and nitrogen excretion (Kerr et al., 2003; Zarate et al., 2003; Fairbrother et al., 2005; Deng et al., 2007a; 2007b; Lallès et al., 2007; Hou et al., 2008; Kang et al., 2008; Yue and Qiao, 2008; Deng et al., 2009; Opapeju et al., 2009; Gallo et al., 2014; Gloaguen et al., 2014; Liu et al., 2014; Recktenwald et al., 2014). Therefore, the CP level in the diet of weaned pigs has been suggested to be one of the main factors that affect their growth performance (Gallo et al., 2014), feed efficiency, and gastrointestinal health (Yin and Tan, 2010; Acciaioli et al., 2011). An increase in the entry of nutrients (e.g. dipeptides, tripeptides, arginine, lysine, and histidine) from the lumen of the small intestine into the enterocyte can enhance tissue protein synthesis and improve the efficiency of utilization of dietary nutrients, and it can selectively modulate the gene expression of AA transporters in intestinal cells (Wang et al., 2009). Some studies have reported that improving the efficiency of protein use and maintaining performance can be achieved by reducing the CP content of the diet while ensuring the AA balance (Kerr and Easter, 1995; Le Bellego and Noblet, 2002; Kerr et al., 2003; Opapeju et al., 2008; Gloaguen et al., 2014). However, Nyachoti et al. (2006) demonstrated that piglet performance may be impaired when dietary CP levels are reduced from 230 to 190 or 170 g/kg, as the AA deficiency may change the efficiency of AA absorption and transportation (Tuitoek et al., 1997; Tan et al., 2009; 2011; Gallo et al., 2014). The present study investigated the effects of reducing dietary CP while supplementing with essential AA on the gene expression of AA transporters in weaned piglets.
2 Materials and methods
2.1 Animals and experimental treatment
The experimental protocol was approved by the Protocol Management and Review Committee of the Institute of Subtropical Agriculture, Chinese Academy of Sciences (CAS). Pigs were cared for according to the guidelines of the Institute of Subtropical Agriculture on Animal Care, CAS.
Eighteen piglets (Duroc×Landrace×Yorkshire, female), from the experimental field of the animal observation station in southern center of China, were weaned at 28 d of age, but the experiment started after a 7-d adaptation period when the piglets were weighed to determine an initial mean body weight (BW; mean±standard deviation (SD), (9.57±0.64) kg), and randomly assigned to three different groups (six pigs per group). Each of the groups was fed diets with different levels of CP (14%, 17%, and 20%), respectively. Their ingredients are shown in Tables 1 and 2. The dietary treatment met the National Research Council (NRC; 2012) nutrient specifications for 11 to 20 kg BW pigs. All of the animals were housed individually in cages, and had free access to feed and drinking water at all times throughout the experimental period.
2.2 Relative quantification of mRNA expression of AA transporters
Primers for the selected genes (Table 3) were designed using Oligo 6.0 software (Molecular Biology Insights, CO, USA). After the quantity of complementary DNA (cDNA) was determined by a NanoDrop® ND-1000 (NanoDrop Technologies, Rockland, DE, USA), real-time quantitative polymerase chain reaction (PCR) analyses were performed on an ABI 7900HT Fast Real-Time PCR System (Applied Biosystems, CA, USA) with a total volume of 10 μl containing 5 ng of cDNA, 5 μl SYBR Green mix, 0.2 μl ROX Reference Dye (50×), and 0.2 μl each of the forward and reverse primers (Yao et al., 2012). The following protocol was used: (1) pre-denaturation (30 s at 95 °C); (2) amplification and quantification, repeated 40 cycles (5 s at 95 °C, 30 s at 60 °C); and (3) a melting curve program (extension at 72 °C). β-Actin was used as an internal reference gene to normalize target gene transcript levels. The efficiency of the real-time reverse-transcription PCR was determined by the amplification of a dilution series of cDNA according to the equation 10(-1/slope). Target mRNA and β-actin mRNA were amplified with comparable efficiencies (Wang et al., 2009; He et al., 2013). Negative controls, in which cDNA was replaced by water, were also tested (Liu et al., 2012).
