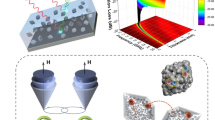
Abstract
Significant reductions recently seen in the size of wide-bandgap power electronics have not been accompanied by a relative decrease in the size of the corresponding magnetic components. To achieve this, a new generation of materials with high magnetic saturation and permeability are needed. Here, we develop gram-scale syntheses of superparamagnetic Fe/FexOy core–shell nanoparticles and incorporate them as the magnetic component in a strongly magnetic nanocomposite. Nanocomposites are typically formed by the organization of nanoparticles within a polymeric matrix. However, this approach can lead to high organic fractions and phase separation; reducing the performance of the resulting material. Here, we form aminated nanoparticles that are then cross-linked using epoxy chemistry. The result is a magnetic nanoparticle component that is covalently linked and well separated. By using this ‘matrix-free’ approach, we can substantially increase the magnetic nanoparticle fraction, while still maintaining good separation, leading to a superparamagnetic nanocomposite with strong magnetic properties.







Similar content being viewed by others
References
W.G. Hurley and W.H. Wolfle: Transformers and Inductors for Power Electronics: Theory, Design and Applications, 1st ed. (John Wiley & Sons Ltd., West Sussex, U.K., 2013).
C. Beatrice, S. Dobak, E. Ferrara, F. Fiorillo, C. Ragusa, J. Fuzer, and P. Kollar: Broadband magnetic losses of nanocrystalline ribbons and powder cores. J. Magn. Magn. Mater. 420, 317–323 (2016).
K. Mandel, F. Hutter, C. Gellermann, and G. Sextl: Modified superparamagnetic nanocomposite microparticles for highly selective Hg(II) or Cu(II) separation and recovery from aqueous solutions. ACS Appl. Mater. Interfaces 4, 5633–5642 (2012).
W. Dong, Y. Li, D. Niu, Z. Ma, J. Gu, Y. Chen, W. Zhao, X. Liu, C. Liu, and J. Shi: Facile synthesis of monodisperse superparamagnetic Fe3O4 core@hybrid@Au shell nanocomposite for bimodal imaging and photothermal therapy. Adv. Mater. 23, 5392–5397 (2011).
W.L. Gu, X. Deng, X.X. Gu, X.F. Jia, B.H. Lou, X.W. Zhang, J. Li, and E.K. Wang: Stabilized, superparamagnetic functionalized graphene/Fe3O4@Au nanocomposites for a magnetically-controlled solid-state electrochemiluminescence biosensing application. Anal. Chem. 87, 1876–1881 (2015).
L.J. Zhu, D.L. Wang, X. Wei, X.Y. Zhu, J.Q. Li, C.L. Tu, Y. Su, J.L. Wu, B.S. Zhu, and D.Y. Yan: Multifunctional pH-sensitive superparamagnetic iron-oxide nanocomposites for targeted drug delivery and MR imaging. J. Controlled Release 169, 228–238 (2013).
B. Cullity: Introduction to Magnetic Materials (Addison-Wesley Pub. Co., Reading, MA, 1972).
D.L. Huber: Synthesis, properties, and applications of iron nanoparticles. Small 1, 482–501 (2005).
M. Knobel, W.C. Nunes, L.M. Socolovsky, E. De Biasi, J.M. Vargas, and J.C. Denardin: Superparamagnetism and other magnetic features in granular materials: A review on ideal and real systems. J. Nanosci. Nanotechnol. 8, 2836–2857 (2008).
J. Pyun: Nanocomposite materials from functional polymers and magnetic colloids. Polym. Rev. 47, 231–263 (2007).
R. Stone, S. Hipp, J. Barden, P.J. Brown, and O.T. Mefford: Highly scalable nanoparticle–polymer composite fiber via wet spinning. J. Appl. Polym. Sci. 130, 1975–1980 (2013).
H. Wakayama and H. Yonekura: Synthesis and magnetic properties of FePt nanocomposite magnets via self-assembled block copolymer templates. Mater. Lett. 171, 268–272 (2016).
S. Behrens and I. Appel: Magnetic nanocomposites. Curr. Opin. Biotechnol. 39, 89–96 (2016).
S. Chen, S. Zhang, T. Jin, and G. Zhao: Synthesis and characterization of novel covalently linked waterborne polyurethane/Fe3O4 nanocomposite films with superior magnetic, conductive properties and high latex storage stability. Chem. Eng. J. 286, 249–258 (2016).
A.C. de Leon, Q. Chen, N.B. Palaganas, J.O. Palaganas, J. Manapat, and R.C. Advincula: High performance polymer nanocomposites for additive manufacturing applications. React. Funct. Polym. 103, 141–155 (2016).
