Abstract
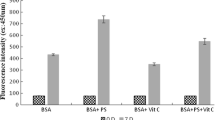
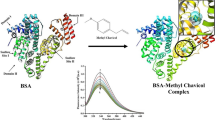
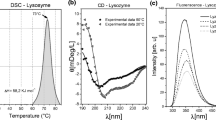
The effects of the mycotoxin patulin on the thermodynamics and kinetics of the transition of bovine serum albumin (BSA) in aqueous solution were studied by Differential Scanning Calorimetry and Photo-luminescence methods. Results show that in the presence of patulin, the free enthalpy change during the transition of BSA was decreased by an average of ~46 kJ/mol, the free energy change was decreased by ~4 kJ/mol, and the activation energy fell from ~1546 to -840 kJ/mol. These results indicate that the bioactivity of patulin is based on the kinetic rather than on the thermodynamic properties of the transition. This is the first evidence of the direct interaction of patulin with the free thiol-containing BSA, a process which could contribute to the adverse cyto- and genotoxic effects induced by patulin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BSA:
-
bovine serum albumin
- PL:
-
photoluminescence
- T m :
-
transition temperature
- ΔHcal:
-
enthalpy
- PAT:
-
patulin
References
Benesi, H., Hildebrand, J. H. (1949) A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. Am. Chem. Soc. 71, 2703–2707.
Bennett, J. W., Klich, M. (2003) Mycotoxins. Clin. Microbiol. Rev. 16, 497–516.
Carter, D. C., Ho, J. X. (1994) Advances in protein chemistry. Academic Press Inc., San Diego.
Chiti, F., Taddei, N., Baronim, F., Capanni, C., Stefani, M., Ramponi, G., Dobson, C. M. (2002) Kinetic partitioning of protein unfolding and aggregation. Nat. Struct. Biol. 9, 137–143.
Dombrink-Kurtzman, M. A., Blackburn, J. A. (2005) Evaluation of several culture media for production of patulin by Penicillium species. Int. J. Food Microbiol. 98, 241–248.
Fliege, R., Metzler, M. (1999) The mycotoxin patulin induces intra- and intermolecular protein crosslinks in vitro involving cysteine, lysine, and histidine side chains, and a-amino groups. Chem. Biol Interact. 123, 85–103.
Francis, G. L. (2010) Albumin and mammalian cell culture: implications for biotechnology applications. Cytotechnology 62, 1–16.
Giancola, C., De Sena, C., Fessas, D., Graziano, G., Barone, G. (1997) DSC studies on bovine serum albumin denaturation. Effects of ionic strength and SDS concentration. Int. J. Biol. Macromol. 20, 193–204.
Horváth, E., Papp, G., Belágyi, J., Gazdag, Z., Vágvölgyi, C., Pesti, M. (2010) In vivo direct patulin-induced fluidization of the plasma membrane of fission yeast Schizosaccharomyces pombe. Food and Chem. Toxicol. 48, 1898–1904.
Iwahashi, Y., Hosoda, H., Park, J. H., Lee, J. H., Suzuki, Y., Kitagawa, E., Murata, S. M., Jwa, N. S., Gu, M. B., Iwahashi, H. (2006) Mechanisms of patulin toxicity under conditions that inhibit yeast growth. J. Agric. Food Chem. 54, 1936–1942.
Kissinger, H. E. (1957) Reaction kinetics in differential thermal analysis. Anal. Chem. 29, 1702–1706.
Kunsági-Máté, S., Iwata, K. (2009) Effect of cluster formation of solvent molecules on the preferential solvatation of anthracene in binary alcoholic solutions. Chem. Phys. Lett. 473, 284–287.
Kunsági-Máté, S., Kumar, A., Sharma, P., Kollár, L., Nikfardjam, M. P. (2009) Effect of molecular environment on the formation kinetics of complexes of malvidin-3-O-glucoside with caffeic acid and catechin. J. Phys. Chem. B113, 7468–7473.
Kunsági-Máté, S., Lecomte, S., Ortmann, E., Kunsági-Máté, E., Desbat, B. (2010) The environment controlled effect of thiacalix[4]arene on the transition thermodynamics and kinetics of bovine serum albumin. J. Incl. Phenom. Macro. Chem. 66, 147–151.
Kunsági-Máté, S., Csók, Zs., Iwata, K., Szász, E., Kollár, L. (2011) Role of the conformational freedom of the skeleton in the complex formation ability of resorcinarene derivatives toward a neutral phenol guest. J. Phys. Chem. B115, 3339–3343.
Moriyama, Y., Watanabe, E., Kobayashi, K., Harano, H., Inui, E., Takeda, K. (2008) Secondary structural change of bovine serum albumin in thermal denaturation up to 130 °C and protective effect of sodium dodecyl sulfate on the change. J. Phys. Chem. B112, 16585–16589.
Murillo, M., Gonzáles-Penas, E., Amezqueta, S. (2008) Determination of patulin in commercial apple juice by micellar electrokinetic chromatography. Food Chem. Toxicol. 46, 57–64.
Privalov, P. L., Gill, S. J. (1988) Stability of protein structure and hydrophobic interaction. Advances in Protein Chemistry 39, 191–234.
Privalov, P. L., Khechinashvili, N. N. (1974) A thermodynamic approach to the problem of stabilization of globular protein structure: A calorimetric study. J. Mol. Biol. 86, 665–684.
Privalov, P. L., Dragan, A. I. (2007) Microcalorimetry of biological macromolecules. Biophys. Chem. 126, 16–24.
Relkin, P., Mulvihill, D. M. (1996) Thermal unfolding of β-lactoglobulin, α-lactalbumin and bovine serum albumin. A thermodynamic approach. Critical Rev. Food Sci. and Nutrition 36, 565–601.
Ricelli, A., Baruzzi, F., Solfrizzo, M., Morea, M., Fanizzi, F. P. (2007) Biotransformation of patulin by Gluconobacter oxydons. Appl. Environ. Microbiol. 73, 785–792.
Schumacher, D. M., Metzler, M., Lehmann, L. (2005) Mutagenicity of the mycotoxin patulin in cultured Chinese hamster V79 cells, and its modulation by intracellular glutathione. Arch. Toxicol. 79, 110–121.
Tapia, M. O., Stern, M. D., Koski, R. L., Bach, A., Murphy, M. J. (2002) Effects of patulin on microbial fermentation in continuous culture fermenters. Animal Feed Sci. and Techn. 97, 239–246.
Tong, G. C. (2003) Characterization of cysteine-34 in serum albumin. Thesis. The Ohio State Univ.
Virág, E., Pesti, M., Kunsági-Máté, S. (2010) Competitive hydrogen bonds associated with the effect of primycin antibiotic on oleic acid as a building block of plasma membranes. J. Antib. 63, 113–117.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Horváth, E., Kálmán, N., Pesti, M. et al. Thermodynamic and Kinetic Processes during the Unfolding of BSA in the Presence of the Mycotoxin Patulin. BIOLOGIA FUTURA 63, 389–398 (2012). https://doi.org/10.1556/ABiol.63.2012.3.9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.63.2012.3.9