Abstract
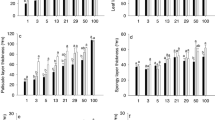
After clearcutting xerothermic oakwoods once natural in the forest-steppe loess regions of Hungary, the perennial understorey grass Brachypodium pinnatum has been persisting for decades by establishing microhabitats from shade to full sun. In this paper, we explore variation in leaf anatomy for plants growing in different microhabitat light regimes (full shade under oak canopy, half shade near shrubs, and in unshaded grassland) in situ, and for plants reciprocally transplanted between these microhabitats. Leaf lamina thickness and mesophyll thickness were about 1.5 times greater in the grassland in situ than in oak subcanopy due to an additional layer of mesophyll cells and to 25–32% taller mesophyll cells. Mesophyll thickness and the proportion of veins plus sclerenchyma were lower for plants transplanted from either full or half shade to full sun than in situ plants in the grassland. Parenchymatous bundle sheath tended to be thicker in the grassland than in the two other microhabitats. Mean intervenial distance remained invariable among microsites. These adjustments in leaf anatomy may be a considerable part, but presumably not the dominant component of the medium-term (one year) light acclimation of B. pinnatum and the species’ success in microsites with contrasting light climate appearing side-by-side during secondary vegetation succession.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ETR:
-
Electron transport rate
- LMA:
-
Leaf mass per area
References
Boardman, N. K. 1977. Comparative photosynthesis of sun and shade plants. Annu. Rev. Plant Physiol. 28:355–377.
Bobbink, R. 1991. Effects of nutrient enrichment in Dutch chalk grassland. J. Appl. Ecol. 28:28–41.
Bobbink, R. and J. H. Willems. 1987. Increasing dominance of Brachypodium pinnatum (L.) Beauv. in chalk grasslands: a threat to a species-rich ecosystem. Biol. Conserv. 40:301–314.
Castro-Díez, P., J. P. Puyravaud and J. H. C. Cornelissen. 2000. Leaf structure and anatomy as related to leaf mass per area variation in seedlings of a wide range of woody plant species and types. Oecologia 124:476–486.
Chazdon, R. L. and S. Kaufmann. 1993. Plasticity of leaf anatomy of two rain forest shrubs in relation to photosynthetic light acclimation. Funct. Ecol. 7:385–394.
Choong, M. F., P. W. Lucas, J. S. Y. Ong, B. Pereira, H. T. W. Tan and I. M. Turner. 1992. Leaf fracture toughness and sclerophylly: their correlations and ecological implications. New Phytol. 121:597–610.
De Kroon, H. and J. Knops. 1990. Habitat exploration through morphological plasticity in two chalk grassland perennials. Oikos 59:39–49.
De Kroon, H. and F. Schieving. 1990. Resource partitioning to clonal growth in relation to clonal growth strategy. In: J. Van Groenendael and H. De Kroon (eds.), Clonal Growth in Plants: Regulation and Function. SPB Acad. Publ., The Hague. pp. 79–94.
Fekete, G. and J. Szujkó-Lacza. 1973. Leaf anatomical and photosynthetical reactions of Quercus pubescens Willd. to environmental factors in various ecosystems. I. Leaf anatomical reactions. Acta Bot. Acad. Sci. Hung. 18(1–2):59–89.
Fekete, G., K. Virágh, R. Aszalós and L. Orlóci. 1998. Landscape and coenological differentiation of Brachypodium pinnatum grasslands in Hungary. Coenoses 13(1):39–53.
Fekete G., K. Virágh, R. Aszalós and I. Précsényi. 2000. Static and dynamic approaches to landscape heterogeneity in the Hungarian forest-steppe zone. J. Veg. Sci. 11:375–382.
Garnier, E. and G. Laurent. 1994. Leaf anatomy, specific mass and water content in congeneric annual and perennial grass species. New Phytol. 128:725–736.
Givnish, J. T. 1988. Adaptation to sun and shade: a whole-plant perspective. Aust. J. Plant Physiol. 15:63–92.
GraphPad Software. 2000. GraphPad Instat version 3.05 for Windows 95/NT. GraphPad Software, Inc., San Diego, California, USA, https://doi.org/www.graphpad.com.
