Abstract
A membrane bioreactor (MBR) was established for treating harbor oily wastewater. It showed good removal performance for chemical oxygen demand (COD), oil content, suspended solids (SS), and other pollutants. However, serious membrane fouling occurred. It was recognized that the extracellular polymeric substances (EPS) accumulated on the membrane surface, especially the proteins, were of great importance for the transmembrane pressure (TMP) increment and membrane fouling. The MBR was optimized via improving aeration rate and reducing the ratio of Ar/Ad (Ar and Ad are the cross-sectional areas of the riser and the downcomer of the MBR). The increasing rate of TMP was slowed, indicating that the optimization strategy could effectively mitigate membrane fouling. Microbial community evolution was monitored and analyzed by polymerase chain reaction-denaturing gradient gel electrophoresis (PCR-DGGE), cloning, and sequencing of 16S ribosomal ribonucleic acid (rRNA) fragments. Results revealed that low community shift occurred during the whole operational period. Geobacter sp. and Rhodocyclales sp., which have also been identified by other studies in a petroleum refinery wastewater MBR or an infiltration basin receiving highway runoff, dominated in the MBR system throughout. Comamonas sp. was thought to accommodate the lower aeration rate in this study, while Rhodocyclales sp. preferred the higher aeration rate. In addition, during the operational time under high organic loading rate, a few species were present in abundance, and may have been responsible for the good removal performance at this time.
中文概要
目 的
采用膜生物反应器处理港口含油废水。考察运行过程中膜污染行为特征, 分析引起膜污染的关键影响因素, 研究减弱膜污染的途径与方法; 考察反应器内群落结构演变, 分离鉴定优势菌种, 揭示运行条件和进水水质等宏观环境与微生物微环境之间的对应关系。
创新点
1. 分析出膜表面累积的胞外蛋白是膜污染严重和过膜压力增大的关键诱因; 2. 基于Yusuf Chisti和Murray Moo Young 提出的气提液体上升流速模型, 结合胞外蛋白变化趋势, 提出降低过膜压力上升速度和缓解膜污染进程的有效措施; 3. 针对处理港口含油废水的生物系统进行种群结构研究及优势菌属的分离与鉴定。
方 法
1. 借助环境扫描电子显微镜(SEM)、能量色散X射线光谱仪(EDX)和傅里叶红外光谱仪(FTIR)等分析手段表征膜污染物的主要成分; 2. 基于Yusuf Chisti 和Murray Moo Young 提出的气提液体上升流速模型, 提出缓解膜污染进程的措施, 并验证所提措施的有效性(图3 和9); 3. 通过巢式PCR-DGGE 技术对处理港口含油废水的膜生物反应器不同运行阶段的微生物群落结构进行研究, 并采用克隆技术对优势微生物进行菌种鉴定(图10 和表3)。
结 论
1. 采用一体化厌氧/好氧-膜生物反应器来处理港口含油废水的优点是对有机污染物和油类等污染物的去除效率高, 但在其运行过程中膜污染严重。2. 分析发现胞外聚合物是引起膜污染物的主要因素(图3∼6); 组分分析进一步表明膜表面累积的胞外蛋白是膜污染严重和过膜压力增大的关键诱因(图7∼9)。3. 通过增大反应器曝气量, 调整反应器结构参数可有效降低过膜压力上升速度, 缓解膜污染进程。4. 反应器内细菌群落结构平稳。5. 克隆测序结果表明: 膜生物反应器内丛毛单胞菌属在低曝气环境下繁殖能力强, 且可能是引起膜污染的特征微生物; 红环菌科细菌更适应高曝气条件, 是降解含有油类污染物的特征细菌。
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
A membrane bioreactor (MBR) is able to provide a small footprint, perfect effluent, high biodegradation efficiency, and little excess sludge (Xia et al., 2010). Thus, this technology has attracted increased interest and been widely applied in wastewater treatment and reuse. However, inevitable membrane fouling, consequently leading to a decline in membrane permeability and an increase in operational cost, is still the major obstacle that hampers further development of MBR technology.
Extensive research has been carried out to better understand the fouling mechanism. It has been generally accepted that the membrane fouling is due to particle deposition on membrane surface and/or colloidal particle deposition inside membrane pores (Cosenza et al., 2013). These processes are mainly affected by membrane materials, feedwater characteristics, sludge characteristics, and operating conditions (Judd, 2004; Meng et al., 2009; Kim et al., 2010; Cosenza et al., 2013; Mannina and Cosenza, 2013; Estrada-Arriaga, et al., 2015). Among them, sludge characteristics are considered to be directly affected by the feedwater characteristics and operating conditions (i.e., aeration, hydraulic conditions, sludge retention time (SRT), hydraulic retention time (HRT), organic loading rate, and food microorganism ratio (F/M)). Thus, it is assumed that sludge characteristics, including viscosity, mixed liquor suspended solids (MLSS) concentration, amount of filamentous bacteria, extracellular polymeric substances (EPS), and soluble microbial products (SMP), serve as the vital causes for membrane fouling. More specifically, it is believed that the EPS may have a primary role in membrane fouling (Cosenza et al., 2013). However, due to the complexity of MBR systems and various wastewaters to be treated, the complex relationship of those fouling factors is still not clear. For some wastewater, a detailed understanding on how and which part of EPS has more significant influence on membrane fouling is still lacking (Delrue et al., 2011; Cosenza et al., 2013; Di Bella et al., 2015). A better elucidation, clarifying the key fouling factors, will help in defining the optimum approach to mitigate membrane fouling in an MBR system.