2.3 Statistical analysis
All data were subjected to a one-way analysis of variance (ANOVA; SAS Version 9.2) and data were presented as mean±standard error of the mean (SEM). Differences with P values of <0.05 were considered to be statistically significant.
3 Results
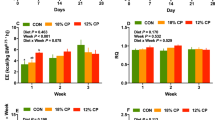
3.1 Growth performance of piglets fed diets with different levels of CP (internal data)
The growth performance of weaned piglets fed diets with different CP levels is unpublished data. After the animals were fed different diets for 45 d, significant differences were noted in the average daily gain (ADG) among the three groups (P<0.01), and the 20% CP group had a significantly higher ADG than the other groups (P<0.01). Average daily feed intake (ADFI) showed the same trend as ADG: the 20% CP group had the highest ADFI and ADG, and 14% CP group had the lowest ADFI and ADG. However, there was no significant difference in ADFI between the 17% and 20% CP groups. While the 20% CP group had the lowest feed to gain (F/G) ratio, there were no significant differences between the 14% and 17% CP groups.
3.2 mRNA expression of AA transporters
Changes in the abundance of ASCT2, EAAC1, B0+AT, y+LAT1, 4F2hc, and ATB0 mRNA in the jejunum of the piglets are shown in Table 4. While the abundance of ASCT2, EAAC1, B0+AT, and ATB0 mRNA significantly differed among the three groups (P<0.05), there were no differences in the expression of y+LAT1 and 4F2hc mRNA (P>0.05). Compared with the values in the 14% CP group, the expression of ASCT2, 4F2hc, and ATB0 mRNA in the jejunum was increased by 23.00%, 12.00%, 6.00% and 48.00%, 47.00%, 56.00% in the 17% and 20% CP groups, respectively. The 17% CP group showed the lowest expression of EAAC1 mRNA, and the highest expression of B0+AT mRNA. Furthermore, while the expression of y+LAT1 in the 17% and 20% CP groups was decreased by 13.00% and 8.00%, respectively, compared with that in the 14% CP group, there were no significant differences among the three groups (P>0.05).
4 Discussion
AAs regulate key metabolic pathways that are crucial for the maintenance, health, and growth of animals (Wu, 1998; Hu et al., 2008). The absorption of AA requires many transporter systems that differ with respect to their substrate specificity and driving force (Wang et al., 2009). It has been reported that some genes (such as ASCT2, EAAC1, B0+AT, y+LAT1, 4F2hc, and ATB0) that are involved in the control of growth or AA metabolism are regulated by AA availability (Wu, 1998; Lallès et al., 2007; Zhang et al., 2012; Abdulhussein and Wallace, 2014). In our study, we found that, with an increase in the dietary CP level, the expression of AA transporter genes (ASCT2, EAAC1, B0+AT, 4F2hc, and ATB0) tended to increase, while an opposite trend was seen related to the abundance of y+LAT1 mRNA. A possible reason was that y+LAT1 and EAAC1 have differential expression in our study because the diets with different CP levels related with absorption in piglets. The transport of AA in the small intestine is critical for the supply of AA to all tissues and the homeostasis of serum AA levels (Zhang et al., 2012). Since dietary CP can provide an ideal AA level, the genes of AA transporters that regulate protein metabolism in piglets should be more activated. The present results indicate that the expression of ASCT2, EAAC1, B0+AT, and ATB0 mRNA in the jejunum was influenced by dietary CP levels. Our results regarding the expression of y+LAT1 mRNA were similar to those of He et al. (2013). Under conditions of nutritional stress, the expression of some AA transporters (y+LAT1) may be increased to maintain normal growth of animals under adverse conditions (He et al., 2013). Skalli et al. (2014) suggest that dietary protein levels regulate the expression of AA transporter genes by a complex regulatory system, which may also affect the energy balance and endocrine system. AAs and hormones can play an important role in the regulation of gene expression. Overall, the use of low-CP (14%) AA-supplemented diets in piglets may decrease the transport capacity of AA in the small intestine. However, the absorption and transport of AA is a complex physiological process that is influenced by many factors (Wang et al., 2009; He et al., 2013; Zhang et al., 2013; Chen et al., 2014), including the dietary CP level and the AA balance. Thus, the transport of these nutrients is a key regulatory step in the use of dietary protein by weaned piglets. An excess decrease in dietary CP with AA supplementation resulted in significant reductions in the expression of AA transporter genes in the jejunum. This result suggests that there is an appropriate range within which CP in the diet may be reduced, and any further decrease may dramatically reduce the efficiency of energy and AA utilization in the small intestine (Jensen, 1998). This understanding may also make it possible to increase productivity and reduce the impact of livestock on the environment (Franklin et al., 2002). While this finding has important practical implications, further research will be required to elucidate the underlying mechanisms.
