Abstract
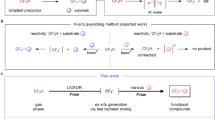
Herein, we present the development of a continuous-flow process for a difluoromethylation with difluorocarbene as the reactive reagent. The difluoromethylated product is a key intermediate during the synthesis of eflornithine, a pharmaceutical that is on the World Health Organization’s Model List of Essential Medicines. The developed procedure uses inexpensive and commercially available chlorodifluoromethane (CHF2Cl, Freon 22) as difluorocarbene source. Deprotonation of CHF2Cl with NaOH in a biphasic mixture of organic solvent-water generates the carbene. A fast subsequent reaction of the difluorocarbene with the substrate generates the desired product with excellent selectivity.
Similar content being viewed by others
References
Organofluorine Compounds in Medicinal Chemistry & Biomedical Applications, R. Filler; Z. Kobayashi; L. M. Yagupolskii, Eds.; Elsevier: Amsterdam, 19
Meyskens, F. L.; Gerner, E. W. Jr. Clin. Cancer Res. 1999, 5, 945–951
Furet, Y.; Duong, T. H.; Combescot, C.; Breteau, M. Pathol. Biol. 1987, 35, 398–404.
Mettler, H.; Greth, E. (Lonza AG), 2-Difluormethyl-2-cyanethylmalonsäureniedrigalkylester, CH672124A5, Oct. 1989.
Ni, C.; Hu, J. Synthesis 2014, 46, 842–863
Brahms, D. L. S.; Dailey, W. P. Chem. Rev. 1996, 96, 1585–1632.
Hydrochlorofluorocarbons, such as CHF2Cl, have high global warming and ozone depletion potential. Their production and consumption are controlled under the Montreal Protocol.
Shen, T. Y.; Lucas, S.; Sarett, L. H. Tetrahedron Lett. 1961, 2, 43–47
Kosuge, S.; Nakai, H.; Kurono, M. Prostaglandins 1979, 18, 737–743
Tsushima, T.; Kawada, K.; Ishihara, S.; Uchida, N.; Shiratori, O.; Hiagaki, J.; Hirata, M. Tetrahedron 1988, 44, 5375–5387
McDonald, I. A.; Lacoste, J. M.; Bey, P.; Palfreyman, M. G.; Zreika, M. J. Med. Chem. 1985, 28, 186–193
McDonald, I. A.; Bey, P. Tetrahedron Lett. 1985, 26, 3807–3810
Bey, P.; Fozard, J.; Lacoste, J. M.; McDonald, I. A.; Zreika, M.; Palfreyman, M. G. J. Med. Chem. 1984, 27, 9–10.
Bey, P.; Schirlin, D. Tetrahedron Lett. 1978, 52, 5225–5228
Seki, M.; Suzuki, M.; Matsumoto, K. Biosci. Biotechnol. Biochem. 1993, 57, 1024–1025
Bey, P.; Gerhart, F.; Dorsselaer, V. V.; Dansin, C. J. Med. Chem. 1983, 26, 1551–1556
Schirlin, D.; Gerhart, F.; Hornsperger, J. M.; Hamon, M.; Wagner, J.; Jung, M. J. J. J. Med. Chem. 1988, 31, 30–36
Tsushima, T.; Kawada, K.; Shiratori, O.; Uchida, N. Heterocycles 1985, 23, 45–49
Nishide, K.; Kobori, T.; Tunemoto, D.; Kondo, K. Heterocycles 1987, 26, 633–640.
Bey, P.; Vevert, J.-P. Tetrahedron Lett. 1978, 14, 1215–1218
Bey, P.; Vevert, J.-P.; Dorsselaer, V. V.; Kolb, M. J. Org. Chem. 1979, 44, 2732–2742.
Tsushima, T.; Kawada, K. Tetrahedron Lett. 1985, 26, 2445–2448.
Jończyk, A.; Nawrot, E.; Kisielewski, M. J. Fluorine Chem. 2005, 126, 1587–1591.
Nawrot, E.; Jonczyk, A. J. Fluorine Chem. 2006, 127, 943–947.
Nawrot, E.; Jonczyk, A. J. Fluorine Chem. 2009, 130, 466–469.
The advantages of continuous-flow processes were thoroughly reviewed: (a) Wiles, C.; Watts, P. Green Chem. 2014, 16, 55–62
Ley, S. V.; Fitzpatrick, D. E.; Ingham, R. J.; Myers, R. M. Angew. Chem., Int. Ed. 2015, 54, 3449–3464
Gutmann, B.; Cantillo, D.; Kappe, C. O. Angew. Chem., Int. Ed. 2015, 54, 6688–6728
Hessel, V.; Kralisch, D.; Kockmann, N.; Noël, T.; Wang, Q. ChemSusChem. 2013, 6, 746–789
Pastre, J. C.; Browne, D. L.; Ley, S. V. Chem. Soc. Rev. 2013, 42, 8849–8869
McQuade, D. T.; Seeberger, P. H. J. Org. Chem. 2013, 78, 6384–6389
Movsisyan, M.; Delbeke, E. I. P.; Berton, J. K. E. T.; Battilocchio, C.; Ley, S. V.; Stevens, C. V. Chem. Soc. Rev. 2016, 45, 4892–4928.
Dehmlow, E. V. Angew. Chem., Int. Ed. Engl. 1974, 13, 170–179.
The application of continuous-flow microreactors for organic synthesis under biphasic conditions has been previously demonstrated. For selected examples, see: (a) Ahmed, B.; Barrow, D.; Wirth, T. Adv. Synth. Catal. 2006, 348, 1043–1048
Damm, M.; Gutmann, B.; Kappe, C. O. ChemSusChem 2013, 6, 978–982
Plouffe, P.; Roberge, D. M.; Macchi, A. Chem. Eng. J. 2016, 300, 9–19
Reichart, B.; Kappe, C. O.; Glasnov, T. N. Synlett 2013, 24, 2393–2396
Illg, T.; Hessel, V.; Löb, P.; Schouten, J. C. Chem. Eng. J. 2011, 167, 504–509.
http://www.ehrfeld.com; Ehrfeld Mikrotechnik BTS GmbH homepage (accessed on June 9th of 2017).
Mielke, E.; Roberge, D. M.; Macchi, A. J. Flow Chem. 2016, 6, 279–287.
http://www.uniqsis.com; Uniqsis Ltd homepage (accessed on June 9th of 2017).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Gutmann, B., Hanselmann, P., Bersier, M. et al. Continuous-Flow Difluoromethylation with Chlorodifluoromethane under Biphasic Conditions. J Flow Chem 7, 46–51 (2017). https://doi.org/10.1556/1846.2017.00005
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/1846.2017.00005