Abstract
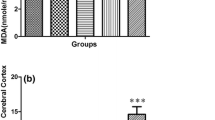
Our aim was to evaluate the effects of marijuana (Mar) and diazepam (Dz) on lipid peroxidation (TBARS), Na+, K+ ATPase activity, levels of glutathione (GSH) and 5-hydroxytryptophan (5-HTP). Male Wistar rats were given a single dose per group: extract of Mar (100 μL/kg), Dz (5 mg/kg), Mar plus Dz, and NaCl for control. Sixty mins after treatment, animals were sacrificed, and their brains extracted and homogenised to measure GSH, TBARS and 5-HTP levels. Na+, K+ ATPase and total ATPase activities. GSH and TBARS did not show differences respect to controls. Na+, K+ ATPase activity was similar as well. However, groups treated with Mar, total ATPase activity decreased significantly (p < 0.05). Levels of 5-HTP decreased significantly (p = 0.0001) in rats treated either with Mar and or Dz. Mar and Dz induced biochemical effects on the serotonergic metabolism, which can alter the development and function in rat brain, because it has also been involved in scavenging free radicals present there.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beckman, J. S., Beckman, T. W., Chen, J., Marshall, P. A., Freeman, B. A. (1994) Apparent hydroxyl radical production by peroxynitrite: Implications for endothelial injury from nitric oxide and superoxides. Proc. Natl. Acad Sci. USA. 87, 1629–1634.
Ben de Kruijff (1987) Polymorphic regulation of membrane lipid composition. Nature 329, 587–588.
Brase, D. A. (1990) Is intracellular sodium involved in the mechanism of tolerance to opioid drugs? Med. Hypotheses 32, 161–167.
Calderon, G. D., Toledo, L. A., Hernandez, I. J., Barragan, M. G., Rodriguez, P. R., Hernandez, G. E. (2001) Determination de triptofano en cerebro de ratas expuestas a ozono. Arch. Neurosci. (Mex) 6, 2–5.
Calderon, G. D., Hernandez, I. J., Espitia, V. I., Barragan, M. G., Hernandez, G. E., Juarez Olguin, H. (2004) Pyridoxine, regardless of serotonin levels, increases production of 5-hydroxytryptophan in rat brain. Arch. Med. Res. 35, 271–274.
Calderón, G. D., Espitia, V. I., López, D. A., Hernández, G. E., Huerta, G. B., Juarez Olguin, H. (2005) Effect of toluene and nutritional status on serotonin, lipid peroxidation levels and Na+/K+-ATPase in adult rat brain. Neurochem. Res. 30, 619–624.
Ebadi, M., Govitrapong, P., Phansuwan-Pujito, P., Nelson, F., Reiter, R. J. (1998) Pineal opioid receptors and analgesic action of melatonin. J. Pineal Res. 24, 193–200.
Enrico, P., Mura, M. A., Esposito, G., Serra, P., Migheli, R. (1998) Effect of naloxone on morphine-induced changes in striatal dopamine metabolism and glutamate, ascorbic acid and uric release in freely moving rats. Brain Res. 797, 94–102.
Fernstrom, J. D., Wurtman, R. J. (1972) Brain serotonin content: Physiological regulation by plasma neutral amino acids. Science 178, 414–416.
Fiske, C. H., Subbarow, Y. (1925) The colorimetric determination of phosphorus. J. Biol. Chem. 66, 375–400.
Garcia, C. A., Serrano, M., Perez, D. B., Gonzalez, V., Ormazabal, A., Pineda, M. (2007) Secondary abnormalities of neurotransmitters in infants with neurological disorders. Dev. Med. Child Neurol. 49, 740–744.
Goudas, L. C., Carr, D. B., Maszczynska, I., Marchand, J. E., Wurm, W. H., Greenblatt, D. J. (1997) Differential effect of central versus parenteral administration of morphine sulfate on regional concentrations of reduced glutathione in rat brain. Pharmacology 54, 92–97.
Goudas, L. C., Langlade, A., Serrie, A., Matson, W., Milbury, P., Thurel, C. (1999) Acute decreases in cerebrospinal fluid glutathione levels after intracerebroventricular morphine for cancer pain. Anesth. Analg. 89, 1209–1215.
Gutteridge, M. C., Halliwell, B. (1990) The measurement and mechanism of lipid peroxidation in biological systems. Trends Biochem. Sci. 5, 129–135.
Halford, J. C., Harrold, J. A., Boyland, E. J., Lawton, C. L., Blundell, J. E. (2007) Serotonergic drugs: effects on appetite expression and use for the treatment of obesity. Drugs 67, 27–55.
Hampson, A. J., Grimaldi, M., Axelrod, J., Wink, D. (1998) Cannabidiol and (-) D9-tetrahydrocanna-binol are neuroprotective antioxidants. Proc. Natl. Acad Sci. USA. 95, 8268–8273.
