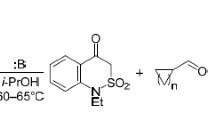
Abstract
Dodecanoyl isothiocyanate (I) reacts additively with anthranilic acid to afford derivatives of thiourea II and benzothiazine IIIin a one-pot reaction. The cyclisation of thiourea II was achieved using acetic anhydride to form quinazoline derivative IV. The heating of quinazoline IV in acetic anhydride or butan-1-ol gave quinazoline derivatives V or VI, respectively. Benzothiazine III underwent trans-acylation to benzothiazine VII in boiling acetic anhydride. The treatment of IV with hydrazine hydrate, anthranilic acid or ethyl carbazate afforded derivatives of triazoloquinazoline VIII, quinazolinoquinazoline XI or thiosemicarbazide X, respectively. The reaction of I with 2-aminophenol or 2-aminothiophenol afforded thiourea derivative XIII or benzothiazole derivative XIV, respectively. Most of the synthesised compounds bear a lauroyl (dodecanoyl) group (a hydrocarbon moiety). The structures of the synthesised compounds were confirmed by microanalytical and spectral data.
Similar content being viewed by others
References
Alagarsamy, V., Pathak, U. S. (2007). Synthesis and an-tihypertensive activity of novel 3-benzyl-2-substituted-3H-[1,2,4]triazolo[5,1-b]quinazolin-9-ones. Bioorganic & Medicinal Chemistry, 15, 3457–3462. DOI: 10.1016/j.bmc.2007.03. 007.
Alagarsamy, V., Murugesan, S., Dhanabal, K., Murugan, M., de Clercq, E. (2007). AntiHIV, antibacterial and an-tifungal activities of some novel 2-methyl-3-(substituted methylamino)-(3H)-quinazolin-4-ones. Indian Journal of Pharmaceutical Sciences, 69, 304–307. DOI: 10.4103/0250-474x.33167.
Balzarini, J., Van Daele, I., Negri, A., Solaroli, N., Karls-son, A., Liekens, S., Gago, F., & Van Calenbergh, S. (2009). Human mitochondrial thymidine kinase is selectively inhibited by 3’-thiourea derivatives of /3-thymidine: Identification of residues crucial for both inhibition and catalytic activity. Molecular Pharmacology, 75, 1127–1136. DOI: 10.1124/mol.108.053785.
Barluenga, J., Tomás, M., Ballesteros, A., & López, L. A. (1994). A simple approach to pyrimidine and quinazoline derivatives by [4+2] cycloaddition of 1,3-diazadienes and enamines. Heterocycles, 37, 1109–1120. DOI: 10.3987/com-93-s123.
Bukvic Krajacic, M., Peric, M., Smith, K. S., Ivezic Schönfeld, Z., Ziher, D., Fajdetic, A., Kujundžic, N., Schönfeld, W., Lan-dek, G., Padovan, J., Jelic, D., Ager, A., Milhous, W. K., Ellis, W., Spaventi, R., Ohrt, C. (2011). Synthesis, structure-activity relationship, and antimalarial activity of ureas and thioureas of 15-membered azalides. Journal of Medicinal Chemistry, 54, 3595–3605. DOI: 10.1021/jm2001585.
Butin, A. V., Tsiunchik, F. A., Abaev, V. T., Gutnov, A. V., Cheshkov, D. A. (2009). Aryl ring migration reaction in the synthesis of 2,4-diaryl-4H-3,1-benzothiazines. Synthesis, 2009, 2616–2626. DOI: 10.1055/s-0029-1217399.
Butler, K., Partridge, M. W. (1959). Cyclic amidines. Part VIII. Derivatives of 12H-6:7:12a-triazabenz[a]anthracene and 5aH-5:6:11a-triazanaphthacene. Journal of Chemical Society, 1959, 1512–1521.
Canon, K. K. (1984). Japan Patent No. JP59197051. Tokyo, Japan: Japan Patent Office.
Chandrika, P. M., Yakaiah, T., Rao, A. R. R., Narsaiah, B., Reddy, N. C., Sridhar, V., Rao, J. V. (2008). Synthesis of novel 4,6-disubstituted quinazoline derivatives, their anti-inflammatory and anti-cancer activity (cytotoxic) against U937 leukemia cell lines. European Journal of Medicinal Chemistry, 43, 846–852. DOI: 10.1016/j.ejmech.2007.06.010.
Cohen, E., Klarberg, B., Vaughan, J. R., Jr. (1960). Quina-zolinone sulfonamides. A new class of diuretic agents. Journal of the American Chemical Society, 82, 2731–2735. DOI: 10.1021/ja01496a020.
