Abstract
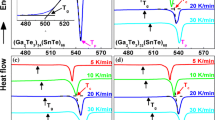
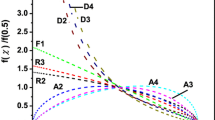
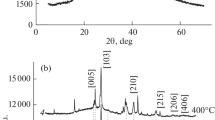
Nonisothermal crystallization kinetics of amorphous chalcogenide Ga–Sb–Te films with compositions along the pseudo-binary tie-lines connecting Sb7Te3-GaSb and Sb2Te3–GaSb of the ternary phase diagram were investigated by means of differential scanning calorimetry. Powder samples were prepared firstly by film deposition using a co-sputtering method; the films were then stripped from the substrate. The activation energy (Ea) and rate factor (Ko) were evaluated from the heating rate dependency of the crystallization temperature using the Kissinger method. The kinetic exponent (n) was deduced from the exothermic peak integrals using the Ozawa method. The crystallization temperature (Tx = 181 to 327 °C) and activation energy (Ea= 2.8 to 6.5 eV) increased monotonically with increasing GaSb content andreached a maximum value in compositions located at the vicinity of GaSb. The kinetic exponent is temperature dependent and shows higher values in the SbTe-rich compositions. Promising media compositions worthy of further studies were identified through the determined kinetics parameters.
Similar content being viewed by others
References
K. Yusu, S. Ashida, N. Nakamura, N. Oomachi, N. Morishita, A. Ogawa, and K. Ichihara: Advanced phase change media for blue laser recording of 18 GB capacity for 0.65 numerical aperture and 30 GB capacity for 0.85 numerical aperture. Jpn. J. Appl. Phys. 42, 858 (2003).
C-M. Lee, W-S. Yen, R-H. Liu, and T-S. Chin: Performance of Ge-Sb–Bi-Te-B recording media for phase-change optical disks. Jpn. J. Appl. Phys. 40, 5321 (2001).
C.M. Lee, T.S. Chin, and E.Y. Huang: Optical properties and structure of tellurium-germanium-bismuth-antimony compounds with fast phase-change capability. J. Appl. Phys. 89, 3290 (2001).
T. Narahara, S. Kobayashi, M. Hattori, Y. Shimpuku, G.J. van den Enden, J.A.H.M. Kahlman, M. van Dijk, and R. van Woudenberg: Optical disc system for digital video recording. Jpn. J. Appl. Phys. 39, 912 (2000).
B. Tieke, M. Dekker, N. Pfeffer, R. van Woudenberg, G-F. Zhou, and I.P.D. Ubbens: High data-rate phase-change media for the digital video recording system. Jpn. J. Appl. Phys. 39, 762 (2000).
C-M. Lee and T-S. Chin: New optical media based on Ge4Sb1-x BixTe5, in Proc. 12th Int. Conf. Ternary and Multinary Compounds. Jpn. J. Appl. Phys. Suppl. 39-1, 513 (2000).
C-M. Lee, T-S. Chin, Y-Y. Huang, I-C. Tung, T-R. Jeng, D-Y. Chiang, and D-R. Huang: Optical properties of Ge40Sb10Te50Bx (x _ 0-2) Films. Jpn. J. Appl. Phys. 38, 6369 (1999).
C-M. Lee, Y-I. Lin, and T-S. Chin: Crystallization kinetics of amorphous Ga-Sb-Te films: Part II. Isothermal studies by a timeresolved optical transmission method. J. Mater. Res. 19, 2938 (2004).
M. Avrami: Kinetics of phase change. I, General theory. J. Chem. Phys. 7, 1103 (1939).
M. Avrami: Kinetics of phase change. II, Transformation-time relations for random distribution of nuclei. J. Chem. Phys. 8, 212 (1940).
M. Avrami: Kinetics of phase change. III, Granulation, phase change, and microstructure. J. Chem. Phys. 9, 177 (1941).
W.A. Johnson and R.F. Mehl: Reaction kinetics in processes of nucleation and growth. Trans. Am. Inst. Min. Metall. Pet. Eng. 135, 416 (1939).
H.E. Kissinger: Reaction kinetics in differential thermal analysis. Anal. Chem. 29, 1702 (1957).
T. Ozawa: Kinetics of non-isothermal crystallization. Polym. 12, 150 (1971).
Y. Li, S.C. Ng, C.K. Ong, H.H. Hng, and T.T. Goh: Glass forming ability of bulk glass forming alloys. Scripta Mater. 36, 783 (1997).
J.W. Christian: The Theory of Transformations in Metals and Alloys, 2nd ed. (Pergamon, Oxford, U.K., 1975), p. 542.
N. Ohshima: Crystallization of germanium-antimony-tellurium amorphous thin film sandwiched between various dielectric protective films. J. Appl. Phys. 79, 8357 (1996).
C.M. Lee and T.S. Chin: (unpublished).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, CM., Lin, YI. & Chin, TS. Crystallization kinetics of amorphous Ga–Sb–Te chalcogenide films: Part I. Nonisothermal studies by differential scanning calorimetry. Journal of Materials Research 19, 2929–2937 (2004). https://doi.org/10.1557/JMR.2004.0378
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.2004.0378