Abstract
Cell-releasing hydrogel carriers enhance cell-based therapies by improving retention and viability within target tissues. We previously demonstrated tunable cell release from a hydrolytically labile β-thioester-containing polyethylene glycol-based hydrogel. However, the macromer synthesis dependent on thiol-Michael addition leads to batch variability and heterogeneous hydrogel properties. Herein, we report a new synthetic route with improved control of macromer molecular weight and characterize the resulting hydrogel properties. The resulting PEG dithioester acrylamide (PEGDTEA) hydrogel demonstrates a more controlled synthetic route to generate targeted molecular weights with reduced couplings, creating a more predictable and tunable hydrogel microenvironment for precise cell delivery applications.
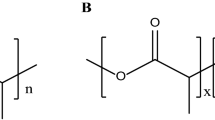
Graphical abstract

Synthesis of thioester containing PEG macromer for more predictable and tunable cell-releasing hydrogels



Similar content being viewed by others
Data availability
Please email the authors for full access to the data.
References
X. Wang, N. Rivera-Bolanos, B. Jiang, G.A. Ameer, Advanced functional biomaterials for stem cell delivery in regenerative engineering and medicine. Adv. Funct. Mater. 29, 1809009 (2019). https://doi.org/10.1002/adfm.201809009
A. Golchin, T.Z. Farahany, Biological products: cellular therapy and FDA approved products. Stem Cell Rev. Rep. 15, 166–175 (2019). https://doi.org/10.1007/s12015-018-9866-1
D. Tonkin, A. Yee-Goh, R. Katare, Healing the ischaemic heart: a critical review of stem cell therapies. RCM 24, 122 (2023). https://doi.org/10.31083/j.rcm2404122
A. Gratwohl, M.C. Pasquini, M. Aljurf et al., One million haemopoietic stem-cell transplants: a retrospective observational study. Lancet Haematol. 2, e91–e100 (2015). https://doi.org/10.1016/S2352-3026(15)00028-9
L.M. Marquardt, S.C. Heilshorn, Design of injectable materials to improve stem cell transplantation. Curr. Stem Cell Rep. 2, 207–220 (2016). https://doi.org/10.1007/s40778-016-0058-0
M. Iwasaki, J.T. Wilcox, Y. Nishimura et al., Synergistic effects of self-assembling peptide and neural stem/progenitor cells to promote tissue repair and forelimb functional recovery in cervical spinal cord injury. Biomaterials 35, 2617–2629 (2014). https://doi.org/10.1016/j.biomaterials.2013.12.019
J.M. Singelyn, K.L. Christman, Injectable materials for the treatment of myocardial infarction and heart failure: the promise of decellularized matrices. J. Cardiovasc. Transl. Res. 3, 478–486 (2010). https://doi.org/10.1007/s12265-010-9202-x
J. Yang, X. Sun, Y. Zhang, Y. Chen, Chapter 10—the application of natural polymer-based hydrogels in tissue engineering, in Hydrogels Based on Natural Polymers. ed. by Y. Chen (Elsevier, Amsterdam, 2020), pp.273–307
M. Whitely, S. Cereceres, P. Dhavalikar et al., Improved in situ seeding of 3D printed scaffolds using cell-releasing hydrogels. Biomaterials 185, 194–204 (2018). https://doi.org/10.1016/j.biomaterials.2018.09.027
S. Cereceres, Z. Lan, L. Bryan et al., In vivo characterization of poly(ethylene glycol) hydrogels with thio-β esters. Ann. Biomed. Eng. 48, 953–967 (2020). https://doi.org/10.1007/s10439-019-02271-8
N.A. Peppas, P. Bures, W. Leobandung, H. Ichikawa, Hydrogels in pharmaceutical formulations. Eur. J. Pharm. Biopharm. 50, 27–46 (2000). https://doi.org/10.1016/s0939-6411(00)00090-4