Ding, Q., Liu, X., Yu, J., Zhang, Q., Wang, D., Cao, B., Peng, Y. (2012). Access to functionalized 4-benzylidene-4H-benzo[d][1,3]thiazines via tandem addition-cyclization/cross-coupling reactions. Tetrahedron, 68, 3937–3941. DOI: 10. 1016/j.tet.2012.03.098.
Ding, Q., Lin, Y., Ding, G., Liao, F., Sang, X., & Peng, Y. Y. (2013). New simple synthesis of ring-fused 4-alkyl-4H-3,1-benzothiazine-2-thiones: Direct formation from carbon disulfide and (E)-3-(2-aminoaryl)acrylates or (E)-3-(2-aminoaryl)acrylonitriles. Beilstein Journal of Organic Chemistry, 9, 460–466. DOI: 10.3762/bjoc.9.49.
El-Bordany, E. A. (2012). Addition-cyclization of lauroyl isoth-iocyanate with hydrazine derivatives as a source of 1,2,4-triazoles. Middle-East Journal of Scientific Research, 11, 266–271.
El-Hiti, G. A., Hussain, A., Hegazy, A. S., Alotaibi, M. H. (2011). Thioxoquinazolines: synthesis, reactions and biological activities. Journal of Sulfur Chemistry, 32, 361–395. DOI: 10.1080/17415993.2011.601417.
El-Sayed, R., Khairou, K. S. (2015). Propoxylated fatty thi-azole, pyrazole, triazole, and pyrrole derivatives with antimicrobial and surface activity. Journal of Surfactants and Detergents, 18, 661–673. DOI: 10.1007/s11743-015-1684-8.
Fahmy, A. F., Ali, N., Abdelhamid, H., Shiba, S., Hemdan, M. M. (2010). The utility of p-N-succinimidobenzoyl isoth-iocyanate in synthesis of benzoxazole, quinazoline, pyrimi-dine, 1,2,4-triazoline, 1,3-thiazolidine, and thiourea derivatives. Phosphorus, Sulfur, and Silicon and the Related Elements, 185, 1536–1542. DOI: 10.1080/10426500903127557.
Gažák, R., Purchartová, K., Marhol, P., Zivná, L., Sedmera, P., Valentová, K., Kato, N., Matsumura, H., Kaihatsu, K., & Kren, V. (2010). Antioxidant and antiviral activities of silybin fatty acid conjugates. European Journal of Medicinal Chemistry, 45, 1059–1067. DOI: 10.1016/j.ejmech.2009.11.056.
Ghorab, M. M., Ismail, Z. H., Abdalla, M., Radwan, A. A. (2013). Synthesis, antimicrobial evaluation and molecular modelling of novel sulfonamides carrying a biologically active quinazoline nucleus. Archives of Pharmacal Research, 36, 660–670. DOI: 10.1007/s12272-013-0094-6.
Gimbert, C., Vallribera, A. (2009). A straightforward synthesis of benzothiazines. Organic Letters, 11, 269–271. DOI: 10.1021/ol802346r.
Grover, G., & Kini, S. G. (2006). Synthesis and evaluation of new quinazolone derivatives of nalidixic acid as potential antibacterial and antifungal agents. European Journal of Medicinal Chemistry, 41, 256–262. DOI: 10.1016/j.ejmech.2005.09.002.
Gütschow, M., Schlenk, M., Gäb, J., Paskaleva, M., Alnouri, M. W., Scolari, S., Iqbal, J., Müller, C. E. (2012). Ben-zothiazinones: A novel class of adenosine receptor antagonists structurally unrelated to xanthine and adenine derivatives. Journal of Medicinal Chemistry, 55, 3331–3341. DOI: 10.1021/jm300029s.
Hadj Salem, J., Humeau, C., Chevalot, I., Harscoat-Schiavo, C., Vanderesse, R., Blanchard, F., Fick, M. (2010). Effect of acyl donor chain length on isoquercitrin acylation and biological activities of corresponding esters. Process Biochemistry, 45, 382–389. DOI: 10.1016/j.procbio.2009.10.012.
Hayao, S., Havera, H. J., Strycker, W. G., Leipzig, T. J., Kulp, R. A., Hartzler, H. E. (1965). New sedative and hypoten-sive 3-substituted 2,4(1H,3H)-quinazolinediones. Journal of Medicinal Chemistry, 8, 807–811. DOI: 10.1021/jm00330a 017.
