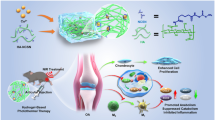
Abstract
In this study, a hybrid dual drug-loaded hydroxyapatite-oxidized dextran methacrylate core–shell nanocarrier was formulated and explored for combinatorial delivery of doxorubicin (DOX) and methotrexate (MTX) to bone cancer. The synthesized nanocarrier was well characterized by different techniques. In vitro drug release studies in both acidic (pH 5) and alkaline (pH 7.4) conditions showed sequential release of MTX followed by DOX in a sustained manner for 10 days. Biocompatibility and cytotoxicity studies performed using drug-loaded nanoparticles (NPs) on fibroblast L929 cells and osteosarcoma MG63 cells (OMG63) showed that the NPs were highly biocompatible and showed concentration-dependent toxicity. Gene expression studies in OMG-63 cells exhibited the upregulation of caspase-3 and BAX which confirmed the apoptosis induced by dual drug-loaded NPs. The nanocarrier is expected to be a potential bone void filling material, as well as a platform for sequential delivery of DOX and MTX for the treatment of bone cancer.











Similar content being viewed by others
References
T. Ibrahim, L. Mercatali, and D. Amadori: Bone and cancer: The osteoncology. Clin. Cases Miner. Bone Metab. 10, 121 (2013).
J.T. Buijs and G. van der Pluijm: Osteotropic cancers: From primary tumor to bone. Cancer Lett. 273, 177–193 (2009).
T. Kimura: Multidisciplinary approach for bone metastasis: A review. Cancers 10, 156 (2018).
W. Gu, C. Wu, J. Chen, and Y. Xiao: Nanotechnology in the targeted drug delivery for bone diseases and bone regeneration. Int. J. Nanomedicine 8, 2305–2317 (2013).
F. Macedo, K. Ladeira, F. Pinho, N. Saraiva, N. Bonito, L. Pinto, and F. Gonçalves: Bone metastases: An overview. Oncol. Rev. 11, 321 (2017).
S. Hossen, M.K. Hossain, M.K. Basher, M.N. Mia, M.T. Rahman, and M.J. Uddin: Smart nanocarrier-based drug delivery systems for cancer therapy and toxicity studies: A review. J. Adv. Res. 15, 1–18 (2019).
C. Marques, J.M. Ferreira, E. Andronescu, D. Ficai, M. Sonmez, and A. Ficai: Multifunctional materials for bone cancer treatment. Int. J. Nanomedicine 9, 2713–2725 (2014).
E.M. Alexandrino, S. Ritz, F. Marsico, G. Baier, V. Mailänder, K. Landfester, and F.R. Wurm: Paclitaxel-loaded polyphosphate nanoparticles: A potential strategy for bone cancer treatment. J. Mater. Chem. B. 2, 1298 (2014).
H. Ma, C. He, Y. Cheng, Z. Yang, J. Zang, J. Liu, and X. Chen: Localized co-delivery of doxorubicin, cisplatin, and methotrexate by thermosensitive hydrogels for enhanced osteosarcoma treatment. ACS Appl. Mater. Interfaces 7, 27040–27048 (2015).
J.A. Kemp, M.S. Shim, C.Y. Heo, and Y.J. Kwon: “Combo” nanomedicine: Co-delivery of multi-modal therapeutics for efficient, targeted, and safe cancer therapy. Adv. Drug Deliv. Rev. 98, 3–18 (2016).
P. Parhi, C. Mohanty, and S.K. Sahoo: Nanotechnology-based combinational drug delivery: An emerging approach for cancer therapy. Drug Discov. Today. 17, 1044–1052 (2012).
A. Wicki, D. Witzigmann, V. Balasubramanian, and J. Huwyler: Nanomedicine in cancer therapy: Challenges, opportunities, and clinical applications. J. Control. Release 28, 138–157 (2015).
C.M. Hu, S. Aryal, and L. Zhang: Nanoparticle-assisted combination therapies for effective cancer treatment. Ther. Deliv 1, 323–334 (2010).
S.S. Qi, J.H. Sun, H.H. Yu, and S.Q. Yu: Co-delivery nanoparticles of anti-cancer drugs for improving chemotherapy efficacy. Drug Deliv. 24, 1909–1926 (2017).
M. Estanqueiro, M.H. Amaral, J. Conceicao, and J.M. Lobo: Nanotechnological carriers for cancer chemotherapy: The state of the art. Colloids Surf. B: Biointerfaces 126, 631–648 (2015).