N. Zaquen, B. Wenn, K. Ranieri et al., Facile design of degradable poly(β-thioester)s with tunable structure and functionality. J. Polym. Sci. A 52, 178–187 (2014). https://doi.org/10.1002/pola.26986
A.J. Engler, S. Sen, H.L. Sweeney, D.E. Discher, Matrix elasticity directs stem cell lineage specification. Cell 126, 677–689 (2006). https://doi.org/10.1016/j.cell.2006.06.044
J.S. Choi, B.P. Mahadik, B.A.C. Harley, Engineering the hematopoietic stem cell niche: frontiers in biomaterial science. Biotechnol. J. 10, 1529–1545 (2015). https://doi.org/10.1002/biot.201400758
A.E. Gilchrist, S. Lee, Y. Hu, B.A.C. Harley, Soluble signals and remodeling in a synthetic gelatin-based hematopoietic stem cell niche. Adv. Healthc. Mater. 8, 1900751 (2019). https://doi.org/10.1002/adhm.201900751
M.S. Hahn, L.J. Taite, J.J. Moon et al., Photolithographic patterning of polyethylene glycol hydrogels. Biomaterials 27, 2519–2524 (2006). https://doi.org/10.1016/j.biomaterials.2005.11.045
S.H. Frayne, B.H. Northrop, Evaluating nucleophile byproduct formation during phosphine- and amine-promoted thiol-methyl acrylate reactions. J. Org. Chem. 83, 10370–10382 (2018). https://doi.org/10.1021/acs.joc.8b01471
J. Magano, Large-scale amidations in process chemistry: practical considerations for reagent selection and reaction execution. Org. Process Res. Dev. 26, 1562–1689 (2022). https://doi.org/10.1021/acs.oprd.2c00005
G.J. Rodriguez-Rivera, A. Post, M. John, et al., Towards prevention of re-entrant arrhythmias: injectable hydrogel electrodes enable direct capture of previously inaccessible cardiac tissue (2023). https://doi.org/10.1101/2021.11.03.467102
D.P. Nair, M. Podgórski, S. Chatani et al., The thiol-Michael addition click reaction: a powerful and widely used tool in materials chemistry. Chem. Mater. 26, 724–744 (2014). https://doi.org/10.1021/cm402180t
G.-Z. Li, K.R. Randev, H.A. Soeriyadi, et al., Investigation into thiol-(meth)acrylate Michael addition reactions using amine and phosphine catalysts. Polym. Chem. 1, 1196–1204 (2010). https://doi.org/10.1039/C0PY00100G
M. Liu, B. Hoon Tan, P.R. Burford, B.A. Lowe, Nucleophilic thiol-Michael chemistry and hyperbranched (co)polymers: synthesis and ring-opening metathesis (co)polymerization of novel difunctional exo-7-oxanorbornenes with in situ inimer formation. Polym. Chem. 4, 3300–3311 (2013). https://doi.org/10.1039/C3PY00110E
J.W. Chan, B. Yu, C.E. Hoyle, A.B. Lowe, The nucleophilic, phosphine-catalyzed thiol-ene click reaction and convergent star synthesis with RAFT-prepared homopolymers. Polymer 50, 3158–3168 (2009). https://doi.org/10.1016/j.polymer.2009.04.030
F.C. Nising, S. Bräse, Recent developments in the field of oxa-Michael reactions. Chem. Soc. Rev. 41, 988–999 (2012). https://doi.org/10.1039/C1CS15167C
M.B. Browning, S.N. Cereceres, P.T. Luong, E.M. Cosgriff-Hernandez, Determination of the in vivo degradation mechanism of PEGDA hydrogels. J. Biomed. Mater. Res. A 102, 4244–4251 (2014). https://doi.org/10.1002/jbm.a.35096
Funding
This work was supported by the National Institutes of Health (NIH/NIAMS R21 AR076708) and the Early Career Provost Fellowship Program from the Provost's Office of Diversity at the University of Texas at Austin. Graphical abstract was created using BioRender.com.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Green, M., Hicks, A., Sullivan, T. et al. New synthetic route to improve uniformity of cell-releasing PEG-based hydrogel carriers. MRS Communications 13, 901–906 (2023). https://doi.org/10.1557/s43579-023-00446-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/s43579-023-00446-y