Abstract
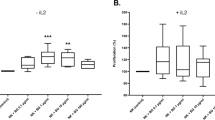
β-Glucan is a natural carbohydrate, which is produced by the variety of different organisms - bacteria, fungi, plants, etc. β-Glucan from different sources has been recognized as an active material, which is an immune stimulator for plants, invertebrates and mammals. Saccharomyces cerevisiae, the baker’s yeast, is one of the commonly used sources of β-1,3-glucan. The aim of the present work was to investigate how the different S. cerevisiae β-glucan preparations affect proliferation, phagocytosis and cytokine production of murine macrophages and dendritic cells. In our experiments, BALB/c macrophages and dendritic cells were treated with different β-glucan preparations in vitro. Then cell proliferation (AlamarBlue reagent), ability to phagocytose zymosan particles and production of IL-12 and IFN7 (Western blot) were investigated. Our results showed that β-glucan preparations stimulated proliferation of BALB/c macrophages and dendritic cells in vitro, but acted on phagocytosis and cytokine synthesis in different ways. This study demonstrated that S. cerevisiae β-glucan preparations propelled proliferation of the murine macrophages and dendritic cells and influenced phagocytosis and cytokine production of these cells. These effects can depend on the size of β-glucan molecules.
Similar content being viewed by others
Abbreviations
- CR3:
-
complement receptor-3
- DC:
-
dendritic cell
- IN:
-
water-insoluble S. cerevisiae β-glucan preparation
- LPS:
-
lipopolysaccharide
- PAMP:
-
pathogen-associated molecular pattern
- PBS:
-
phosphate-buffered saline
- S1–S5:
-
soluble S. cerevisiae β-glucan preparations.
References
Abbas A.K., Lichtman A.H. & Pillai S. 2015. Cellular and Molecular Immunology. 7th Ed, Saunders Elsevier, Philadelphia, PA, USA.
Abdi K., Singh N.J. & Matzinger P. 2012. Lipopolysaccharide-activated dendritic cells: “exhausted” or alert and waiting? J. Immunol. 188: 5981–5989.
Ahmad A., Anjum F.M., Zahoor T., Nawaz H. & Dilshad S.M.R. 2012. Beta glucan: a valuable functional ingredient in foods. Crit. Rev. Food. Sci. Nutr. 52: 201–212.
Baran J., Allendorf D.J., Hong F. & Ross D.G. 2007. Oral β-glucan adjuvant therapy converts nonprotective Th2 response to protective Th1 cell-mediated immune response in mammary tumor-bearing mice. Folia Histochem. Cytobiol. 45: 107–114.
Basic A, Fincher G.B. & Stone B.A. 2009. Chemistry, Biochemistry, and Biology of 1–3 β Glucans and Related Polysaccharides. Academic Press, New York, NY, USA.
Brown G.D., Taylor P.R., Reid D.M., Willment J.A., Williams D.L., Martinez-Pomares L., Wong S.Y. & Gordon S. 2002. Dectin-1 is a major β-glucan receptor on macrophages. J. Exp. Med. 196: 407–412.
Budak F., Göral G. & Oral H.B. 2008. Saccharomicies cerevisiae β-glucan induces interferon-7 production in human T-cells via IL12. Turk. J. Immunol. 13: 21–26.
Carmona E.M., Kottom D.M., Hebrink D.M., Moua T., Singh R.D., Pagano R.E. & Limper A.H. 2012. Glycosphingolipids mediate Pneumocystis cell wall β-glucan activation of the IL-23/IL-17 axis in human dendritic cells. Am. J. Respir. Cell. Mol. Biol. 47: 50–59.
Chan G.C., Chan W.C. & Man-Yuen S.D. 2009. The effects of β-glucan on human immune and cancer cells. J. Hematol. Oncol. 2: 25.
Chen J. & Seviour R. 2007. Medicinal importance of fungal β-(1→3),(1→6)-glucans. Mycol. Res. 3: 635–652.
Chen Y., Chen Y., Yin D., Wang Y., Liu Z., An N., Wen F., Li N., Xin J., Hu X., Zhang H.J. & Yin W. 2016. The Sca-1+ mesenchymal stromal cells modulate macrophage commitment and function. Turk. J. Biol. 40: 473–483.
Dennehy K.M., Willment J.A., Williams D.L. & Brown G.D. 2009. Reciprocal regulation of IL-23 and IL-12 following co-activation of dectin-1 and TLR signaling pathways. Eur. J. Immunol. 39: 1379–1386.
