Abstract
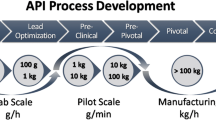
An extended cost study consisting of 14 process scenarios was carried out to envisage the cost impact of microprocessing and microwaves separately or in combination for two liquid-phase model reactions in fine-chemicals synthesis: (1) Ullmann C–O cross-coupling reaction and (2) the aspirin synthesis. The former, a Cu-catalyzed substitution reaction, was based on an experimental investigation, whereas the latter, a noncatalyzed aromatic esterification reaction, was based on literature data. The cost of 4-phenoxypyridine production, as a pharmaceutical intermediate in the synthesis of vancomycin or vancocin, was compared with that of the synthesis of aspirin, a key example of large-scale fine-chemical production plants. The operating costs in the Ullmann synthesis were found to be related to material-based process (reactant excess, pretreatment, and catalyst synthesis), whereas those in the aspirin synthesis appeared to be related to downstream-based process (workup, waste treatment). The impact of an integrated microwave heating and microprocessing system on profitability was demonstrated with respect to operational cost and chemical productivity. Different modes of microwave heating and catalyst supply were studied and compared with conventional oil-bath-heated systems in batch and continuous processes. The overall costs including profitability breakthrough for a competitive market price of product were obtained from various combinations of heating and processing. In case of the Ullmann synthesis, the CAPEX (capital expenditure) was negligible compared to the OPEX (operational expenditure), whereas in the aspirin synthesis, the CAPEX was found around 40%, both at a production scales of 1–10 kg/day using proposed upscale methods. The source of the catalyst strongly determined the profitability of a continuously operated Ullmann process due to its effect on the chemical performance. Higher energy efficiencies could be attained using single-mode microwave irradiation; however, the energy contribution to the overall cost was found to be negligible. Different scenarios provided a cost-feasible and profitable process; nevertheless, an integrated microwave heating and microflow processing led to a cost-efficient system using a micropacked-bed reactor in comparison to wall-coated microreactor, showing a profit margin of 20%.
Article PDF
Similar content being viewed by others
References and Notes
Kappe, C. O.; Stadler, A. In Microwaves in Organic and Medicinal Chemistry; Wiley-VCH Verlag GmbH & Co KGaA: Weinheim, 2006; pp 9–28.
Bogdal, D.; Prociak, A. Chimica Oggi 2007, 25(3), 30–33.
Dallinger, D.; Lehmann, H. r.; Moseley, J. D.; Stadler, A.; Kappe, C. O. Org. Process Res. Dev. 2011, 15(4), 841–854
Lehmann, H.; LaVecchia, L. Org. Process Res. Dev. 2010, 14(3), 650–656
Moseley, J. D. Chimica Oggi 2009, 27(2), 6–10
Moseley, J. D.; Lenden, P.; Lockwood, M.; Ruda, K.; Sherlock, J.-P.; Thomson, A. D.; Gilday, J. P. Org. Process Res. Dev. 2007, 12(1), 30–40
Wolkenberg, S. E.; Shipe, W. D.; Lindsley, C. W.; Guare, J. P.; Pawluczyk, J. M. Curr. Opin. Drug Discovery Dev. 2005, 8(6), 701–708
Loupy, A. Microwaves in Organic Synthesis; Wiley: Weinheim, 2006; Vol. 2
Kremsner, J.; Stadler, A.; Kappe, C. O. Top. Curr. Chem. 2006, 266, 233–278.
Chamoin, S. In Advances in Microwave-Assisted Organic Synthesis: MAOS Conference and Exhibition, Budapest, Hungary, 2006.
Lehmann, H. In Ernst Schering Foundation Symposium Proceedings, Berlin–Heidelberg, Germany, 2007, Vol. 3, pp 133–149.
Kappe, C. O.; Stadler, A., In Microwaves in Organic and Medicinal Chemistry, Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, 2006; pp 57–90.
Marafie, J. A.; Moseley, J. D. Org. Biomol. Chem. 2010, 8(9), 2219–2227
Moseley, J. D.; Woodman, E. K. Org. Process Res. Dev. 2008, 12(5), 967–981
Arvela, R. K.; Leadbeater, N. E.; Collins Jr, M. J. Tetrahedron 2005, 61(39), 9349–9355.