Bacteria are key players in biological wastewater treatments (WWTs), and thus a firm knowledge of the bacterial population dynamics is crucial to understand environmental/operational factors affecting the efficiency and stability of the biological depuration process (Reboleiro-Rivas et al., 2016). In recent years, DNA-based molecular techniques, like denaturing gradient gel electrophoresis (DGGE) of polymerase chain reaction (PCR) amplified 16S ribosomal ribonucleic acid (rRNA) gene fragments, offer a valuable tool for the characterization of the bacterial population diversity in biological wastewater treatment systems (LaPara et al., 2002; Li A.J. et al., 2008; Wei et al., 2012). These techniques have been used to characterize the dynamics of microbial evolution in various wastewater treatment reactors (Li et al., 2010; Zhang et al., 2010; Aydin et al., 2015). The molecular techniques are also applied in the MBR system to study the bacterial community structures. With the PCR-DGGE technique, Xia et al. (2010) investigated the effect of organic loading on the bacterial community composition of membrane biofilms in a submerged polyvinyl chloride MBR. Ziembinska et al. (2012) monitored the ammonia-oxidizing bacterial (AOB) communities in three MBRs for two months, and found that landfill leachate can be effectively treated in an MBR with a higher sludge age for longer periods of time, and that this improvement in performance was correlated with an increase in AOB biodiversity. Tan et al. (2015) reported that the bacterial community in an MBR showed robust performance over ZnO nanoparticles exposure.
In this study, an MBR system was established for harbor oily wastewater treatment, owing to the higher biomass concentration retained by membrane filtration and longer SRT. A possible relationship between fouling and EPS was evaluated by means of scanning electron microscope (SEM) images, energy dispersive X-ray (EDX) analysis and Fourier transform infrared spectroscopy (FTIR) spectra, as well as qualitative analysis. The content and composition of EPS were also examined when the MBR system was optimized according to a model proposed by Chisti and Moo-Young (1993) and Li et al. (2000), so as to further verify the role of EPS in membrane fouling and the function of the optimization strategy. The microbial population dynamic during the whole operation period was monitored, to determine what kinds of bacteria are responsible for the pollutant removal performance and membrane fouling during long-term operation.
2 Materials and methods
2.1 Experimental set-up
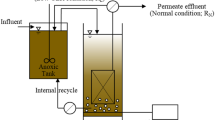
Fig. 1 shows the schematic diagram of the experiment. A pilot-scale anoxic/oxic-MBR (A/O-MBR) with 500 L working volume was used. Two hollow flat-plat polyvinylidene fluoride (PVDF) fiber membrane modules (Motian, China) were submerged in the oxic zone. The nominal pore size of the membrane module was 0.2 µm, and the filtration area of each module was 1 m2. The MBR was operated at a constant permeate flux of 10 L/(m2·h). The feed pump and suction pump were controlled by computer, and an intermittent suction effluent mode of 8 min-on/2 min-off was applied. Fine air bubbles were continuously supplied for aeration through microporous air diffusers at the bottom of the oxic zone. The air flow rate was controlled by a rotermeter, with an air flow rate of 0.1–1.0 m3/h. The membrane module did not experience hydrodynamic and chemical cleaning before the end of the experiment.
Floc-like sludge from an oxidation ditch of a local harbor oily wastewater treatment plant was added to the MBR as inocula, resulting in an initial MLSS of 5.0 g/L. The SRT was maintained at 50 d.
The system was fed with real harbor oily wastewater coming from a local harbor oily wastewater treatment plant in Tianjin Harbor, China. Influent of A/O-MBR had been pre-treated by coagulation-sedimentation. Composition of the influent fed to the reactor is summarized in Table 1.
2.2 Operational strategy
It is known that the improvement of the hydrodynamic condition in MBR systems, including cross-flow velocity (CFV) along the membrane surface, can effectively mitigate membrane fouling (Bacchin and Espinasse, 2005). For this purpose, CFV was usually kept around 1 m/s in MBRs treating municipal wastewater (Li J. et al., 2008). Therefore, it could be inferred that for a sufficiently long membrane life, a higher CFV should be obtained in MBRs treating oily wastewater.
The CFV of the submerged MBR was equated with the liquid circulation velocity ULr, which could be calculated following the procedure reported by Chisti and Moo-Young (1993) and Li et al. (2000):
where ULr is the superficial liquid velocity in the riser of MBR, m/s; g is the gravitational acceleration, m2/s; hD is the gas-liquid dispersion height in the MBR, m; Ar and Ad are the cross-sectional areas of the riser and the downcomer, respectively, m2; εr and εd are the fractional-gas-holdup values of the riser and the downcomer, respectively; KB is the frictional-loss coefficient for the bottom zones.