J.B. Hooper and K.S. Schweizer: Theory of phase separation in polymer nanocomposites. Macromolecules 39, 5133–5142 (2006).
V.N. Mochalin, I. Neitzel, B.J.M. Etzold, A. Peterson, G. Palmese, and Y. Gogotsi: Covalent incorporation of aminated nanodiamond into an epoxy polymer network. ACS Nano 5, 7494–7502 (2011).
B.I. Dach, H.R. Rengifo, N.J. Turro, and J.T. Koberstein: Cross-linked “matrix-free” nanocomposites from reactive polymer-silica hybrid nanoparticles. Macromolecules 43, 6549–6552 (2010).
B.G. Compton and J.A. Lewis: 3D-printing of lightweight cellular composites. Adv. Mater. 26, 5930–5935 (2014).
F.L. Jin, X. Li, and S.J. Park: Synthesis and application of epoxy resins: A review. J. Ind. Eng. Chem. 29, 1–11 (2015).
Y. Sugawa, K. Ishidate, M. Sonehara, and T. Sato: Carbonyl-iron/epoxy composite magnetic core for planar power inductor used in package-level power grid. IEEE Trans. Magn. 49, 4172–4175 (2013).
H. Gu, S. Tadakamalla, Y. Huang, H.A. Colorado, Z. Luo, N. Haldolaarachchige, D.P. Young, S. Wei, and Z. Guo: Polyaniline stabilized magnetite nanoparticle reinforced epoxy nanocomposites. ACS Appl. Mater. Interfaces 4, 5613–5624 (2012).
J.H. Zhu, S.Y. Wei, J. Ryu, L.Y. Sun, Z.P. Luo, and Z.H. Guo: Magnetic epoxy resin nanocomposites reinforced with core–shell structured Fe@FeO nanoparticles: Fabrication and property analysis. ACS Appl. Mater. Interfaces 2, 2100–2107 (2010).
Z.S. Pour and M. Ghaemy: Thermo-mechanical behaviors of epoxy resins reinforced with silane-epoxide functionalized α-Fe2O3 nanoparticles. Prog. Org. Coat. 77, 1316–1324 (2014).
B.T. Naughton, P. Majewski, and D.R. Clarke: Magnetic properties of nickel–zinc ferrite toroids prepared from nanoparticles. J. Am. Ceram. Soc. 90, 3547–3553 (2007).
N. Mikuszeit, E.Y. Vedmedenko, and H.P. Oepen: Multipole interaction of polarized single-domain particles. J. Phys. Condens. Matter 16, 9037–9045 (2004).
G.C. Bleier, J. Watt, C.K. Simocko, J.M. Lavin, and D.L. Huber: Reversible magnetic agglomeration—A mechanism for true thermodynamic control over nanoparticle size. Angew. Chem. Int. Ed. Engl. (2018) DOI: https://doi.org/10.1002/anie.201800959.
B.D. Fellows, S. Sandler, J. Livingston, K. Fuller, L. Nwandu, S. Timmins, K.A. Lantz, M. Stefik, and O.T. Mefford: Extended LaMer synthesis of cobalt-doped ferrite. IEEE Magn. Lett. 9, 1–5 (2018).
E.C. Vreeland, J. Watt, G.B. Schober, B.G. Hance, M.J. Austin, A.D. Price, B.D. Fellows, T.C. Monson, N.S. Hudak, L. Maldonado-Camargo, A.C. Bohorquez, C. Rinaldi, and D.L. Huber: Enhanced nanoparticle size control by extending LaMer’s mechanism. Chem. Mater. 27, 6059–6066 (2015).
M. Unni, A.M. Uhl, S. Savliwala, B.H. Savitzky, R. Dhavalikar, N. Garraud, D.P. Arnold, L.F. Kourkoutis, J.S. Andrew, and C. Rinaldi: Thermal decomposition synthesis of iron oxide nanoparticles with diminished magnetic dead layer by controlled addition of oxygen. ACS Nano 11, 2284–2303 (2017).
T.C. Monson, Q. Ma, T.E. Stevens, J.M. Lavin, J.L. Leger, P.V. Klimov, and D.L. Huber: Implication of ligand choice on surface properties, crystal structure, and magnetic properties of iron nanoparticles. Part. Part. Syst. Char. 30, 258–265 (2013).
G. Concas, F. Congiu, G. Muscas, and D. Peddis: Determination of blocking temperature in magnetization and mössbauer time scale: A functional form approach. J. Phys. Chem. C 121, 16541–16548 (2017).
J. Watt, G.C. Bleier, M.J. Austin, S.A. Ivanov, and D.L. Huber: Non-volatile iron carbonyls as versatile precursors for the synthesis of iron-containing nanoparticles. Nanoscale 9, 6632–6637 (2017).