Kelly, C. B.; Lee, C.; Leadbeater, N. E. Tetrahedron Lett. 2011, 52(2), 263–265
Pipus, G.; Plazl, I.; Koloini, T., Chem. Eng. J. 2000, 76(3), 239–245
Wilson, N. S.; Sarko, C. R.; Roth, G. P. Org. Process Res. Dev. 2004, 8(3), 535–538.
Shore, G.; Morin, S.; Organ, M. G. Angew. Chem. Int. Ed. 2006, 45(17), 2761–2766.
Benaskar, F.; Hessel, V.; Krtschil, U.; Löb, P.; Stark, A. Org. Process Res. Dev. 2009, 13(5), 970–982.
Bowman, M. D.; Holcomb, J. L.; Kormos, C. M.; Leadbeater, N. E.; Williams, V. A. Org. Process Res. Dev. 2007, 12(1), 41–57.
Engels, V.; Benaskar, F.; Patil, N.; Rebrov, E. V.; Hessel, V.; Hulshof, L. A.; Jefferson, D. A.; Vekemans, J. A. J. M.; Karwal, S.; Schouten, J. C.; Wheatley, A. E. H. Org. Process Res. Dev. 2010, 14(3), 644–649.
Illg, T.; Loeb, P.; Hessel, V. Bioorg. Med. Chem. 2010, 18(11), 3707–3719.
Hartman, R. L.; Naber, J. R.; Buchwald, S. L.; Jensen, K. F. Angew. Chem. Int. Ed. 2010, 49(5), 899–903
Roberge, D. M.; Gottsponer, M.; Eyholzer, M.; Kockmann, N. Chim. Oggi 2009, 27(4), 4.
Schmalz, D.; Oldenburg, M. H., N.; Grund, M.; Muntermann, H.; Kunz U. Chem. Ing. Tech. 2005, 77(7), 859–866.
Kockmann, N. In Micro Process Engineering; Wiley-VCH Verlag GmbH: Weinheim, 2008; pp 1–45.
Schmink, J. R.; Kormos, C. M.; Devine, W. G.; Leadbeater, N. E. Org. Process Res. Dev. 2010, 14(1), 205–214
Nakamura, T.; Nagahata, R.; Kunii, K.; Soga, H.; Sugimoto, S.; Takeuchi, K. Org. Process Res. Dev. 2010, 14(4), 781–786
Moseley, J. D.; Woodman, E. K. Energy Fuels 2009, 23(11), 5438–5447
Hessel, V.; Kralisch, D.; Krtschil, U. Energy Environ. Sci. 2008, 1(4), 467–478
Glasnov, T. N.; Kappe, C. O. Macromol. Rapid Commun. 2007, 28(4), 395–410
Krtschil, U.; Hessel, V.; Kralisch, D.; Kreisel, G.; Kuepper, M.; Schenk, R. Chimia 2006, 60(9), 6
Comer, E.; Organ, M. G. Chem. Eur. J. 2005, 11(24), 7223–7227
Comer, E.; Organ, M. G. J. Am. Chem. Soc. 2005, 127(22), 8160–8167
Roberge, D. M.; Ducry, L.; Bieler, N.; Cretton, P.; Zimmermann, B. Chem. Eng. Technol. 2005, 28(3), 318–323
Nuchter, M.; Ondruschka, B.; Bonrath, W.; Gum, A. Green Chem. 2004, 6(3), 128–141.
Benaskar, F.; Engels, V.; Patil, N. G.; Chaibi, M.; Rebrov, E. V.; Meuldijk, J.; Hessel, V.; Hulshof, L. A.; Wheatley, A. E. H.; Schouten, J. C. 2011, (Unpublished results)
Dressen, M. H. C. L.; van de Kruijs, B. H. P.; Meuldijk, J.; Vekemans, J. A. J. M.; Hulshof, L. A. Org. Process Res. Dev. 2010, 14(2), 351–361
Drevina, V. M.; Markitanova, L. I.; Nesterov, V. M. Khimiko-Farmatsevticheskii Zhurnal 1976, 10(10), 53–55
Drevina, V. M.; Nesterov, V. M.; Markitanova, L. I. Khimiko-Farmatsevticheskii Zhurnal 1976, 10(11), 120–122
Markitanova, L. I.; Drevina, V. M.; Nesterov, V. M. Khimiko-Farmatsevticheskii Zhurnal 1976, 10(12), 85–88.
Dressen, M. H. C. L. 2009
Wang, L.-D.; Cui, P. Liaoning Huagong 2009, 38(9), 623.