It could be seen that a higher aeration rate and a lower ratio of Ar/Ad are in favor of higher superficial liquid velocity in the riser of MBR, or CFV. However, the higher aeration rate would also result in higher operational costs, and the lower ratio of Ar/Ad would lead to larger floor area. Thus, proper parameters should be optimized.
The experiment lasted for 320 d in total, including the first stage, suspended stage, and second stage. During the first stage, the aeration rate of the MBR was fixed at 0.40 m3/h, and the ratio of Ar/Ad was 0.21, with a CFV of 0.78 m/s. To gain a higher CFV to mitigate membrane fouling, both the aeration rate and Ar/Ad were optimized in the suspended stage according to the models proposed by Chisti and Moo-Young (1993) and Li et al. (2000). Eventually, the aeration rate of the MBR increased to 0.45 m3/h during the second stage. Ar/Ad decreased to 0.18, with a CFV of 1.13 m/s. Meanwhile, the membrane modules were replaced by new ones. Fresh floc-like sludge was added into the MBR, with the MLSS of 5 g/L, just as at the beginning of the first stage.
2.3 Analytical methods
COD, oil content, Cl− content, SS, and MLSS were measured regularly according to the standard methods (APHA, 1998). pH was determined by a pH meter (Hanna HI9145, Italy). Heavy metal was analyzed by an inductively coupled plasma mass spectrometer (PerkinElmer SCIEX-ELAN DRC-e, USA) and a mercury vapourmeter (Milestone DMA80, Italy). Monocyclic aromatic hydrocarbons (MAH) were tested by a gas chromatography-mass spectrometer (Agilent 7890B-5977A, USA). Polynuclear aromatic hydrocarbons (PAH) were detected by a gas chromatography-mass spectrometer (Agilent 7890A/5975C, USA).
The morphology and structure of the membrane surface were observed using an environmental SEM (Phillips XL30, the Netherlands). EDX analysis was accomplished by an X-ray energy spectrometer (Genesis XM2, USA). A spot size of about 70 nm was used to record EDX spectra and stoichiometric ratios were calculated from atomic percentages given by EDX spectra. EDX calibration standards were run to obtain quantitative analyses of major elements on the membrane surface (Zhang et al., 2008b). The deposition of biopolymers on the membrane surface was characterized by Fourier transform infrared spectroscopy (Nexus™, USA). Sludge morphology was observed under an optical microscope (HIROX KH-7700, USA). Specific resistance to filtration (SRF) of sludge was tested according the method described by Zhang (2000).
The EPS of the mixed liquor in the membrane reactor was extracted using the method described by Wang et al. (2010). The EPS of the cake layer attached to the membrane was extracted according the steps described by Zhang et al. (2008b). The polysaccharides content was determined by the phenol-sulfuric acid method (Gerhardt et al., 1994). The protein content was determined by the revised Lowry method (Frølund et al., 1996).
2.4 DNA extraction, PCR amplification, and DGGE
Activated sludge samples were collected from the MBR system. The sludge from approximately 8 mg of MLSS was transferred into a 1.5-ml Eppendorf tube and then centrifuged at 14 000g for 10 min. The supernatant was then removed, and the pellet was stored at −70 °C until further analysis (Zhang et al., 2010).
Genomic DNA of the biomass in sludge and granule samples was extracted using an E.Z.N.A.™ Soil DNA Kit (OMEGA D5625-01, USA). Subsequently, the 16S rRNA gene was amplified by a nested PCR approach. After the first round of amplification, a nested PCR was performed on the PCR products with a second primer pair allowing the amplification of an internal fragment. These primers and PCR conditions are summarized in Table 2. All PCR amplicons were examined by electrophoresis in 1.5% (weight/volume) agarose with ethidium bromide staining to confirm the product size. PCR amplification was performed with a GoTaq Master Mix (Promega, USA).
The PCR-amplified DNA products were separated by DGGE on polyacrylamide gels (8%, 37.5:1 acrylamide-bisacrylamide) with a linear gradient of 35%–55% denaturant (100% denaturant=7 mol/L urea plus 40% formamide). Gels were run for 7 h at 150 V in 1× TAE buffer maintained at 60 °C. Denaturing gradient gels were poured and run using the DGGE-2001 System (C.B.S. Scientific, Del Mar, USA). Gels were silver-stained, air dried, and scanned following the prescriptions described elsewhere (Sanguinetti et al., 1994).
2.5 Cloning and sequencing analysis
Selected DGGE bands were excised and dissolved in 50 µl Milli-Q water overnight, at 4 °C. DNA was recovered from the gel by freeze-thawing three times. Using the primer set F357/R518 without GC-clamp, 3 µl of these DNA band samples were reamplified by the same PCR procedure described previously. The PCR products were cloned using the PGEMTM-T Easy vector system (Promega, Madison, USA). The positive colonies were amplified with F357/R518. PCR amplicons were submitted for sequencing using ABI 3730 capillary sequencers (PE Applied Biosystems, Invitrogen, China) (Zhang et al., 2010). Sequence data were analyzed in comparison with the 16S rRNA sequences in the GenBank by BLASTN search (National Centre for Biotechnology Information, USA) for species identification.