Compliance with ethics guidelines
Li WU, Liu-qin HE, Zhi-jie CUI, Gang LIU, Kang YAO, Fei WU, Jun LI, and Tie-jun LI declare that they have no conflict of interest.
All institutional and national guidelines for the care and use of laboratory animals were followed.
References
Abdulhussein, A.A., Wallace, H.M., 2014. Polyamines andmembrane transporters. Amino Acids, 46(3):655–660.[doi:10.1007/s00726-013-1553-6]
Acciaioli, A., Sirtori, F., Pianaccioli, L., et al., 2011. Comparisonof total tract digestibility and nitrogen balancebetween Cinta Senese and Large White pigs fed on differentlevels of dietary crude protein. Anim. Feed Sci. Tech., 169(1-2):134–139. [doi:10.1016/j.anifeedsci.2011.05.009]
Chen, G., Zhang, J., Zhang, Y.Z., et al., 2014. Oral MSGadministration alters hepatic expression of genes for lipidand nitrogen metabolism in suckling piglets. Amino Acids,46(1):245–250. [doi:10.1007/s00726-013-1615-9]
Deng, D., Li, A.K., Chu, W.Y., et al., 2007a. Growth performanceand metabolic responses in barrows fed lowproteindiets supplemented with essential amino acids. Livest. Sci., 109(1-3):224–227. [doi:10.1016/j.livsci.2007.01.104]
Deng, D., Huang, R.L., Li, T.J., et al., 2007b. Nitrogen balancein barrows fed low-protein diets supplemented with essentialamino acids. Livest. Sci., 109(1-3):220–223.[doi:10.1016/j.livsci.2007.01.122]
Deng, D., Yao, K., Chu, W.Y., et al., 2009. Impaired translationinitiation activation and reduced protein synthesis inweaned piglets fed a low-protein diet. J. Nutr. Biochem.,20(7):544–552. [doi:10.1016/j.jnutbio.2008.05.014]
Fairbrother, J.M., Nadeau, E., Gyles, C.L., 2005. Escherichiacoli in postweaning diarrhea in pigs: an update on bacterialtypes, pathogenesis, and prevention strategies. Anim. Health Res. Rev., 6(1):17–39. [doi:10.1079/AHR2005105]
Franklin, M.A., Mathew, A.G., Vickers, J.R., et al., 2002.Characterization of microbial populations and volatilefatty acid concentrations in the jejunum, ileum, and cecumof pigs weaned at 17 vs 24 days of age. J. Anim. Sci.,80(11):2904–2910.