Hlwatika, C. N. M. and R. B. Bhat. 2002. An ecological interpretation of the difference in leaf anatomy and its plasticity in contrasting tree species in Orange Kloof, Table Mountain, South Africa. Ann. Bot. 89:109–114.
Hurst, A. and E. John. 1999. The biotic and abiotic changes associated with Brachypodium pinnatum dominance in chalk grassland in South-East England. Biol. Conserv. 88:75–84.
Klich, M. G. 2000. Leaf variations in Elaeagnus angustifolia related to environmental heterogeneity. Environ. Exp. Bot. 44:171–183.
Lambers, H., F. S. Chapin III and T. L.Pons. 1998. Plant Physiological Ecology. Springer-Verlag, New York.
Laboratory Imaging. 1997. Lucia M 3.52a image analysis software. Laboratory Imaging Ltd, Praha, Czech Republic.
Mendes, M. M., L. C. Gazarini and M. L. Rodrigues. 2001. Acclimation of Myrtus communis to contrasting Mediterranean light environments – effects on structure and chemical composition of foliage and plant water relations. Environ. Exp. Bot. 45:165–178.
Mojzes, A., T. Kalapos and K. Virágh. 2003. Plasticity of leaf and shoot morphology and leaf photochemistry for Brachypodium pinnatum (L.) Beauv. growing in contrasting microenvironments in a semiarid loess forest-steppe vegetation mosaic. Flora 198:304–320.
Molnár, E., I. Bagi, Zs. Csintalan and A. Nyakas. 2000. The invasion success of a native grassland species in the Great Hungarian Plain. In: D. Ferenciková, N. Gáborcik, L. Ondrasek, E. Uhlrarová and M. Zimková (eds.), Grassland Ecology V. Proceedings of the 5th Ecological Conference, 23–25 November 1999. Grassland and Mountain Agriculture Research Institute, Banská Bystrica. pp. 423–433.
Niinemets, Ü. 1999. Components of leaf dry mass per area – thickness and density – alter leaf photosynthetic capacity in reverse directions in woody plants. New Phytol. 144:35–47.
Niinemets, Ü. and J. D. Tenhunen. 1997. A model separating leaf structural and physiological effects on carbon gain along light gradients for the shade-tolerant species Acer saccharum. Plant, Cell Environ. 20:845–866.
Oguchi, R., K. Hikosaka and T. Hirose. 2003. Does the photosynthetic light-acclimation need change in leaf anatomy? Plant, Cell Environ. 26:505–512.
Sage, R. F. 2001. Environmental and evolutionary preconditions for the origin and diversification of the C4 photosynthetic syndrome. Plant biol. 3:202–213.
Sims, D. A. and R. W. Pearcy. 1992. Response of leaf anatomy and photosynthetic capacity in Alocasia macrorrhiza (Araceae) to a transfer from low to high light. Am. J. Bot. 79(4):449–455.
Utrillas, M. J. and L. Alegre. 1997. Impact of water stress on leaf anatomy and ultrastructure in Cynodon dactylon (L,) Pers, under natural conditions. Int. J. Plant Sci. 158(3):313–324.
Van Arendonk, J. J. C. M. and H. Poorter. 1994. The chemical composition and anatomical structure of leaves of grass species differing in relative growth rate. Plant, Cell Environ. 17:963–970.
Virágh, K. and S. Bartha. 1998. Interspecific associations in different successional stages of Brachypodium pinnatum grassland after deforestation in Hungary. Tiscia 31:3–12.
Zólyomi, B. and G. Fekete. 1994. The Pannonian loess steppe: differentiation in space and time. Abstracta Botanica 18(1):29–41.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Mojzes, A., Kalapos, T. & Virágh, K. Leaf anatomical plasticity of Brachypodium pinnatum (L.) Beauv. growing in contrasting microenvironments in a semiarid loess forest-steppe vegetation mosaic. COMMUNITY ECOLOGY 6, 49–56 (2005). https://doi.org/10.1556/ComEc.6.2005.1.6
Published:
Issue Date:
DOI: https://doi.org/10.1556/ComEc.6.2005.1.6