3 Results and discussion
3.1 MBR performance
Table 1 and Fig. 2 show the changes and removal efficiencies of main pollutants fed to the system. During the first stage, the organic loading rate (OLR) of the MBR was 0.72–1.25 kg COD/(m3·d). With the acclimatization of microorganisms to influent compounds, COD removal efficiency (RE) gradually increased and stabilized around 90% afterwards.
In the second stage, COD removal efficiency was still about 90%, except in the middle phase with higher shock load. During days 200–230, the OLR increased sharply to 1.63–2.62 kg COD/(m3·d) due to higher influent COD content. The COD removal efficiency decreased, but it resumed to a stable range of 88%–95% in a short period. When the influent COD was (427.6±155.6) mg/L, the effluent concentration was stationary at (48.1±31.4) mg/L.
There were a few pretreatment steps before the MBR for the oil removal but the influent oil content of MBR was still as high as (8.8±6.3) mg/L. In light of the entrapment role of the membrane module, the bioreactor became a relatively closed system as far as the microbial metabolism was concerned (Wang et al., 2008). Therefore, the oil content was not detected after the MBR treating.
The MBR also exhibited good removal performance for recalcitrant compounds (MAH and PAH), SS, and other elements in the list of priority pollutants of USEPA. Ammonium was not a major concern because the average influent concentration of the MBR system was fairly low.
Fig. 2b shows the changes of biomass concentration in the MBR. It increased from 5000 mg/L to 9159 mg/L during the first stage, and from 4939 mg/L to 11 730 mg/L in the second stage.
3.2 Membrane fouling behavior
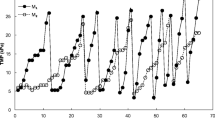
Membrane fouling was usually monitored using transmembrane pressure (TMP) under constant flux mode. The evolution of permeate flux and TMP are depicted in Fig. 3. The flux was fixed at around 10 L/(m2·h) via a constant flow suction pump. The TMP increased sharply, and reached 30 kPa after 78 d.
Specific resistance to filtration (SRF) of sludge is widely used to foresee performances of full-scale filters and to compare the sludge behavior from different plants against filtration processes. It represents the resistance to filtration of a theoretical sludge panel having unitary weight in dry solids per unit of filtering surface (Pontoni et al., 2015). The evolution of SRF in this study is shown in Fig. 3. It did not significantly change within 25 d. The rapid increase of SRF was completed between the 30th day and 70th day, and it was up to 4.18×1013 m/kg on the 70th day, meaning a higher aptitude of sludge to be dehydrated via filtration, and higher energy to be consumed for ensuring the constant flux (Mannina et al., 2016b). In addition, the increase of SRF was closely associated with the TMP augmentation, suggesting that sludge characteristics would be the major pressure exerted on MBR fouling.
In general, membrane fouling is attributed to initial pore blocking followed by cake formation (Meng et al., 2007). The sludge layer was thought to be the main cause of membrane fouling and the increment of transmembrane resistance (Mannina et al., 2016a). The SEM images reveal the outer morphology of foul membrane surface (Fig. 4). It can be seen that multiple sludges were tightly linked together by a cement-like substance forming a compact structure.
EDX and FTIR analyses were performed to confirm the major components of the fouling layer (Figs. 5 and 6). In comparison with the original chemical composition, multi-valent metal elements, such as Zn, Mg, Al, and Ca, were detected. It was well known that EPS would be produced during the microbial metabolism, and prefers to bind multi-valent metals due to the formation of stable complexes (Rudd et al., 1984).
Additionally, the spectrum showed peaks mainly between 3500 cm−1 and 600 cm−1. The broad peak at 3284.25 cm−1 is due to the stretching vibration of the O-H bond in hydroxyl functional groups, and the sharp peak at 2921.17 cm−1 is due to stretching of C-H bond (Meng et al., 2007). The peak at 1632.63 cm−1 is sensitive to C=O stretching vibration of the protein amide I. The peak at 1543.72 cm−1 is assigned to the out-of-phase combination of the NH in-plane bend and the CN stretching vibration of the protein amide II (Barth and Zscherp, 2002). The peaks in the spectral range from 1250 cm−1 to 1000 cm−1 are due to the contribution of nucleic acid phosphodiester groups and phospholipids, as well as to the C-O absorption of carbohydrates (Ami et al., 2014). In Fig. 6 the FTIR spectra of membrane foulants show that the major components of the foulants included proteins and polysaccharide materials. Proteins and carbohydrates are the dominant components typically found in EPS.
Taken together with the analysis above, it could be inferred that EPS would be the major component of the fouling layer, and a probable index for membrane fouling.
3.3 Relationship between EPS content and membrane fouling behavior
The qualitative composition of the EPS was determined to investigate the deposition behavior of EPS on the membrane surface, and further membrane fouling, so as to get further insights into the assumed relationship between EPS and membrane fouling behavior. Fig. 7 shows the EPS composition of the mixed liquor in the membrane reactor. The original concentrations of proteins (PN) and polysaccharides (PS) were 31.9 mg/g VSS and 7.9 mg/g VSS, respectively. The polysaccharides concentration did not show significant change subsequently, and stabilized at 9.2–11.0 mg/g VSS. Rapid increase of proteins concentration had occurred, and it reached around 98.5 mg/g VSS on the 58th day. The concentration ratio of PN/PS showed similar trends to that of protein concentration.
