Abstract
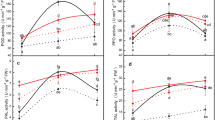
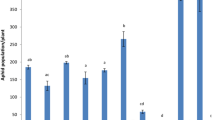
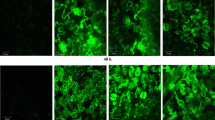
The oxidation of fatty acids, which is responsible for changes in lipid composition, were investigated in a comparative study using the Russian wheat aphid (RWA) (Diuraphis noxia), infested and un-infested resistant (Tugela DN) and close isogenic-susceptible (Tugela) wheat cultivars. LOX, which catalyzes the first step of the lipoxygenase pathway, was selectively induced in the infested resistant wheat. A pathogen-induced oxygenase protein was also found to be induced during the wheat-RWA interaction. The involvement of oxylipins in the RWA resistance response was confirmed by inhibition studies using indomethacin, which is known to inhibit prostanoid biosynthesis in mammalian tissue. Downstream defense reactions, e.g. LOX and POD activities, were inhibited upon indomethacin treatment. These results emphasize the importance of fatty acid oxidation as an essential process for the establishment of a successful defense response in wheat to the RWA.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Andreou, A., Brodhum, F., Feussner, I. 2009. Biosynthesis of oxylipins in non-mammals. Prog. Lipid Res. 48:148–170.
Blée, M. 2002. Impact of phyto-oxylipins in plant defence. Trends Plant Sci 7:315–322.
Bohland, C., Balkenhohl, T., Loers, G., Feussner, I., Grambow, H.J. 1997. Differential induction of lipoxygenase isoforms in wheat upon treatment with rust fungus elicitor, chitin oligosaccharides, chitosan and methyl jasmonate. Plant Physiol. 114:679–685.
Bradford, M.M. 1976. A rapid and sensitive method for the quantification of micron quantities of protein utilizing the principle of protein dye binding. Anal. Biochem. 72:248–254.
Catalá, A. 2009. Lipid peroxidation of membrane phospholipids generates hydroxy-alkenals and oxidized phospholipids active in physiological and/or pathological conditions. Chem. Phys. Lipids 157:1–11.
Du Toit, F. 1989. Inheritance in two Triticum aestivum lines to Russian wheat aphid (Homo: aphididae). J. Econ. Entomol. 82:1251–1253.
Gardner, H.W. 1991. Recent investigations into the lipoxygenase pathway of plants. Biochem. Biophys. Acta 1084:221–239.
Göbel, C., Feussner, I., Schmidt, A., Scheel, D., Sanchez-Serrano, J., Hanberg, M., Rosahl, S. 2001. Oxylipin profiling reveals the preferential stimulation of the 9-lipoxygenase pathway in elicitor-treated potato cells. J. Biol. Chem. 276:6267–6273.
Grossmann, S., Zakut, R. 1979. Determination of lipoxygenase (lipoxydase). Meth. Biochem. Anal. 25:303–329.
Haley, S.D., Peairs, F.B., Walker, C.B., Rudolph, J.B., Randolph, T.L. 2004. Occurrence of a new Russian wheat aphid biotype in Colorado. Crop Sci. 44:1589–1592.
Hamberg, M., Samuelson, B. 1967. On the specificity of the oxygenation of unsaturated fatty acids catalysed by soybean lipoxidase. J. Biol. Chem. 242:5329–5335.
Hannun, Y.A., Obeid, L.M. 2008. Principles of bioactive lipid signalling: Lessons from sphingolipids. Nature Rev. Mol. Cell. Biol. 9:139–150.
Harizi, H., Corcuff, J-B., Gualde, N. 2008. Arachidonic-acid-derived eicosanoids: Roles in biology and immunopathology. Trends Mol. Med. 14:461–469.
Heath, R.L., Parker, L. 1968. Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 125:89–198.
Hofer, M., Pospíšil, M., Hoferová, Z., Weiterová, L., Komůrková, D. 2012. Stimulatory action of cyclooxygenase inhibitors on Hematopoiesis: A review. Molecules 17:5615–5625.
Khattab, H. 2007. The defence mechanism of cabbage plant against phloem-sucking aphid (Brevicoryne brassicae L.). Aus. J. Basic and Appl. Sci. 1:56–62.
Kim, Y-C., Yi, S-Y., Mang, H.G., Seo, Y.S., Kim, WT, Choi, D. 2002. Pathogen-induced expression of cyclo-oxygenase homologue in hot pepper (Capsicum annuum cv. Pukang). J. Exp. Bot. 53:383–385.
Kuhn, H., Sprecher, H., Brash, A.R. 1990. On singular or dual positional specificity of lipoxygenases. The number of chiral products varies with alignment of methylene groups at the active site of the enzyme. J. Biol. Chem. 265:16300–16305.
Laemmli, U.K. 1970. Cleavage of structure proteins during the assembly of the head bacteriophage T4. Nature 227:680–685.
Leone, A., Melillo, M.T., Bleve-Zacheo, T. 2001. Lipoxygenase in pea roots subjected to biotic stress. Plant Sci. 161:703–717.
Montillet, J-L., Agnel, J.P., Ponchet, M., Vailleau, F., Roby, D., Triantaphylidès, C. 2002. Lipoxygenase-mediated production of fatty acid hydroperoxides is a specific signature of the hypersensitive reaction in plants. Plant Physiol. Biochem. 40:633–639.
