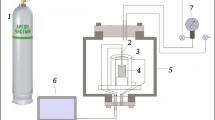
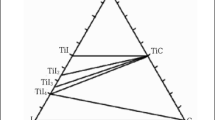
Abstract
Titanium carbonitride, TiC0.5N0.5, is synthesized directly by a self-propagating reaction between titanium and carbon in a nitrogen atmosphere. Complete conversion to the carbonitride phase is achieved with the addition of TiN as diluent and with a nitrogen pressure ≥0.6 MPa. Thermodynamic phase-stability calculations and experimental characterizations of quenched samples support a proposed mechanism in which the formation of the carbonitride is a two-step process. The first step involves the formation of the nonstoichiometric carbide, TiC0.5, and is followed by the formation of the product by the incorporation of nitrogen in the defect-structure carbide to form the carbonitride solid solution.
Similar content being viewed by others
References
L. E. Toth, Transition Metal Carbides and Nitrides (Academic Press, New York, 1971).
H. Pastor, Mater. Sci. Eng. A105/106, 401 (1988).
T. Watanabe, T. Doutsu, K. Shoubu, and Y. Kai, Mater. Sci. Forum 34–36, 561 (1988).
L. Maya, in Better Ceramics Through Chemistry II, edited by C. J. Brinker, D. E. Clark, and D. R. Ulrich (Mater. Res. Soc. Symp. Proc. 73, Pittsburgh, PA, 1986), p. 401.
V. D. Parkhomenko, G. N. Serdyuk, and Yu. I. Krasnokutskii, Fiz. Khim. Obrab. Mater. (Russian) 5, 78–82 (1986).
A. V. Bolotov, V. N. Musolin, A. V. Kolensnikov, and M. N. Filkov, Sixth Symposium on Plasma Chemistry 1, 237–243 (1983).
D. Seyfeth and G. Mignani, Government Report Announcement Index (US) 88 (11), (1988).
M. Yoshimura, M. Nishioka, and S. Sõmiya, I. Mater. Sci. Lett. 6, 463–465 (1987).
V. D. Lyubimov, G. K. Moiseev, and T. A. Timoshchuk, Neorg. Khim. (Russian) 21 (8), 1321–1324 (1985).
O. Matsumoto and H. Taki, Proc. The Electrochemical Society 88–5, 486–493 (1988).
H. Yoshimura, Jpn. Kokai Tokyo Koho J P 61 17, 471 (1986).
I. Zalite, Tezisy Dokl. -Konf. Molodykh Nanchn. Rab. Inst. Neorg. Khim., Akad. Nauk Latv. SSSR 5th (Russian), 22–3 (1976).
P. Grieveson, Proc. The British Ceramic Society 8, 137–153 (1967).
J. B. Holt and Z. A. Munir, J. Mater. Sci. 21 (1), 251–259 (1986).
A. G. Merzhanov and I. P. Borovinskaya, Dok. Akad. Nauk SSSR (Chem. ) 204, 429–432 (1972).
A. I. Kirdyashkin, Y. M. Maksinov, and E. A. Nekrasov, Combust. Explos. Shock Waves 17, 33 (1981).
O. Yamada, Y. Miyamoto, and M. Koizumi, J. Am. Ceram. Soc. 70 (9), C206–C208 (1987).
S. L. Kharatyan, Y. S. Grigor’ev, and A. G. Merzhanov, Combust. Explos. Shock Waves 11, 21–26 (1975).
Z. A. Munir, S. Deevi, and M. Eslamloo-Grami, High Temp. –High Press. 20, 19–24 (1988).
M. Eslamloo-Grami and Z. A. Munir, J. Am. Ceram. Soc. 73 (5), 1235–1239 (1990).
M. Eslamloo-Grami and Z. A. Munir, J. Am. Ceram. Soc. 73 (8), 2222–2227 (1990).
A. B. Avakian, A. R. Bagramian, I. P. Borovinskaya, S. L. Grigorian, and A. G. Merzhanov, in Combustion Process in Chemical Technology and Metallurgy, Chernogolovka, 1975.
Z. A. Munir, Ceram. Bull. 67, 342 (1988).
Z. A. Munir and U. Anselmi-Tamburini, Mater. Sci. Rep. 3, 277–365 (1989).
H. C. Yi and J. J. Moore, J. Mater. Sci. 25, 1159 (1990).
J. B. Holt and S. D. Dunmead, Annu. Rev. Mater. Sci. 21, 305 (1991).
Selected Powder Diffraction Data for Metals and Alloys, Data Book First Edition (JCPDS, 1978), Vol. I, pp. 1-260–19-515.
H. J. Goldschmidt, Interstitial Alloys (Plenum Press, New York, 1967), p. 535.
The CSIRO Thermochemistry System (Version V. l), CSIRO Institute of Energy and Earth Resources, Division of Mineral Chemistry, Melbourne, Australia, 1990.
O. Kubaschewski and C. B. Alcock, Metallurgical Thermochemistry (Pergamon Press, New York, 1979).
JANAF Thermochemical Tables, National Bureau of Standards, NBS-37, Washington, DC, June 1971.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eslamloo-Grami, M., Munir, Z.A. The mechanism of combustion synthesis of titanium carbonitride. Journal of Materials Research 9, 431–435 (1994). https://doi.org/10.1557/JMR.1994.0431
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1557/JMR.1994.0431