Taken in conjunction with the data shown in Figs. 3 and 7, it can be observed that protein concentration of mixed liquor was dramatically related to SRF. The exponential relationship is illustrated in Fig. 8 by a positive correlation coefficient of 0.934 (data of the second stage were used here, and would be discussed later). These results clearly implied that proteins, the major component of EPS of the mixed liquor, would stimulate the increase of sludge SRF in a very significant way.
The EPS composition of the cake layer attached to the membrane is depicted in Fig. 9. On the 42nd day, the concentrations of proteins and polysaccharides were 133.9 mg/g VSS and 38.1 mg/g VSS, respectively. These parameters increased to 157.4 mg/g VSS and 49.6 mg/g VSS on the 65th day, implying that the EPS had significantly accumulated on the membrane surface.
Based on the above discussion, it could be inferred that the EPS of the mixed liquor or accumulated on the membrane surface (mainly the proteins) were of great importance for increase of sludge SRF and TMP, and consequent membrane fouling. But which one played the effective trigger for membrane fouling still needs to be fully understood.
3.4 Optimization of the MBR system
To achieve a longer membrane life, many manufacturers of hollow fiber membrane have made strict user notes that influent oil content (in terms of N-Hexane Extract, N-HEX) should be eliminated to a level of below 3 mg/L (MOTIMO, 2006). As a matter of fact, the influent oil content of the MBR was as high as (8.8±6.3) mg/L in this study, which would aggravate the fouling step, as demonstrated by the sharp increment of TMP in Fig. 3. To weaken the effect of oil pollutant on membrane fouling, the MBR was optimized by way of increasing aeration rate and reducing Ar/Ad. The CFV increased from 0.78 m/s to 1.13 m/s. As a result, the TMP of the MBR system still increased, but slowly. The change in TMP from 0.35 kPa to 30 kPa took about 180 d (Fig. 3), over twice the time taken before the operation optimization (75 d), indicating that the optimization could effectively reduce the rate of transmembrane pressure under constant flux mode, and significantly mitigate membrane fouling.
The deposition behaviors of EPS of the mixed liquor and SRF change after adjustment are illustrated in Figs. 3 and 7. In comparison with the situation in the first stage, the increasing rate of protein content and SRF was the same, or even higher, meaning that the optimization exerted no or positive effect on EPS secretion of the mixed liquor and sludge SRF in the MBR system. This was inconsistent with the fouling behavior.
The deposition behaviors of EPS accumulated on the membrane surface exposed to the adjustment are shown in Fig. 9. Protein content increased slowly, implying that the optimization played a crucial role in slowing down the increasing rate of EPS content (mainly the proteins content) of the fouling layer. This phenomenon could explain why the CFV was improved after optimization, because EPS can be removed by the shear stress of fluid mechanics on the membrane surfaces (Ying et al., 2010).
A solid conclusion can be drawn that the EPS which accumulated on the membrane surface, especially the proteins, were of great importance for the TMP increment and membrane fouling.
3.5 Microbial population dynamics at different operational stages
Bacterial community DNA was extracted from the sludge, which was sampled once every few days. Using the DNA obtained from DNA Kit method as template, 250-bp fragments of the V3 hypervariable region of the 16S rRNA gene were obtained as expected. DGGE was carried out to determine the genetic fingerprint of these PCR amplification products, and the profiles are illustrated in Fig. 10. From the similarity diagram and cluster analysis by unweighted pair group method (UPGMA), it is noticeable that low community shift occurred during the whole operational period, and marginal changes of DGGE bands and patterns were observed at different operational stages. It can be seen that several dominant bands appeared across all biomass samples. This indicates that the species represented by these bands were predominant populations throughout the experiment.
DGGE profiles of the bacterial communities in the MBR at different operational stages
(a) DGGE profile (the predominant bands are labeled A-L, and bands a-n are dynamic species existing in partial samples); (b) Similarity diagram of sample lanes (DGGE pattern of day 232 is defined as 100%, and values along the bottom indicate the similarity coefficient with day 232); (c) Cluster analysis of DGGE by UPGMA (values in the cluster indicate the similarity coefficient with each other). Lane labels along the top or right side show sampling time (in days) from startup of the bioreactor
There were also some relatively persistent species, like band_A, which were very intense during the first stage with low aeration rate, but became depleted or not detectable during the second stage with higher aeration rate. This phenomenon indicates that the population represented by band_A accommodated the low aeration rate. Conversely, some species, like band_D, band_E, and band_F, were very intense in the sludge samples at the second stage, indicating that these species may prefer the higher aeration rate. In addition, species marked by band_B and band_H were particularly intense in lane 212. Taken together, the results shown in Fig. 3 and Table 2 suggest that the corresponding species may metabolically respond to high OLR.