Dubois M., Gilles K.A., Hamilton J.K., Rebers P.A. & Smith F. 1956. Colorimetric method for determination of sugars and related substances. Anal. Chem. 28: 350–356.
Fuentes A.L., Millis L. & Sigola L.B. 2011. Laminarin, a soluble β-glucan, inhibits macrophage phagocytosis of zymosan but has no effect on lipopolysaccharide mediated augmentation of phagocytosis. Int. Immunopharmacol. 11: 1939–1945.
Gersuk G.M., Underhill D.M., Zhu L. & Marr K.A. 2006. Dectin-1 and TLRs permit macrophages to distinguish between different Aspergillus fumigatus cellular states, J. Immunol. 176: 3717–3724.
Giaimis J., Lombard Y., Fonteneau P., Muller C.D., Levy R. & Makaya-Kumba M. 1993. Both mannose and β-glucan receptors are involved in phagocytosis of unopsonized, heat-killed Saccharomyces cerevisiae by murine macrophages. J. Leukoc. Biol. 54: 564–571.
Gorman A., McCarthy J., Finucane J., Reville W. & Cotter T. 1996. Technics in Apoptosis, A User’s Guide. Portland Press, London, UK.
Granucci F., Ostuni R. & Zanoni I. 2012. Generation of mouse bone marrow-derived dendritic cells (BM-DCs). Bio-protocol 2: e226.
Havrlentová M., Petruláková Z., Burgárová A., Gago F., Hlinková A. & Šturdík E. 2012. Cereal β-glucans and their significance for the preparation of functional foods - a review. Czech J. Food Sci. 29: 1–14.
Huang H., Ostroff G.R., Lee C.K., Agarwal S., Ram S., Rice P.A., Specht C.A. & Levitz S.M. 2012. Relative contributions of dectin-1 and complement to immune responses to particulate β-glucans, J. Immunol. 189: 312–317.
Huang H., Ostroff C.R., Lee C.K., Wang J.P., Specht CA. & Levitz S.M. 2009. Distinct patterns of dendritic cell cytokine release stimulated by fungal β-glucans and toll-like receptor agonists. Infect. Immun. 77: 1774–1781.
Hunter Jr. K.W., Gault R.A. & Berner M.D. 2002. Preparation of microparticulate β-glucan from Saccharomyces cerevisiae for use in immune potentiation. Lett. Appl. Microbiol. 35: 267–271.
Javmen A., Grigiškis S. & Gliebuė R. 2012. β-Glucan extraction from Saccharomyces cerevisiae yeast using Actinomyces rut-gersensis 88 yeast lysing enzymatic complex. Biologija 58: 51–59.
Javmen A., Grigiškis S., Rudenkov M. & Mauricas M. 2013. Purification and partial characterization of a novel β-1,3-endoglucanase from Streptomyces rutgersensis. Protein J. 32: 411–417.
Javmen A., Nemeikaitė-Čėnienė A., Grigiškis S., Jonauskienė L, Rudenkov M., Kačianauskas D. & Mauricas M. 2015. S. cerevisiae β-glucan reduced viability of mouse hepatoma cells in vitro, Turk. J. Biol. 39: 562–566.
Jiang H.R., Muckersie E., Robertson M., Xu H., Liversidge J. & Forrester J.V. 2002. Secretion of interleukin-10 or interleukin-12 by LPS-activated dendritic cells is critically dependent on time of stimulus relative to initiation of purified DC culture. J. Leukoc. Biol. 72: 978–985.
Kikuchi T., Ohno N. & Ohno T. 2002. Maturation of dendritic cells induced by Candida β-D-glucan. Int. Immunopharma-col., 2: 1503–1508.
Li B., Cai Y., Qi C., Hansen R., Ding C, Mitchell C.T. & Yan J. 2010. Orally administered particulate β-glucan modulates tumor-capturing dendritic cells and improves antitumor T-cell responses in cancer. Clin. Cancer. Res. 16: 5153–5164.
Masuda Y., Inoue H., Ohta H., Miyake A., Konishi M. & Nanba H. 2013. Oral administration of soluble β-glucans extracted from Grifola frondosa induces systemic antitumour immune response and decreases immunosuppression in tumour-bearing mice. Int. J. Cancer. 133: 108–120.
Miller G.L. 1959. Use of dinitrosalicylic acid reagent for determination of reducing sugar, Anal. Chem. 31: 426–428.