C.Y. Zhao, R. Cheng, Z. Yang, and Z.M. Tian: Nanotechnology for cancer therapy based on chemotherapy. Molecules. 23, 826 (2018).
M. Sadat-Shojai, M.T. Khorasani, E. Dinpanah-Khoshdargi, and A. Jamshidi: Synthesis methods for nanosized hydroxyapatite with diverse structures. Acta Biomater. 9, 7591–7621 (2013).
X. Cheng and L. Kuhn: Chemotherapy drug delivery from calcium phosphate nanoparticles. Int. J. Nanomedicine 2, 667 (2007).
L. Yang, B.W. Sheldon, and T.J. Webster: Nanophase ceramics for improved drug delivery. Am. Ceram. Soc. Bull. 89, 24–32 (2010).
K.D. Son and Y.J. Kim: Anticancer activity of drug-loaded calcium phosphate nanocomposites against human osteosarcoma. Biomater. Res. 21, 13 (2017).
W.M. Li, C.W. Su, Y.W. Chen, and S.Y. Chen: In situ DOX-calcium phosphate mineralized CPT-amphiphilic gelatin nanoparticle for intracellular controlled sequential release of multiple drugs. Acta Biomater. 15, 191–199 (2015).
A. Lebugle, A. Rodrigues, P. Bonnevialle, J.J. Voigt, P. Canal, and F. Rodriguez: Study of implantable calcium phosphate systems for the slow release of methotrexate. Biomaterials. 23, 3517–3522 (2002).
M.F. Cipreste and E.M.B. Sousa: Poly(vinyl alcohol)/collagen/hydroxyapatite nanoparticles hybrid system containing yttrium-90 as a potential agent to treat osteosarcoma. J. Biomater. Nanobiotechnol. 5, 24–30 (2014).
Z.J. Rong, L.J. Yang, B.T. Cai, L.X. Zhu, Y.L. Cao, G.F. Wu, and Z.J. Zhang: Porous nano-hydroxyapatite/collagen scaffold containing drug-loaded ADM-PLGA microspheres for bone cancer treatment. J. Mater. Sci. Mater. Med. 27, 89 (2016).
Y. Wang, X. Zhang, J. Yan, Y. Xiao, and M. Lang: Surface modification of hydroxyapatite with poly(methyl methacrylate) via surface-initiated ATRP. Appl. Surf. Sci. 257, 6233–6238 (2011).
Q. Liu, J.R. de Wijn, and C.A. Van Blitterswijk: Covalent bonding of PMMA, PBMA, and poly (HEMA) to hydroxyapatite particles. J. Biomed. Mater. Res. 40, 257–263 (1998).
Q. Liu, J.R. de Wijn, K. de Groot, and C.A. van Blitterswijk: Surface modification of nano-apatite by grafting organic polymer. Biomaterials 19, 1067–1072 (1998).
S. Kango, S. Kalia, A. Celli, J. Njuguna, Y. Habibi, and R. Kumar: Surface modification of inorganic nanoparticles for development of organic–inorganic nanocomposites–a review. Prog. Polym. Sci. 38, 1232–1261 (2013).
J. Wei, P. He, A. Liu, X. Chen, X. Wang, and X. Jing: Surface modification of hydroxyapatite nanoparticles with thermal-responsive PNIPAM by ATRP. Macromol. Biosci. 9, 1237–1246 (2009).
A. Matsuda, T. Furuzono, D. Walsh, A. Kishida, and J. Tanaka: Surface modification of a porous hydroxyapatite to promote bonded polymer coatings. J. Mater. Sci. Mater 14, 973–978 (2003).
S.R. Prasad, A. Jayakrishnan, and T.S. Sampath Kumar: Hydroxyapatite-poly(vinyl alcohol) core-shell nanoparticles for dual delivery of methotrexate and gemcitabine for bone cancer treatment. J. Drug Deliv. Sci. Technol. 51, 629–638 (2019).
S.S. Dhaneshwar, K. Mini, N. Gairola, and S.S. Kadam: Dextran: A promising macromolecular drug carrier. Indian J. Pharm. Sci. 68, 705 (2006).
J.D. Cabral, M. Roxburgh, Z. Shi, L. Liu, M. McConnell, G. Williams, N. Evans, L.R. Hanton, J. Simpson, S.C. Moratti, and B.H. Robinson: Synthesis, physiochemical characterization, and biocompatibility of a chitosan/dextran-based hydrogel for postsurgical adhesion prevention. J. Mater. Sci. Mater. Med. 25, 2743–2756 (2014).