Gallo, L., Monta, G.D., Carraro, L., et al., 2014. Growth performanceof heavy pigs fed restrictively diets with decreasingcrude protein and indispensable amino acidscontent. Livest. Sci., 161:130–138. [doi:10.1016/j.livsci.2013.12.027]
Gloaguen, M., Le Floc'h, N., Corrent, E., et al., 2014. The useof free amino acids allows formulating very low crudeprotein diets for piglets. J. Anim. Sci., 92(2):637–644.[doi:10.2527/jas.2013-6514]
He, L.Q., Yang, H.S., Hou, Y.Q., et al., 2013. Effects of dietaryL-lysine intake on the intestinal mucosa and expressionof cat genes in weaned piglets. Amino Acids, 45(2):383–391. [doi:10.1007/s00726-013-1514-0]
Hou, Z.P., Yin, Y.L., Huang, R.L., et al., 2008. Rice proteinconcentrate partially replaces dried whey in the diet forearly-weaned piglets and improves their growth performance. J. Sci. Food Agric., 88(7):1187–1193. [doi:10.1002/jsfa.3196]
Hu, C.A., Khalil, S., Zhaorigetu, S., et al., 2008. HumanΔ1-pyrroline-5-carboxylate synthase: function and regulation. Amino Acids, 35(4):665–672. [doi:10.1007/s00726-008-0075-0]
Jensen, B.B., 1998. The impact of feed additives on the microbialecology of the gut in young pigs. J. Anim. FeedSci., 7:45–64.
Kang, P., Yin, Y.L., Ruan, Z., et al., 2008. Effect of replacementof lactose with partially hydrolysed rice syrup onsmall intestine development in weaned pigs from 7 to 21days. J. Sci. Food Agric., 88(11):1932–1938. [doi:10.1002/jsfa.3298]
Kerr, B.J., Easter, R.A., 1995. Effects of feeding reducedprotein, amino acid-supplemented diets on nitrogen andenergy-balance in grower pigs. J. Anim. Sci., 73(10):3000–3008.
Kerr, B.J., Southern, L.L., Bidner, T.D., et al., 2003. Influenceof dietary protein level, amino acid supplementation, anddietary energy levels on growing-finishing pig performanceand carcass composition. J. Anim. Sci., 81(12):3075–3087.
Lallès, J.P., Bosi, P., Smidt, H., et al., 2007. Nutritional management of gut health in pigs around weaning. Proc. Nutr. Soc., 66(2):260–268. [doi:10.1017/S0029665107005484]
Le Bellego, L., Noblet, J., 2002. Performance and utilization ofdietary energy and amino acids in piglets fed low proteindiets. Livest. Prod. Sci., 76(1-2):45–58. [doi:10.1016/S0301-6226(02)00008-8]
Liu, M.B., Li, X.L., Liu, Y., et al., 2014. Detection of crudeprotein, crude starch, and amylose for rice by hyperspectral reflectance. Spectrosc. Lett., 47(2):101–106. [doi:10.1080/00387010.2013.779283]
Liu, X.D., Wu, X., Yin, Y.L., et al., 2012. Effects of dietaryL-arginine or N-carbamylglutamate supplementationduring late gestation of sows on the miR-15b/16,miR-221/222, VEGFA and eNOS expression in umbilicalvein. Amino Acids, 42(6):2111–2119. [doi:10.1007/s00726-011-0948-5]
Nyachoti, C.M., Omogbenigun, F.O., Rademacher, M., et al.,2006. Performance responses and indicators of gastrointestinal health in early-weaned pigs fed low-proteinamino acid-supplemented diets. J. Anim. Sci., 84(1):125–134.