Muramatsu D., Kawata K., Aoki S., Uchiyama H., Okabe M., Miyazaki T., Kida H. & Iwai A. 2014. Stimulation with the Aureobasidium, pullulans-produced β-glucan effectively induces interferon stimulated genes in macrophage-like cell lines. Sci. Rep. 4: 4777.
Nguyen T.H., Fleet G.H. & Rogers P.L. 1998. Composition of the cell walls of several yeast species. Appl. Microbiol. Biotechnol. 50: 206–212.
Noss I., Doekes G., Thorne P.S., Heederik D.J.J. & Wouters I.M. 2013. Comparison of the potency of a variety of β-glucans to induce cytokine production in human whole blood. Innate Immun. 19: 10–19.
Novak M. & Vetvicka V. 2008. β-Glucans, history and the present: immunomodulatory aspects and mechanisms of action. J. Im-munotoxicol. 5: 47–57.
Novak M. & Vetvicka V. 2009. Glucans as biological response modifiers. Endocr. Metab. Immune Disord. Drug Targets 9: 67–75.
Pelizon A.C., Kaneno R., Soares A.M.V.C., Meira D.A. & Sar-tori A. 2005. Immunomodulatory activities associated with β-glucan derived from Saccharomyces cerevisiae. Physiol. Res. 54: 557–564.
Saito Y., Yanagawa Y., Kikuchi K., Iijima N., Iwabuchi K. & Onoé K. 2006. Low-dose lipopolysaccharide modifies the production of IL-12 by dendritic cells in response to various cytokines. J. Clin. Exp. Hematop. 46: 31–36.
Shokri H., Asadi F. & Khosravi A.R. 2008. Isolation of β-glucan from the cell wall of Saccharomyces cerevisiae. Nat. Prod. Res. 22: 414–421.
Sima P., Vannucci L. & Vetvicka V. 2015. Glucans and cancer: historical perspective. Cancer Transi. Med. 1: 209–214.
Soltanian S., Stuyven E., Cox E., Sorgeloos P. & Bossier P. 2009. β-Glucans as immunostimulant in vertebrates and invertebrates. Crit. Rev. Microbiol. 35: 109–138.
Trouvelot S., Heloir M.C., Poinssot B., Gauthier A., Paris F., Guillier C, Combler M., Trdâ L., Daire X. & Adrian M. 2014. Carbohydrates in plant immunity and plant protection: roles and potential application as foliar sprays. Front. Plant Sci. 5: 592.
Vetvicka V 2011. Glucan-immunostimulant, adjuvant, potential drug. World J. Clin. Oncol. 2: 115–119.
Vetvicka V, Terayama K., Mandeville R., Brousseau P., Kourni-kakis B. & Ostroff G. 2002. Pilot study: orally-administered yeast 1,3-glucan prophylactically protects against anthrax infection and cancer in mice. J. Am. Nutraceut. Assoc. 5: 1–6.
Vetvicka V. & Vetvickova J. 2012. β-1,3-Glucan in cancer treatment. Am. J. Immunol. 8: 38–43.
Vetvicka V. & Vetvickova J. 2014. Comparison of immunological effects of commercially available β-glucans. Appl. Sci. Rep. 1: 2.
Vetvicka V & Vetvickova J. 2016. Comparison of immunological effects of commercially available β-glucans: part II. Int. Clin. Pathol. J. 2: 00053.
Vetvicka V & Vetvickova J. 2016. Comparison of immunological effects of commercially available β-glucans: part III. Int. Clin. Pathol. J. 2: 00046.
Yiannikouris A., François J., Poughon L., Dussap C.G., Bertin G., Jeminet G. & Jouany J.P. 2004. Alkali extraction of β-D-glucans from Saccharomyces cerevisiae cell wall and study of their adsorptive properties toward zearalenone. J. Agric. Food Chem. 52: 3666–3673.
Zanoni I., Ostuni R. & Granucci F. 2012. Generation of mouse bone marrow-derived macrophages (BM-MFs). Bio-protocol. 2: e225.
Zhang J.M. & An J. 2007. Cytokines, inflammation and pain. Int. Anesthesiol. Clin. 45: 27–37.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Javmen, A., Nemeikaitė-Čėnienė, A., Grigiškis, S. et al. The effect of Saccharomyces cerevisiae β-glucan on proliferation, phagocytosis and cytokine production of murine macrophages and dendritic cells. Biologia 72, 561–568 (2017). https://doi.org/10.1515/biolog-2017-0063
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2017-0063