Hemdan, M. M., Fahmy, A. F., Ali, N. F., Hegazi, E., Abd-Elhaleem, A. (2008). Synthesis of some new heterocycles derived from phenylacetyl isothiocyanate. Chinese Journal of Chemistry, 25, 388–391. DOI: 10.1002/cjoc.200890074.
Hemdan, M. M. (2010). Synthesis and antimicrobial activities of some heterocyclic systems from 2-furoyl isothiocyanate. Phosphorus, Sulfur, and Silicon and the Related Elements, 185, 620–627. DOI: 10.1080/10426500902893209.
Hemdan, M. M., Fahmy, A. F., El-Sayed, A. A. (2010). Synthesis and antimicrobial study of 1,2,4-triazole, quinazoline and benzothiazole derivatives from 1-naphthoylisothiocya-nate. Journal of Chemical Research, 34, 219–221. DOI: 10.3184/030823410x12707543946812.
Hemdan, M. M., Fahmy, A. F. M., Aly, N. F., Hegazi, I. A., El-Sayed, A. A. (2012). Utility of phthalimidoacyl isoth-iocyanate in synthesis of quinazolines, benzoxazoles, benz-imidazoles, 1,2,4-triazoles, and oxatriazepines. Phosphorus, Sulfur, and Silicon and the Related Elements, 187, 181–189. DOI: 10.1080/10426507.2011.597804.
Hemdan, M. M., El-Sayed, A. A. (2015). Synthesis of some new heterocycles derived from novel 2-(1,3-dioxisoindolin-2-yl)benzoyl isothiocyanate. Journal of Heterocyclic Chemistry. DOI: 10.1002/jhet.2287. (in press)
Hemdan, M. M., Abd El-Mawgoude, H. K. (2015a). Uses of 1-(3-cyano-4,5,6,7-tetrahydrobenzo[b]-thiophen-2-yl)-3-dode-canoylthiourea as a building block in the synthesis of fused pyrimidine and thiazine systems. Chemical and Pharmaceutical Bulletin, 63, 450–456. DOI: 10.1248/cpb.c15-00047.
Hemdan, M. M., Abd El-Mawgoude, H. K. (2015b). Synthesis and antimicrobial evaluation of thieno[2,3-d]-pyrimidine, thieno[2/,3/:4,5]pyrimido[1,2-a][1,3,5]triazine, thieno[2,3-d]-1,3-thiazine and 1,2,4-triazole systems. Chemical and Pharmaceutical Bulletin, 63, 812–818. DOI: 10.1248/cpb.c15-00463.
Khalil, A. A., Abdel Hamide, S. G., Al-Obaid, A. M., El-Subbagh, H. I. (2003). Substituted quinazolines, part 2. Synthesis and in-vitro anticancer evaluation of new 2-substituted mercapto-3H-quinazoline analogs. Archiv der Pharmazie, 336, 95–103. DOI: 10.1002/ardp.200390011.
Kidwai, M., Kukreja, S., Rastogi, S., Singhal, K. (2007). Microwave accelerated multicomponent synthesis for a novel scaffold of monastrol analogues. Letters in Organic Chemistry, 4, 357–361. DOI: 10.2174/157017807781212085.
Laddha, S. S., Bhatnagar, S. P. (2008). Rapid microwave-assisted solution phase synthesis of 6,8-disubstituted 2-phenyl-3-(substituted benzothiazol-2-yl)-4-[3H] quinazolino-nes as novel anticonvulsants. Phosphorus, Sulfur, and Silicon and the Related Elements, 183, 2262–2273. DOI: 10.1080/10426500801957766.
Leistner, S., Michael Gütschow, M., & Stach, J. (1990). Mehrcyclische Azine mit Heteroatomen in 1- und 3-Stellung, 25. Mitt.: 2-Amino-4-oxo-4H-3,1-benzothiazine: Darstellung, Dimroth-Umlagerung zu 4-Oxo-2-thioxo-1,2,3,4-tetrahydro-chinazolinen und MS/MS-Fragmentierung. Archiv der Pharmazie, 323, 857–862. DOI: 10.1002/ardp.19903231009. (in German)
Maggio, B., Daidone, G., Raffa, D., Plescia, S., Mantione, L., Cutuli, V. M. C., Mangano, N. G., Caruso, A. (2001). Synthesis and pharmacological study of ethyl 1-methyl-5-(substituted 3,4-dihydro-4-oxoquinazolin-3-yl)-1H-pyrazole-4-acetates. European Journal of Medicinal Chemistry, 36, 737–742. DOI: 10.1016/s0223-5234(01)01259-4.