3.6 Sequencing results and analysis
Nine bands were carefully excised, amplified, and sequenced to identify microbial species, and the nucleotide sequences were compared with previously identified 16S rRNA gene sequences in the GenBank database using BLASTN. The results are shown in Table 3. It can be seen that band_A was most closely related to uncultured Comamonadaceae bacterium clone PII8C, which was identified in a petroleum refinery wastewater membrane bioreactor fed with a high phenolic load (Silva et al., 2010). Previous studies reported that dominant microbial species of the membrane cake layer contained Comamonadaceae (Gao and Xin, 2014). Comamonas sp. would excrete an amount of slime-EPS and capsular-EPS, and result in severe fouling (Zhang et al., 2008a). These findings were identical to the results in this study. Band_A, representing Comamonadaceae, was intense in the sludge samples under serious membrane fouling, and died away during the second stage after operation optimization, further implying that the optimization could efficiently slow down the membrane fouling process.
Band_C was closely related to uncultured sludge bacterium clone ASB54, which was thought of as being one isolated lipolytic gene from activated sludge (Liaw et al., 2010). Band_D and Band_F had high homologies in the nucleotide sequence with uncultured Geobacteraceae bacterium clone PM5_7.0-02, which was isolated from an infiltration basin receiving highway runoff (Rotaru et al., 2012). Band_L was closely related to uncultured Rhodocyclaceae bacterium clone PM5_-0.3-05, which was also detected in the infiltration basin receiving highway runoff (Rotaru et al., 2012). Band_c was most closely related to Hydrogenophaga sp.
4 Conclusions
-
1.
An A/O-MBR was used to treat harbor oily wastewater. It revealed good removal performances for COD, oil content, monocyclic aromatic hydrocarbons, polynuclear aromatic hydrocarbons, SS, and other elements in the list of priority pollutants of USEPA. However, serious membrane fouling occurred when the TMP increased to 30 kPa under constant flux mode in a short time.
-
2.
It is reasonable to consider that the EPS content would be a probable index for membrane fouling by means of SEM images, EDX analysis, and FTIR spectra. The qualitative analysis further verified that the EPS accumulated on the membrane surface, especially the proteins, were of great importance for the TMP increment and membrane fouling.
-
3.
The MBR was optimized by way of increasing aeration rate and reducing Ar/Ad. The evolution of TMP in the MBR system, and the deposition behavior of EPS substantiated that the optimization strategy could effectively reduce the rate of TMP under constant flux mode, and significantly mitigate membrane fouling.
-
4.
Results of PCR-DGGE indicated that low community shift occurred during the whole operational period. The species marked by band_A accommodated the low aeration rate, and were predominant during the first stage, and may metabolically respond to severe fouling. Some species, like band_D, band_E, and band_F, preferred the higher aeration rate, and were very intense in the sludge samples at the second stage. Species marked by band_B and band_H may metabolically respond to high OLR.
-
5.
Sequencing analysis demonstrated that some dominant species were also identified by other researchers in a petroleum refinery wastewater membrane bioreactor, or an infiltration basin receiving highway runoff.
References
Ami, D., Posteri, R., Mereghetti, P., et al., 2014. Fourier transform infrared spectroscopy as a method to study lipid accumulation in oleaginous yeasts. Biotechnology for Biofuels, 7(1): 12. http://dx.doi.org/10.1186/1754-6834-7-12
APHA (American Public Health Association), 1998. Standard Methods for the Examination of Water and Wastewater, 20th Edition. APHA, Washington DC,USA.
Aydin, S., Shahi, A., Ozbayram, E.G., et al., 2015. Use of PCR-DGGE based molecular methods to assessment of microbial diversity during anaerobic treatment of antibiotic combinations. Bioresource Technology, 192: 735–740. http://dx.doi.org/10.1016/j.biortech.2015.05.086
Bacchin, P., Espinasse, B., 2005. Distributions of critical flux: modeling, experimental analysis and consequences for cross-flow membrane filtration. Journal of Membrane Science, 250(1–2):223–234. http://dx.doi.org/10.1016/j.memsci.2004.10.033
Barth, A., Zscherp, C., 2002. What vibrations tell us about proteins. Quarterly Reviews of Biophysics, 35(4): 369–430. http://dx.doi.org/10.1017/S0033583502003815
Chisti, Y., Moo-Young, M., 1993. Improve the performance of airlift reactors. Chemical Engineering Process, 6: 38–45.
Cosenza, A., Di Bella, G., Mannina, G., et al., 2013. The role of EPS in fouling and foaming phenomena for a membrane bioreactor. Bioresource Technology, 147: 184–192. http://dx.doi.org/10.1016/j.biortech.2013.08.026
Delrue, F., Stricker, A.E., Mietton-Peuchot, M., et al., 2011. Relationships between mixed liquor properties, operating conditions and fouling on two fullscale MBR plants. Desalination, 272(1–3):9–19. http://dx.doi.org/10.1016/j.desal.2010.12.056
Di Bella, G., Di Prima, N., Di Trapani, D., et al., 2015. Performance of membrane bioreactor (MBR) systems for the treatment of shipboard slops: assessment of hydrocarbon biodegradation and biomass activity under salinity variation. Journal of Hazardous Materials, 300: 765–778. http://dx.doi.org/10.1016/j.jhazmat.2015.08.021
Estrada-Arriaga, E.B., Nacheva, P.M., García-Sánchez, L., 2015. Effect of mixed liquor volatile suspended solids on membrane fouling during short and long-term operation of membrane bioreactor. Ingeniería y Ciencia, 11(21): 137–155. http://dx.doi.org/10.17230/ingciencia.11.21.7
Frølund, B., Palmgren, R., Keiding, K., et al., 1996. Extraction of extracellular polymers from activated sludge using a cation exchange resin. Water Research, 30(8): 1749–1758. http://dx.doi.org/10.1016/0043-1354(95)00323-1
Gao, D.W., Xin, X.D., 2014. Analysis of microbial community structure and metabolites during the MBR membrane fouling process. Journal of Harbin Institute of Technology, 46(2): 26–32 (in Chinese).