Patil, N. G.; Hermans, A. I. G.; Benaskar, F.; Rebrov, E. V.; Meuldijk, J.; Hessel, V.; Hulshof, L. A.; Schouten, J. C. AIChE Journal: Chemical Engineering Research and Development 2011, (Unpublished results).
Protasova, L. N.; Rebrov, E. V.; Glazneva, T. S.; Berenguer-Murcia, A.; Ismagilov, Z. R.; Schouten, J. C. J. Catal. 2010, 271(2), 161–169
Shore, G.; Tsimerman, M.; Organ, M. G. Beilstein J. Org. Chem. 2009, 5, 35
Glazneva, T.; Rebrov, E.; Schouten, J.; Paukshtis, E.; Ismagilov, Z. Thin Solid Films 2007, 515(16), 6391–6394.
D’Angelo, N. D.; Peterson, J. J.; Booker, S. K.; Fellows, I.; Dominguez, C.; Hungate, R.; Reider, P. J.; Kim, T.-S. Tetrahedron Lett. 2006, 47(29), 5045–5048.
Benaskar, F.; Engels, V.; Rebrov, E. V.; Patil, N. G.; Meuldijk, J.; Magusin, P. C. M. M.; Mezari, B.; Hessel, V.; Hulshof, L. A.; Thüne, P. C.; Hensen, E. J. M.; Wheatley, A. E. H.; Schouten, J. C. Chem. Eur. J. 2010, in press.
Stankiewicz, A. Chem. Eng. Res. Des. 2006, 84(7), 511–521.
Hoogenboom, R.; Wilms, T. F. A.; Schubert, U. S. Polymer Preprints 2008, 49(2), 930–931
Graus, W. H. J.; Voogt, M.; Worrell, E. Energy Policy 2007, 35(7), 3936–3951.
After personal discussion with Lauda Company, pump energy for internal circulation of heating medium was determined.
In this study, the minimum heating medium volume required for uniform and stable heating was experimentally found to be around fourfold heating medium volume regarding the reaction mixture. At lower heat medium volumes, temperature fluctuations and high temperature gradients result. In conventional lab-scale systems, a much higher volume ratio of medium and reaction mixture is used.
Gronnow, M. J.; White, R. J.; Clark, J. H.; Macquarrie, D. J. Org. Process Res. Dev. 2005, 9(4), 516–518
Simmons, H. E.; Smith, R. D., J. Am. Chem. Soc. 1958, 80(19), 5323–5324.
Moseley, J. D.; Kappe, C. O. Green Chem. 2011, 13(4), 794–806
Nüchter, M.; Müller, U.; Ondruschka, B.; Tied, A.; Lautenschläger, W. Chem. Eng. Technol. 2003, 26(12), 1207–1216.
Strauss, C. R. Org. Process Res. Dev. 2009, 13(5), 915–923.
Hoogenboom, R.; Wilms, T. F. A.; Erdmenger, T.; Schubert, U. S. Aust. J. Chem. 2009, 62(3), 236–243.
Razzaq, T.; Kappe, C. O. ChemSusChem 2008, 1(1–2), 123–132.
Godwin, D. R.; Lawton, S. J.; Moseley, J. D.; Welham, M. J.; Weston, N. P. Energy Fuels 2010, 24(10), 5446–5453.
Bruggink, A. Chim. Oggi 1998, 16, 44–47
Bruggink, A. In CphI Conference Proceedings 1993, 38–47
Berkoff, C. E.; Kamholz, K.; Rivard, D. E.; Wellman, G.; Winicov, H. Chemtech 1986, 1986, 552–559.
Benaskar, F.; Engels, V.; Patil, N.; Rebrov, E. V.; Meuldijk, J.; Hessel, V.; Hulshof, L. A.; Jefferson, D. A.; Schouten, J. C.; Wheatley, A. E. H. Tetrahedron Lett. 2010, 51(2), 248–251.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Benaskar, F., Ben-Abdelmoumen, A., Patil, N.G. et al. Cost Analysis for a Continuously Operated Fine Chemicals Production Plant at 10 Kg/Day Using a Combination of Microprocessing and Microwave Heating. J Flow Chem 1, 74–89 (2011). https://doi.org/10.1556/jfchem.2011.00015
Published:
Issue Date:
DOI: https://doi.org/10.1556/jfchem.2011.00015