Narumiya, S., Furuyashiki, T. 2011. Fever, inflammation, pain and beyond: Prostanoid receptor research during these 25 years. The FASEB Journal 25:813–818.
Nicolaou, K.C., Ramphal, J.Y., Petasis, N.A., Serhan, M.H. 1991. Lipoxins and related eicosanoids: Biosynthesis, biological properties, and chemical synthesis. Angew. Chem. Int. Ed. Engl. 30:1100–1116.
Ntambi, J.M. 2005. Polyunsaturated fatty acid regulation of genes of lipid metabolism. Annu. Rev. Nutr. 25:317–340.
Ocampo, C.A., Moerschbacher, B.M., Grambow, H.J. 1986. Increased lipoxygenase activity is involved in the hypersensitive response of wheat leaf cells infected with the avirulent rust fungi or treated with fungal elicitor. Z. Naturforsch. 41c:559–563.
Polkowska-Kowalczyk, L., Montilletb, J-L., Agnelb, J-P., Triantaphylides, B.C., Wielgat, B., Maciejewska, U. 2008. Changes in the initial phase of lipid peroxidation induced by elicitor from Phytophthora infestans in Solanum species. J. Plant Physiol. 165:1929–1939.
Prinsloo, G.J. 2000 Host and host instar preference of Aphelinus sp. nr. varipes (Hymenoptera: Aphelinidae) in South Africa. Afr. Entomol. 8:57–61.
Quick, J.S., Ellis, G.E., Normann, R.M., Stormberger, J.A., Shanahan, J.F., Peairs, F.B., Lorenz, K. 1996. Registration of ‘Halt’ wheat. Crop Sci. 36:210.
Rancé, I., Fourier, J., Esquerré-Tugayé, M-T. 1998. The incompatible interaction between Phytophthora parasitica var. nicotianae race 0 and tobacco is suppressed in transgenic plants expressing antisense lipoxygenase sequences.. Proc. Natl Acad. Sci. USA 95:6554–6559.
Rao, M.V., Paliyath, G., Ormond, D.G., Murr, D.P., Watkins, C.B. 1997. Influence of salicylic acid on H2 O2 production, oxidative stress, and H2 O2-mediating enzymes. Plant Physiol. 115:137–149.
Rustérucci, C., Montillet, J-L., Agnel, J-P., Battesti, C., Alonso, B., Knoll, A., Bessoules, J-J., Etienne, P., Suty, L., Blein, J-P., Triantaphylides, C. 1999. Involvement of lipoxygenase-dependent production of fatty acid hydroperoxides in the development of the hypersensitive cell death induced by cryptogein on tobacco leaves. J. Biol. Chem. 274 51:36446–36455.
Rybutt, D.B., Parish, C.R. 1982. Protein determination on an automatic spectrophotometer. Anal. Biochem. 121:213–214.
Sanz, A., Moreno, J.I., Castresana, C. 1998. PIOX, a new pathogen-induced oxygenase with homology to animal pathogen-inducible oxygenases. The Plant Cell 10:1523–1537.
Shah, J. 2005. Lipids, lipases, and lipid-modifying enzymes in plant disease resistance. Annu. Rev. Phytopathol. 43:229–260.
Shimizu, T., Radmark, O., Samuelsson, B. 1984. Enzyme with dual lipoxygenases activities catalyses leukotriene A4 synthesis from arachidonic acid. Proc. Natl Acad. Sci. USA 81:689–693.
Tolmay, V.L., Lindeque, R.C., Prinsloo, G.J. 2007. Preliminary evidence of a resistance-breaking biotype of the Russian wheat aphid Diuraphis noxia (Kurdjumov) (Homoptera: Aphididae), in South Africa. Afr. Entomol. 15:228–230.
Tsitsigiannis, D.I., Keller, N.P. 2007 Oxylipins as developmental and host-fungal communication signals. Trends in Microbiol. 15:109–118.
Vick, B.A. 1993. Oxygenated fatty acids of the lipoxygenase pathway. In: Moore, T.S. (ed.), Lipid Metabolism in Plants. CRC Press, Boca Raton, FL, USA, pp. 167–191.
Wasternack, C. 2007. Jasmonates: An update on biosynthesis, signal transduction and action in plant stress response, growth and development. Ann. Bot. 100:681–697.
Weber, H. 2002. Fatty acid-derived signals in plants. Trends in Plant Sci. 7:217–224.
Zieslin, N., Ben-Zaken, R. 1991. Peroxidase, phenylalanine ammonia-lyase and lignification in peduncles of rose flowers. Plant Physiol. Biochem. 29:147–151.
Zoeller, M., Stingl, N., Krischke, M., Fekete, A., Waller, F., Berger, S., Mueller, M. 2012. Lipid profiling of the Arabidopsis hypersensitive response reveals lipid peroxidation and fragmentation processes: Biogenesis of pimelic and azelic acid. Plant Physiol. 160:365–378.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Goyal
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Berner, J.M., van der Westhuizen, A.J. Oxidation of Fatty Acids are Associated with Russian Wheat Aphid Resistance in Wheat. CEREAL RESEARCH COMMUNICATIONS 43, 84–96 (2015). https://doi.org/10.1556/CRC.2014.0031
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/CRC.2014.0031