Gerhardt, P., Murray, R.G.E., Wood, W.A., et al., 1994. Methods for General and Molecular Bacteriology. American Society for Microbiology, Washington DC, USA, p.518.
Judd, S., 2004. A review of fouling of membrane bioreactors in sewage treatment. Water Science Technology, 49(2): 229–235.
Kim, H.G., Jang, H.N., Kim, H.M., et al., 2010. Effects of the sludge reduction system in MBR on the membrane permeability. Desalination, 250(2): 601–604. http://dx.doi.org/10.1016/j.desal.2009.09.032
LaPara, T.M., Nakatsu, C.H, Pantea, L.M., et al., 2002. Stability of the bacterial communities supported by a severnstage biological process treating pharmaceutical wastewater as revealed by PCR-DGGE. Water Research, 36(3): 638–646. http://dx.doi.org/10.1016/S0043-1354(01)00277-9
Li, A.J., Yang, S.F., Li, X.Y., et al., 2008. Microbial population dynamics during aerobic sludge granulation at different organic loading rates. Water Research, 42(13): 3552–3560. http://dx.doi.org/10.1016/j.watres.2008.05.005
Li, J., Xi, D.L., Shi, Y., 2008. Resistance distribution and fouling mechanism of dynamic membrane in wastewater treatment. Journal of Chemical Industry and Engineering, 59(9): 2309–2315 (in Chinese).
Li, P., Wang, Y.X., Wang, Y.H., et al., 2010. Bacterial community structure and diversity during establishment of an anaerobic bioreactor to treat swine wastewater. Water Science and Technology, 61(1): 243–252. http://dx.doi.org/10.2166/wst.2010.807
Li, R., Huang, X., Wang, Z.Q., 2000. Hydraulic characteristics in a submerged membrane bioreactor. Environmental Science, 21(5): 47–50 (in Chinese).
Liaw, R.B., Cheng, M.P., Wu, M.C., et al., 2010. Use of metagenomic approaches to isolate lipolytic genes from activated sludge. Bioresource Technology, 101(21): 8323–8329. http://dx.doi.org/10.1016/j.biortech.2010.05.091
Mannina, G., Cosenza, A., 2013. The fouling phenomenon in membrane bioreactors: assessment of different strategies for energy saving. Journal of Membrane Science, 444: 332–344. http://dx.doi.org/10.1016/j.memsci.2013.05.047
Mannina, G., Cosenza, A., Di Trapani, D., et al., 2016a. Membrane bioreactors for treatment of saline wastewater contaminated by hydrocarbons (diesel fuel): an experimental pilot plant case study. Chemical Engineering Journal, 291: 269–278. http://dx.doi.org/10.1016/j.cej.2016.01.107
Mannina, G., Capodici, M., Cosenza, A., et al., 2016b. Sequential batch membrane bio-reactor for wastewater treatment: the effect of increased salinity. Bioresource Technology, 209: 205–212. http://dx.doi.org/10.1016/j.biortech.2016.02.122
Meng, F.G., Zhang, H.M., Yang, F.L., et al., 2007. Characterization of cake layer in submerged membrane bioreactor. Environmental Science and Technology, 41(11): 4065–4070. http://dx.doi.org/10.1021/es062208b
Meng, F.G., Chae, S.R., Drews, A., et al., 2009. Recent advances in membrane bioreactors (MBRs): membrane fouling and membrane material. Water Research, 43(6): 1489–1512. http://dx.doi.org/10.1016/j.watres.2008.12.044
MOTIMO, 2006. User’s Guide for Membrane Using. Tianjin MOTIMO Membrane Technology Co. Ltd., Tianjin, China (in Chinese).
Muyzer, G., DeWaal, E.C., Uitterlinden, A.G., 1993. Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reationamplified genes coding for 16S rRNA. Applied and Environmental Microbiology, 59(3): 695–700.
Pontoni, L., D’Alessandro, G., d’Antonio, G., et al., 2015. Effect of anaerobic digestion on rheological parameters and dewaterability of aerobic sludges from MBR and conventional activated sludge plants. Chemical Engineering Transactions, 43: 2311–2316. http://dx.doi.org/10.3303/CET1543386
Reboleiro-Rivas, P., Martín-Pascual, J., Morillo, J.A., et al., 2016. Interlinkages between bacterial populations dynamics and the operational parameters in a moving bed membrane bioreactor treating urban sewage. Water Research, 88: 796–807. http://dx.doi.org/10.1016/j.watres.2015.10.059
Rotaru, C., Woodard, T.L., Choi, S., et al., 2012. Spatial heterogeneity of bacterial communities in sediments from an infiltration basin receiving highway runoff. Microbial Ecology, 64(2): 461–473. http://dx.doi.org/10.1007/s00248-012-0026-x
Rudd, T., Sterritt, R.M., Lester, J.N., 1984. Complexation of heavy metals by extracellular polymers in the activated sludge process. Journal of Water Pollution Control Federation, 56: 1260–1268.