Mosaad, S. M., Mohammed, K. I., Ahmed, M. A., AbdelHamide, S. G. (2004). Synthesis of certain new 6-iodoquin azolines as potential antitubercular agents. Journal of Applied Sciences, 4, 302–307 DOI: 10.3923/jas.2004.302.307.
Obayashi, T., Okawa, A. (2001). Japan Patent No. JP200125 3172. Tokyo, Japan: Japan Patent Office.
Okuda, K., Zhang, Y. X., Ohtomo, H., Hirota, T., Sasaki, K. (2010). Polycyclic N-heterocyclic compounds. Part 62: Reaction of N-(quinazolin-4-yl)amidine derivatives with hydrox-ylamine hydrochloride and anti-platelet aggregation activity of the products. Chemical and Pharmaceutical Bulletin, 58, 369–374. DOI: 10.1248/cpb.58.369.
Raffa, D., Edler, M. C., Daidone, G., Maggio, B., Merick-ech, M., Plescia, S., Schillaci, D., Bai, R., & Hamel, E. (2004). Synthesis, cytotoxicity, and inhibitory effects on tubulin polymerization of a new 3-heterocyclo substituted 2-styrylquinazolinones. European Journal of Medicinal Chemistry, 39, 299–304. DOI: 10.1016/j.ejmech.2003.12.009.
Roopan, S. M., Maiyalagan, T., & Khan, F. N. (2008). Solvent-free syntheses of some quinazolin-4(3H)-ones derivatives. Canadian Journal of Chemistry, 86, 1019–1025. DOI: 10.1139/v08-149.
Schleiss, M., Eickhoff, J., Auerochs, S., Leis, M., Abele, S., Rechter, S., Choi, Y., Anderson, Y., Scott, G., Rawlin-son, W., Michel, D., Ensminger, S., Klebl, B., Stamminger, T., & Marschall, M. (2008). Protein kinase inhibitors of the quinazoline class exert anti-cytomegaloviral activity in vitro and in vivo. Antiviral Research, 79, 49–61. DOI: 10.1016/j.antiviral.2008.01.154.
Sharma, S. K., Wu, Y., Steinbergs, N., Crowley, M. L., Hanson, A. S., Casero, R. A., Jr., Woster, P. M. (2010). (Bis)urea and (bis)thiourea inhibitors of lysine-specific demethylase 1 as epigenetic modulators. Journal of Medicinal Chemistry, 53, 5197–5212. DOI: 10.1021/jm100217a.
Shestakov, A. S., Prezent, M. A., Zlatoustovskaya, E. O., Shikhaliev, K. S., Falaleev, A. V., Sidorenko, O. E. (2015). Alkylation of 1,3-benzothiazin-4-one 2-oxo-, 2-arylimino-, and 2-thioxo derivatives. Chemistry of Heterocyclic Compounds, 51, 370–376. DOI: 10.1007/s10593-015-1709-2.
Simerpreet, Cannoo, D. S. (2013). Synthesis and biological evaluation of 1,3-thiazines — a review. Pharmacophore, 4, 70–88.
Uher, M., Berkeš, D., Leško, J., Floch, I. (1983). Reactions of carbonyl isothiocyanates with nucleophilic bifunctional reagents. Collection of Czechoslovak Chemical Communications, 48, 1651–1658. DOI: 10.1135/cccc19831651.
Yadav, L. D. S., Rai, V. K., Yadav, B. S. (2009). The first ionic liquid-promoted one-pot diastereoselective synthesis of 2,5-diamino-/2-amino-5-mercapto-1,3-thiazin-4-ones using masked amino/mercapto acids. Tetrahedron, 65, 1306–1315. DOI: 10.1016/j.tet.2008.12.050.
Yavari, I., Nematpour, M., & Hossaini, Z. (2010). Ph3P-mediated one-pot synthesis of fu nctionalized 3,4-dihydro-2H-1,3-thiazines from N,N-dialkylthioureas and activated acetylenes in water. Monatshefte für Chemie - Chemical Monthly, 141, 229–232. DOI: 10.1007/s00706-009-0247-y.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hemdan, M.M., El-Bordany, E.A. Use of dodecanoyl isothiocyanate as building block in synthesis of target benzothiazine, quinazoline, benzothiazole and thiourea derivatives. Chem. Pap. 70, 1117–1125 (2016). https://doi.org/10.1515/chempap-2016-0042
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/chempap-2016-0042