Hissin, P. J., Hilf R. (1976) A fiuorometric method for determination of oxidized and reduced glutathione in tissue. Anal. Biochem. 74, 214–226.
Jhamandas, J. H., Harris, K. H., Petrov, T., Jhamandas, K. H. (1996) Activation of nitric oxide-synthesizing neurones during precipitated morphine withdrawal. Neuroreport 7, 2843–2846.
Keromov, B. F. (2004) Glutathione-deficient state of nervous tissues in starved animals intensifies lipid peroxidation and oxidation of protein SH-groups. Ukr Biokhim. Zh 76, 108–113.
Lue, W. M., Su, M. T., Lin, W. B., Tao, P. L. (1999) The role of nitric oxide in the development of morphine tolerance in rat hippocampal slices. Eur. J. Pharmacol. 383, 129–135.
Masocha, W., González, L. G., Baeyens, J. M., Agil, A. (2002) Mechanisms involved in morphine-induced activation of synaptosomal Na+, K+-ATPase. Brain Res. 957, 311–319.
Masocha, W., Horvath, G., Agil, A., Ocaña, M., Pozo, E., Szikszay, M. (2003) Role of Na+, K+-ATPase in morphine-induced antinociception. J. Pharmacol. Exp. Ther 306, 1122–1128.
Muñoz, C. J., Montilla, P., Padillo, F. J., Bujalance, I., Muñoz, M. C., Muntané, J. (2006) Role of serotonin in cerebral oxidative stress in rats. Acta Neurobiol. Exp. 66, 1–6.
Ossipov, M. H., Lai, J., King, T., Vanderah, T. W., Porreca, F. (2005) Underlying mechanisms of pronociceptive consequences of prolonged morphine exposure. Biopolymers 80, 319–324.
Pillai, N. P., Ross, D. H. (1986) Effects of opiates on high-affinity Ca2+, Mg2+-ATPase in brain membrane subfractions. J. Neurochem. 47, 1642–1646.
Puppala, B. L., Matwyshyn, G., Bhalla, S., Gulati, A. (2004) Evidence that morphine tolerance may be regulated by endothelin in the neonatal rat. Biol. Neonate 86, 138–144.
Prunet, M. B., Desbazeille, M., Bros, A., Louche, K., Delagrange, P., Renard, P. (2003) Melatonin reduces body weight gain in sprague dawley rats with diet-induced obesity. Endocrinology 144, 5347–5352.
Sadee, W., Wang, D., Bilsky, E. J. (2005) Basal opioid receptor activity, neutral antagonists, and therapeutic opportunities. Life Sci. 76, 1427–1437.
Sakabe, T., Dahlgren, N., Carlsson, A., Siesjö, B. K. (1982) Effect of diazepam on cerebral monoamine synthesis during hypoxia and hypercapnia in the rat. Physiol. Scand. 115, 57–65.
Saner, A., Pletscher, A. (1979) Effect of diazepam on cerebral 5-hydroxytryptamine synthesis. Eur. J. Pharmacol. 55, 315–318.
Seamon, M. J., Fass, J. A., Maniscalco-Feichtl, M., Abu-Shraie, N. A. (2007) Medical marijuana and the developing role of the pharmacist. Am. J. Health Sys. Pharm. 64, 1037–1044.
Sethi, B. B., Trivedi, J. K., Kumar, P., Gulati, A., Agarwal, A. K., Sethi, N. (1996) Antianxiety effect of cannabis: involvement of central benzodiazepine receptors. Biol. Psychiatry 21, 3–10.
The Statistical Discovery Software SDS, Version 7.0 by SAS Institute Inc, 2003.
Sun, Y. (1990) Free radicals, antioxidants enzymes and carcinogenesis. Free Rad Biol. Med. 8, 583–599.
Wan-Kan, O., Hosein, E. A. (1981) Synaptosomal Na+, K+ ATPase as a membrane probe in studying the in vivo action of morphine. Can. J. Biochem. 59, 687–692.
Wall, M. E., Brine, D. R., Perez, R. M. (1976) Metabolism of cannabinoids in man. In: Braude, M. C., Szara, S. (eds) Pharmacol of Marijuana. Raven Press, New York, pp. 93–116.
Wickelgren, I. (1997) Marijuana: harder than throught? Science 276, 1967–1968.
Acknowledgement
We thank to Dr Radames Alemon for helping us to translate the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Calderón, G.D., Esquivel, G.J., García, E.H. et al. Effects of Marijuana and Diazepam on Lipid Peroxidation, Na+, K+ Atpase, and Levels of Glutathione and 5-HTP in rat Brain. BIOLOGIA FUTURA 61, 135–144 (2010). https://doi.org/10.1556/ABiol.61.2010.2.2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/ABiol.61.2010.2.2