J. Varshosaz: Dextran conjugates in drug delivery. Expert Opin. Drug Deliv. 9, 509–523 (2012).
X. Feng, D. Li, J. Han, X. Zhuang, and J. Ding: Schiff base bond-linked polysaccharide-doxorubicin conjugate for upregulated cancer therapy. Mater. Sci. Eng. C Mater. Biol. Appl. 76, 1121–1128 (2017).
T. Doane and C. Burda: Nanoparticle mediated non-covalent drug delivery. Adv. Drug Deliv. Rev. 65, 607–621 (2013).
L. Qiu, C.Y. Hong, and C.Y. Pan: Doxorubicin-loaded aromatic imine-contained amphiphilic branched star polymer micelles: synthesis, self-assembly, and drug delivery. Int. J. Nanomedicine 10, 3623–3640 (2015).
T. Miao, J. Wang, Y. Zeng, G. Liu, and X. Chen: Polysaccharide-based controlled release systems for therapeutics delivery and tissue engineering: From bench to bedside. Adv. Sci. 5, 700513 (2018).
Y. Zhang, M. Huo, J. Zhou, A. Zou, W. Li, C. Yao, and S. Xie: DDSolver: An add-in program for modeling and comparison of drug dissolution profiles. AAPS J. 12, 263–271 (2010).
M. Gierszewska-Drużyńska, and J. Ostrowska-Czubenko: Mechanism of water diffusion into noncrosslinked and ionically crosslinked chitosan membranes. Prog. Chem. Appl. Chitin Deriv. 17, 63–70 (2012).
R.Y. Basha, T.S. Sampath Kumar, and M. Doble: Dual delivery of tuberculosis drugs via cyclodextrin conjugated curdlan nanoparticles to infected macrophages. Carbohydr. Polym. 218, 53–62 (2019).
International Organization for Standardization. ISO 10993-5. Biological Evaluation of Medical Devices–Part 5: Tests for In vitro Cytotoxicity (ISO, Geneva, 2009).
T.C. Chou: Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol. Rev. 58, 621–681 (2006).
W. Song, Z. Tang, M. Li, S. Lv, H. Sun, M. Deng, H. Liu, and X. Chen: Polypeptide-based combination of paclitaxel and cisplatin for enhanced chemotherapy efficacy and reduced side-effects. Acta Biomater. 10, 1392–1402 (2014).
M. Rahimi, K.D. Safa, and R. Salehi: Co-delivery of doxorubicin and methotrexate by dendritic chitosan-g-mPEG as a magnetic nanocarrier for multi-drug delivery in combination chemotherapy. Polym. Chem. 8, 7333–7350 (2017).
S.W. Lowe and A.W. Lin: Apoptosis in cancer. Carcinogenesis. 21, 485–495 (2000).
S.H. Kim and C.C. Chu: Synthesis and characterization of dextran–methacrylate hydrogels and structural study by SEM. J. Biomed. Mater. Res. 49(4), 517–27 (2000).
Y. Zhang and J. Lu: A simple method to tailor spherical nanocrystal hydroxyapatite at low temperature. J. Nanoparticle Res. 9, 589–594 (2006).
A.P. Francis, S. Gurudevan, and A. Jayakrishnan: Synthetic polymannose as a drug carrier: synthesis, toxicity and anti-fungal activity of polymannose-amphotericin B conjugates. J. Biomater Sci. Polym. 29, 1529–1548 (2018).
C.K. Balavigneswaran, S.K. Mahto, B. Subia, A. Prabhakar, K. Mitra, V. Rao, M. Ganguli, B. Ray, P. Maiti, and N. Misra: Tailored chemical properties of 4-Arm star shaped poly(d,l-lactide) as cell adhesive three-dimensional scaffolds. Bioconjugate Chem. 28, 1236–1250 (2017).
Acknowledgments
The authors thank Prof. Nitish R. Mahapatra for providing cell culture facility and thanks are due to Mr. Abrar Ali Khan and Mrs. Hemalatha who helped in gene expression studies. The authors thank Professor Sakthi Kumar of Toyo University, Japan for the TEM analysis. The authors also thank Central XRD Facility and Sophisticated Analytical Instrument Facility (SAIF) of IIT Madras for analytical support. This research did not receive any specific grant from any funding agencies in the public, commercial or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding authors
Supplementary materials
Supplementary materials
To view supplementary material for this article, please visit https://doi.org/10.1557/jmr.2020.193
Rights and permissions
About this article
Cite this article
Ram Prasad, S., Jayakrishnan, A. & Sampath Kumar, T.S. Hydroxyapatite-dextran methacrylate core/shell hybrid nanocarriers for combinatorial drug therapy. Journal of Materials Research 35, 2451–2465 (2020). https://doi.org/10.1557/jmr.2020.193
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2020.193