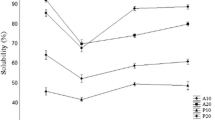
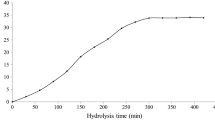
Abstract
The housefy is an important resource insect and the housefy larvae are ideal source of food additives. The housefy larvae protein hydrolysates were obtained by enzymatic hydrolysis by alcalase and neutral proteinase. Their antioxidant activities were investigated, including the superoxide and hydroxyl radical-scavenging activity, 1,1-diphenyl-2-picrylhydrazyl (DPPH) scavenging activity, reducing power and metal chelating activity. The antioxidant activities of both hydrolysates increased with their increasing concentrations. The alcalase hydrolysate (AH) showed higher scavenging activities against hydroxyl radical and superoxide anion radical at low concentrations and higher metal-chelating activity than the neutral proteinase hydrolysate (NPH). The NPH exhibited higher scavenging activity against DPPH free radical and higher reducing power than the AH. Both hydrolysates showed more than 50% superoxide anion radical-scavenging activity at 10 μg/mL. These results indicate that both housefy larvae protein hydrolysates display high antioxidant activities and they could serve as potential natural antioxidant food additives.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beckman, K. B., Ames, B. N. (1998) The free radical theory of aging matures. Physiol. Rev. 78, 547–581.
Smith, M. A., Perry, G., Richey, P. L., Sayre, L. M., Anderson, V. E., Beal, M. F., Kowall, N. (1996) Oxidative damage in Alzheimer’s. Nature 382, 20–121.
Lin, C. C., Liang, J. H. (2002) Effect of antioxidants on the oxidative stability of chicken breast meat in a dispersion system. J. Food Sci. 67, 530–533.
Wang, B., Pace, R. D., Dessai, A. P., Bovell-Benjamin, A., Phillips, B. (2002) Modifed extraction method for determining 2-thiobarbituric acid values in meat with increased specifcity and simplicity. J. Food Sci. 67, 2833–2836.
Yu, L., Haley, S., Perret, J., Harris, M., Wilson, J., Qian, M. (2002) Free radical scavenging properties of wheat extracts. J. Agric. Food Chem. 50, 1619–1624.
Quian, Z., Jung, W., Kim, S. (2008) Free radical scavenging activity of a novel antioxidative peptide purifed from hydrolysate of bullfrog skin, Rana catesbeina Shaw. Bioresour. Technol. 99, 1690–1698.
Giménez, B., Alemán, A., Montero, P., Gómez-Guillén, M. C. (2009) Antioxidant and functional properties of gelatin hydrolysates obtained from skin of sole and squid. Food Chem. 114, 976–983.
Klompong, V., Benjakul, S., Kantachote, D., Shahidi, F. (2007) Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as infuenced by the degree of hydrolysis and enzyme type. Food Chem. 102, 1317–1327.
Sakanaka, S., Tachibana, Y., Ishihara, N., Juneja, L. J. (2004) Antioxidant activity of egg-yolk protein hydrolysates in a linoleic acid oxidation system. Food Chem. 86, 99–103.
Wang, H., Gao, X. D., Zhou, G. C., Cai, L., Yao, W. B. (2008) In vitro and in vivo antioxidant activity of aqueous extract from Choerospondias axillaris fruit. Food Chem. 106, 888–895.
Yu, H. H., Liu, X. G., Xing, R., Liu, S., Guo, Z. Y., Wang, P. B., Li, C. P., Li, P. C. (2006) In vitro determination of antioxidant activity of proteins from jellyfsh Rhopilema esculentum. Food Chem. 95, 123–130.
Zhu, K. X., Zhou, H. M., Qian, H. F. (2006) Antioxidant and free radical-scavenging activities of wheat germ protein hydrolysates (WGPH) prepared with alcalase. Process. Biochem. 41, 1296–1302.
Hao, Y. J., Jing, Y. J., Qu, H., Li, D. S., Du, R. Q. (2008) Purifcation and characterization of a thermal stable antimicrobial protein from housefy larvae, Musca domestica, induced by ultrasonic wave. Acta Biol. Hung. 59, 289–304.
Jing, Y. J., Hao, Y. J., Qu, H., Shan, Y., Li, D. S., Du, R. Q. (2007) Studies on the antibacterial activities and mechanisms of chitosan obtained from cuticles of housefy larvae. Acta Biol. Hung. 58, 75–86.
Ai, H., Wang, F. R., Yang, Q. S., Zhu, F., Lei, C. L. (2008) Preparation and biological activities of chitosan from the larvae of housefy, Musca domestica. Carbohydr. Polym. 72, 419–423.