H. Yun, J. Kim, T. Paik, L.Y. Meng, P.S. Jo, J.M. Kikkawa, C.R. Kagan, M.G. Allen, and C.B. Murray: Alternate current magnetic property characterization of nonstoichiometric zinc ferrite nanocrystals for inductor fabrication via a solution based process. J. Appl. Phys. 119 (2016).
J. Park, J. Joo, S.G. Kwon, Y. Jang, and T. Hyeon: Synthesis of monodisperse spherical nanocrystals. Angew. Chem., Int. Ed. Engl. 46, 4630–4660 (2007).
S. Schonecker, X. Li, B. Johansson, S.K. Kwon, and L. Vitos: Thermal surface free energy and stress of iron. Sci. Rep. 5, 14860 (2015).
G. Grochola, S.P. Russo, I. Yarovsky, and I.K. Snook: “Exact” surface free energies of iron surfaces using a modified embedded atom method potential and lambda integration. J. Chem. Phys. 120, 3425–3430 (2004).
G.K. Tripp, K.L. Good, M.J. Motta, P.H. Kass, and C.J. Murphy: The effect of needle gauge, needle type, and needle orientation on the volume of a drop. Vet. Ophthalmol. 19, 38–42 (2016).
T. Li, A.J. Senesi, and B. Lee: Small angle X-ray scattering for nanoparticle research. Chem. Rev. 116, 11128–11180 (2016).
Y. Xu, Y. Qin, S. Palchoudhury, and Y. Bao: Water-soluble iron oxide nanoparticles with high stability and selective surface functionality. Langmuir 27, 8990–8997 (2011).
H. Nakamura and Z. Tamura: Fluorometric determination of secondary amines based on their reaction with fluorescamine. Anal. Chem. 52, 2087–2092 (1980).
D. Eastwood, C. Fernandez, B.Y. Yoon, C.N. Sheaff, and C.M. Wai: Fluorescence of aromatic amines and their fluorescamine derivatives for detection of explosive vapors. Appl. Spectrosc. 60, 958–963 (2006).
M.G. Gore: Spectrophotometry and Spectrofluorimetry: A Practical Approach, 2nd ed. (Oxford University Press, New York, NY, 2000).
J. Puig, C.E. Hoppe, L.A. Fasce, C.J. Perez, Y. Pineiro-Redondo, M. Banobre-Lopez, M.A. Lopez-Quintela, J. Rivas, and R.J.J. Williams: Superparamagnetic nanocomposites based on the dispersion of oleic acid-stabilized magnetite nanoparticles in a diglycidylether of bisphenol a-based epoxy matrix: Magnetic hyperthermia and shape memory. J. Phys. Chem. C 116, 13421–13428 (2012).
M. Kessler: Advanced Topics in Characterization of Composites, 1st ed. (Trafford Publishing, Bloomington, IN, 2004).
X. Gao, J. Shen, Y. Hsia, and Y. Chen: Reduction of supported iron oxide studied by temperature-programmed reduction combined with mossbauer spectroscopy and X-ray diffraction. J. Chem. Soc., Faraday Trans. 89, 1079–1084 (1993).
C. Bolm, J. Legros, J. Le Paih, and L. Zani: Iron-catalyzed reactions in organic synthesis. Chem. Rev. 104, 6217–6254 (2004).
M. Kin, H. Kura, and T. Ogawa: Core loss and magnetic susceptibility of superparamagnetic Fe nanoparticle assembly. AIP Adv. 6, 125013 (2016).
ACKNOWLEDGMENTS
This work was supported by the Laboratory Directed Research and Development program at Sandia National Laboratories. This work was performed, in part, at the Center for Integrated Nanotechnologies, an Office of Science User Facility operated for the U.S. Department of Energy (DOE) Office of Science. Sandia National Laboratories is a multimission laboratory managed and operated by National Technology and Engineering Solutions of Sandia, LLC., a wholly owned subsidiary of Honeywell International, Inc., for the U.S. Department of Energy’s National Nuclear Security Administration under contract DE-NA-0003525. The views expressed in the article do not necessarily represent the views of the U.S. Department of Energy or the United States Government.
Author information
Authors and Affiliations
Corresponding author
Supporting Information for
Rights and permissions
About this article
Cite this article
Watt, J., Bleier, G.C., Romero, Z.W. et al. Gram scale synthesis of Fe/FexOy core–shell nanoparticles and their incorporation into matrix-free superparamagnetic nanocomposites. Journal of Materials Research 33, 2156–2167 (2018). https://doi.org/10.1557/jmr.2018.139
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/jmr.2018.139