Opapeju, F.O., Rademacher, M., Blank, G., et al., 2008. Effectof low-protein amino acid-supplemented diets on thegrowth performance, gut morphology, organ weights anddigesta characteristics of weaned pigs. Animal, 2(10):1457–1464. [doi:10.1017/S175173110800270X]
Opapeju, F.O., Rademacher, M., Nyachoti, C.M., 2009. Effectof dietary crude protein level on jejunal brush borderenzyme activities in weaned pigs. Arch. Anim. Nutr.,63(6):455–466. [doi:10.1080/17450390903299133]
Recktenwald, E.B., Ross, D.A., Fessenden, S.W., et al., 2014.Urea-N recycling in lactating dairy cows fed diets with 2different levels of dietary crude protein and starch with orwithout monensin. J. Dairy Sci., 97(3):1611–1622.[doi:10.3168/jds.2013-7162]
Skalli, A., Zambonino-Infante, J.L., Kotzamanis, Y., et al.,2014. Peptide molecular weight, distribution of solubleprotein fraction affects growth performance and quality inEuropean sea bass (Dicentracrchus labrax) larvae. Aquacul. Nutr., 20(2):118–131. [doi:10.1111/anu.12058]
Tan, B., Yin, Y., Liu, Z., et al., 2009. Dietary L-arginine supplementation increases muscle gain and reduces body fatmass in growing-finishing pigs. Amino Acids, 37(1):169–175. [doi:10.1007/s00726-008-0148-0]
Tan, B., Yin, Y., Liu, Z., et al., 2011. Dietary L-arginine supplementation differentially regulates expression of lipidmetabolicgenes in porcine adipose tissue and skeletalmuscle. J. Nutr. Biochem., 22(5):441–445. [doi:10.1016/j.jnutbio.2010.03.012]
Tuitoek, K., Young, L.G., de Lange, C.F., et al., 1997. Theeffect of reducing excess dietary amino acids on growing finishingpig performance: an elevation of the ideal proteinconcept. J. Anim. Sci., 75(6):1575–1583.
Wang, W., Gu, W., Tang, X., et al., 2009. Molecular cloning,tissue distribution and ontogenetic expression of theamino acid transporter b0,+ cDNA in the small intestine ofTibetan suckling piglets. Comp. Biochem. Physiol. B Biochem. Mol. Biol., 154(1):157–164. [doi:10.1016/j.cbpb.2009.05.014]
Wu, G., 1998. Intestinal mucosal amino acid catabolism. J. Nutr., 128(8):1249–1252.
Yao, K., Yin, Y., Li, X., et al., 2012. Alpha-ketoglutarate inhibits glutamine degradation and enhances proteinsynthesis in intestinal porcine epithelial cells. Amino Acids,42(6):2491–2500. [doi:10.1007/s00726-011-1060-6]
Yin, Y.L., Tan, B., 2010. Manipulation of dietary nitrogen,amino acids and phosphorus to reduce environmentalimpact of swine production and enhance animal health. J. Food Agric. Environ., 8(3-4):447-462.
Yue, L.Y., Qiao, S.Y., 2008. Effects of low-protein diets supplemented with crystalline amino acids on performanceand intestinal development in piglets over the first 2weeks after weaning. Livest. Sci., 115(2-3):144–152.[doi:10.1016/j.livsci.2007.06.018]
Zarate, A.J., Moran, E.T., Burnham, D.J., 2003. Reducingcrude protein and increasing limiting essential amino acidlevels with summer-reared, slow- and fast-feathering broilers. J. Appl. Poultry Res., 12(2):160–168. [doi:10.1093/japr/12.2.160]
Zhang, J., Yin, Y.L., He, Q.H., et al., 2012. Effects of MSG supplementation on free amino acids in plasma of growingfinishing pigs. J. Food Agric. Environ., 10(3-4):600–605.
Zhang, J., Yin, Y.L., Shu, X.G., et al., 2013. Oral administrationof MSG increases expression of glutamate receptorsand transporters in the gastrointestinal tract of youngpiglets. Amino Acids, 45(5):1169–1177. [doi:10.1007/s00726-013-1573-2]
Acknowledgements
We apologize to colleagues whose work was not cited here due to our oversight.
Author information
Authors and Affiliations
Corresponding author
Additional information
The two authors contributed equally to this work
Project supported by the National Basic Research Program (973) of China (No. 2013CB127301), the Natural Science Foundation of Hunan Province, China (No. 12JJ2014), and the National Natural Science Foundation of China (No. 31402088)
ORCID: Peng LIAO, http://orcid.org/0000-0001-5740-7272
Rights and permissions
About this article
Cite this article
Wu, L., He, Lq., Cui, Zj. et al. Effects of reducing dietary protein on the expression of nutrition sensing genes (amino acid transporters) in weaned piglets. J. Zhejiang Univ. Sci. B 16, 496–502 (2015). https://doi.org/10.1631/jzus.B1400259
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.B1400259