Hou, L. X., Shi, Y. H., Zhai, P., Le, G. W. (2007) Antibacterial activity and in vitro anti-tumor activity of the extract of the larvae of the housefy (Musca domestica). J. Ethnopharmacol. 111, 227–231.
Schägger, H. (2006) Tricine-SDS-PAGE. Nat. Protoc. 1, 16–22.
Moore, S. (1963) On the determination of cystine and cysteic acid. J. Biol. Chem. 238, 235–237.
Halliwell, B., Gutteridge, J. M. C., Aruoma, O. I. (1987) The deoxyribose method: A simple “testtube” assay for determination of rate constants for reactions of hydroxyl radical. Anal. Biochem. 165, 215–219.
Oyaizu, M. (1986) Studies on products of browning reactions: antioxidative activities of products of browning reaction prepared from glucosamine. Jap. J. Nutr. 44, 307–315.
Decker, E. A., Welch, B. (1990) Role of ferritin as a lipid oxidation catalyst in muscle food. J. Agric. Food Chem. 38, 674–677.
Chen, H. M., Muramoto, K., Yamauchi, F., Nokihara K. (1996) Antioxidant activity of designed peptides based on the antioxidative peptide isolated from digests of a soybean protein. J. Agric. Food Chem. 44, 2619–2623.
Kim, S. K., Kim, Y. T., Byun, H. G., Nam, K. S., Joo, D. S., Shahidi, F. (2001) Isolation and characterization of antioxidative peptides from gelatin hydrolysate of Alaska pollack skin. J. Agric. Food Chem. 49, 1984–1989.
Mendis, E., Rajapakse, N., Byun, H. G., Kim, S. K. (2005) Investigation of jumbo squid (Dosidicus gigas) skin gelatin peptides for their in vitro antioxidant effects. Life Sci. 70, 2166–2178.
Aruoma, O. I. (1998) Free radicals oxidative stress, and antioxidants in human health and disease. J. Am. Oil Chem. Soc. 75, 199–211.
Aurand, L. W., Boonme, N. H., Gidding, G. G. (1977) Superoxide and singlet oxygen in milk lipid peroxidation. J. Dairy Sci. 60, 363–369.
Gülçin, Ì., Şat, İ. G., Beydemir, Ş., Elmastaş, M., Küfrevioğlu, Ö. İ. (2004) Comparison of antioxidant activity of clove (Eugenia caryophylata Thunb) buds and lavender (Lavandula stoechas L.). Food Chem. 87, 393–400.
Mokbel, M. S., Hashinaga, F. (2006) Evaluation of the antioxidant activity of extracts from buntan (Citrus grandis Osbeck) fruit tissues. Food Chem. 94, 529–534.
Shimada, K., Fujikawa, K., Yahara, K., Nakamura, T. (1992) Antioxidative properties of xanthan on the antioxidation of soy bean oil in cyclodextrin emulsion. J. Agric. Food Chem. 40, 945–948.
Wang, Y. G., Zhu, F. R., Han, F. S., Wang, H. Y. (2008) Purifcation and characterization of antioxidative peptides from salmon protamine hydrolysate. J. Food Biochem. 32, 654–671.
Saiga, A., Tanabe, S., Nishimura, T. (2003) Antioxidant activity of peptides obtained from porcine myofbrillar proteins by protease treatment. J. Agric. Food Chem. 51, 3661–3667.
Dong, S., Zeng, M., Wang, D., Liu, Z., Zhao, Y., Yang, H. (2008) Antioxidant and biochemical properties of protein hydrolysates prepared from Silver carp (Hypophthalmichthys molitrix). Food Chem. 107, 1485–1493.
Samaranayaka, A. G. P., Li-Chan, E. C. Y. (2008) Autolysis-assisted production of fsh protein hydrolysates with antioxidant properties from Pacifc hake (Merluccius productus). Food Chem. 107, 768–776.
Nalinanon, S., Benjakul, S., Kishimura, H., Shahidi, F. (2011) Functionalities and antioxidant properties of protein hydrolysates from the muscle of ornate threadfn bream treated with pepsin from skipjack tuna. Food Chem. 124, 1354–1362.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, H., Wang, P., Zhang, AJ. et al. Antioxidant Activities of Protein Hydrolysates Obtained from the Housefly Larvae. BIOLOGIA FUTURA 67, 236–246 (2016). https://doi.org/10.1556/018.67.2016.3.2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/018.67.2016.3.2