Sanguinetti, C.J., Dias Neto, E., Simpson, A.J., 1994. Rapid silver staining and recovery of PCR products separated on polyacrylamide gels. Biotechniques, 17(5): 914–921.
Silva, C.C., Viero, A.F., Andreote, F.D., et al., 2010. Monitoring the bacterial community dynamics in a petroleum refinery wastewater membrane bioreactor fed with a high phenolic load. Journal of Microbiology and Biotechnology, 20(1): 21–29.
Tan, M., Qiu, G.L., Ting, Y.P., 2015. Effects of ZnO nanoparticles on wastewater treatment and their removal behavior in a membrane bioreactor. Bioresource Technology, 185: 125–133. http://dx.doi.org/10.1016/j.biortech.2015.02.094
Tu, T., Li, L., Mao, G.N., et al., 2012. Analysis of bacterial diversity in the Songhua River based on nested PCR and DGGE. Acta Ecologica Sinica, 32(11): 3505–3515 (in Chinese). http://dx.doi.org/10.5846/stxb201105160635
Wang, J.F., Wang, X., Zhao, Z.G., et al., 2008. Organics and nitrogen removal and sludge stability in aerobic granular sludge membrane bioreactor. Applied Microbiology and Biotechnology, 79(4): 679–685. http://dx.doi.org/10.1007/s00253-008-1466-6
Wang, X., Zhang, B., Shen, Z.Q., et al., 2010. The EPS characteristics of sludge in an aerobic granule membrane bioreactor. Bioresource Technology, 101(21): 8046–8050. http://dx.doi.org/10.1016/j.biortech.2010.05.074
Wei, Y.J., Ji, M., Li, G.Y., et al., 2012. Microbial and hydrodynamic properties of aerobic granules in a sequencing batch reactor treating landfill leachate. Journal of Zhejiang University-SCIENCE A (Applied Physics & Engineering), 13(3): 219–229. http://dx.doi.org/10.1631/jzus.A1100153
Xia, S.Q., Li, J.X., He, S.Y., et al., 2010. The effect of organic loading on bacterial community composition of membrane biofilms in a submerged polyvinyl chloride membrane bioreactor. Bioresource Technology, 101(17): 6601–6609. http://dx.doi.org/10.1016/j.biortech.2010.03.082
Ying, W., Yang, F., Bick, A., et al., 2010. Extracellular polymeric substances (EPS) in a hybrid growth membrane bioreactor (HG-MBR): viscoelastic and adherence characteristics. Environmental Science and Technology, 44(22): 8636–8643. http://dx.doi.org/10.1021/es102309y
Zhang, B., Sun, B.S., Ji, M., et al., 2008a. Analysis and succession of microbial community structure in a membrane bioreactor. Acta Scientiae Circumstantiae, 28(11): 2192–2199 (in Chinese).
Zhang, B., Sun, B.S., Jin, M., et al., 2008b. Extraction and analysis of extracellular polymeric substances in membrane fouling in submerged MBR. Desalination, 227(1–3):286–294. http://dx.doi.org/10.1016/j.desal.2007.06.032
Zhang, B., Sun, B.S., Ji, M., et al., 2010. Quantification and comparison of ammonia-oxidizing bacterial communities in MBRs treating various types of wastewater. Bioresource Technology, 101(9): 3054–3059. http://dx.doi.org/10.1016/j.biortech.2009.12.048
Zhang, Z.J., 2000. Wastewater Engineering, 2nd Edition. China Construction Industry Press, Beijing, China, p.386–387 (in Chinese).
Ziembinska, A., Ciesielski, S., Gnida, A., et al., 2012. Comparison of ammonia-oxidizing bacterial community structure in membrane-assisted bioreactors using PCRDGGE and FISH. Journal of Microbiology and Biotechnology, 22(8): 1035–1043. http://dx.doi.org/10.4014/jmb.1201.01014
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by the Science and Technology Project of the Ministry of Transport (No. 2011-329-224-330) and the National Nonprofit Institute Research Grants of Tianjin Research Institute for Water Transport Engineering (Nos. TKS100216, TKS130206, and TKS160215), China
ORCID: Yan-jie WEI, http://orcid.org/0000-0002-9007-450X
Rights and permissions
About this article
Cite this article
Wei, Yj., Li, Gy. Membrane fouling behavior and microbial community succession in a submerged membrane bioreactor treating harbor oily wastewater. J. Zhejiang Univ. Sci. A 17, 745–757 (2016). https://doi.org/10.1631/jzus.A1500289
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1631/jzus.A1500289
Keywords
- Membrane bioreactor (MBR) fouling
- Extracellular polymeric substances (EPS)
- Harbor oily wastewater
- Microbial community