Abstract
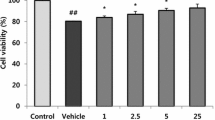
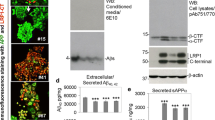
Hypercholesterolemia has been considered as a risk factor for Alzheimer’s disease (AD). In addition to low density lipoprotein (LDL), oxidized LDL plays some roles in AD pathology. Neurodegenerative effect of oxidized LDL was supported by the increased oxidative stress in neurons. To further investigate the role of oxidized LDL, the present study aimed to test its effect on amyloid precursor protein (APP) processing. The release of soluble APP (sAPP) was evaluated in differentiated SH-SY5Y neuroblastoma cells exposed to native (non-oxidized) or oxidized human LDL including mildly and fully oxidized LDL (mox- and fox-LDL). Non-amyloidogenic and amyloidogenic pathways were investigated using specific antibody against sAPPα and sAPPβ. The results demonstrate that oxidized LDL induced neuronal death in dose-dependent patterns. Mox-LDL mediated caspase-3 dependent apoptosis, whereas fox-LDL notably damaged cell membrane. At subtoxic concentration, only native but not oxidized LDL induced the release of sAPP dominantly in amyloidogenic pathway with no change in β-secretase activity. These results suggest that LDL and oxidized LDL play critical roles in AD pathogenesis via different pathways. Elevated serum LDL level together with high oxidative stress may aggravate the progression of AD.
Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- ADAM:
-
A disintegrin and metalloproteinases
- APP:
-
amyloid precursor protein
- Aβ:
-
amyloid beta
- BACE-1:
-
β-site APP cleaving enzyme 1
- BBB:
-
blood-brain barrier
- EDTA:
-
ethylenediaminetetraacetic acid
- FBS:
-
foetal bovine serum
- fox-LDL:
-
fully oxidized-low density lipoprotein
- mox-LDL:
-
mildly oxidized-low density lipoprotein
- sAPPα:
-
soluble amyloid precursor protein alpha
- sAPPβ:
-
soluble amyloid precursor protein beta
References
Bereczki E., Bernat G., Csont T., Ferdinandy P., Scheich H. & Santha M. 2008. Overexpression of human apolipoprotein B-100 induces severe neurodegeneration in transgenic mice. J. Proteome Res. 7: 2246–2252.
Bjelik A., Bereczki E., Gonda S., Juhasz A., Rimanoczy A., Zana M., Csont T., Pakaski M., Boda K., Ferdinandy P., Dux L., Janka Z., Santha M. & Kalman J. 2006. Human apoB overexpression and a high-cholesterol diet differently modify the brain APP metabolism in the transgenic mouse model of atherosclerosis. Neurochem. Int. 49: 393–400.
Chasseigneaux S. & Allinquant B. 2012. Functions of Aβ, sAPPα and sAPPβ: similarities and differences. J. Neurochem. 120 (Suppl. 1): 99–108.
Chen X., Hui L. & Geiger J.D. 2014. Role of LDL cholesterol and endolysosomes in amyloidogenesis and Alzheimer’s disease. J. Neurol. Neurophysiol. 5: 236.
Chen X., Wagener J.F., Morgan D.H., Hui L., Ghribi O. & Geiger J.D. 2010. Endolysosome mechanisms associated with Alzheimer’s disease-like pathology in rabbits ingesting cholesterol-enriched diet. J. Alzheimers Dis. 22: 1289–1303.
Dias I.H., Mistry J., Fell S., Reis A., Spickett C.M., Polidori M.C., Lip G.Y. & Griffiths H.R. 2014. Oxidized LDL lipids increase β-amyloid production by SH-SY5Y cells through glutathione depletion and lipid raft formation. Free Radic. Biol. Med. 75: 48–59.
Gamba P., Testa G., Gargiulo S., Staurenghi E., Poli G. & Leonarduzzi G. 2015. Oxidized cholesterol as the driving force behind the development of Alzheimer’s disease. Front. Aging Neurosci. 7: 119.
Ghribi O., Larsen B., Schrag M. & Herman M.M. 2006. High cholesterol content in neurons increases BACE, β-amyloid, and phosphorylated tau levels in rabbit hippocampus. Exp. Neurol. 200: 460–467.
Hui L., Chen X. & Geiger J.D. 2012. Endolysosome involvement in LDL cholesterol-induced Alzheimer’s disease-like pathology in primary cultured neurons. Life Sci. 91: 1159–1168.
Imtiaz B., Tolppanen A.M., Kivipelto M. & Soininen H. 2014. Future directions in Alzheimer’s disease from risk factors to prevention. Biochem. Pharmacol. 88: 661–670.
Kankaanpaa J., Turunen S.P., Moilanen V., Horkko S. & Remes A.M. 2009. Cerebrospinal fluid antibodies to oxidized LDL are increased in Alzheimer’s disease. Neurobiol. Dis. 33: 467–472.
Keller J.N., Hanni K.B. & Markesbery W.R. 1999. Oxidized lowdensity lipoprotein induces neuronal death: implications for calcium, reactive oxygen species, and caspases. J. Neurochem. 72: 2601–2609.
Koyama A., Stone K. & Yaffe K. 2013. Serum oxidized lowdensity lipoprotein level and risk of cognitive impairment in older women. Neurobiol. Aging 34: 634–635.e2.
Lichtenthaler S.F. 2011. α-Secretase in Alzheimer’s disease: molecular identity, regulation and therapeutic potential. J. Neurochem. 116: 10–21.
Marques F., Sousa J.C., Sousa N. & Palha J.A. 2013. Bloodbrain-barriers in aging and in Alzheimer’s disease. Mol. Neurodegener. 8: 38.
Marwarha G. & Ghribi O. 2015. Does the oxysterol 27-hydroxycholesterol underlie Alzheimer’s disease–Parkinson’s disease overlap? Exp. Gerontol. 68: 13–18.
Matsuura E., Hughes G.R.V. & Khamashta M.A. 2008. Oxidation of LDL and its clinical implication. Autoimmun. Rev. 7: 558–566.
Maulik M., Westaway D., Jhamandas J.H. & Kar S. 2013. Role of cholesterol in APP metabolism and its significance in Alzheimer’s disease pathogenesis. Mol. Neurobiol. 47: 37–63.
Multhaup G., Huber O., Buee L. & Galas M.C. 2015. Amyloid precursor protein (APP) metabolites APP intracellular fragment (AICD), Aβ42, and tau in nuclear roles. J. Biol. Chem. 290: 23515–23522.
Namba Y., Tsuchiya H. & Ikeda K. 1992. Apolipoprotein B immunoreactivity in senile plaque and vascular amyloids and neurofibrillary tangles in the brains of patients with Alzheimer’s disease. Neurosci. Lett. 134: 264–266.
Nowicki M., Muller K., Serke H., Kosacka J., Vilser C., Ricken A. & Spanel-Borowski K. 2010a. Oxidized low-density lipoprotein (oxLDL)-induced cell death in dorsal root ganglion cell cultures depends not on the lectin-like oxLDL receptor-1 but on the toll-like receptor-4. J. Neurosci. Res. 88: 403–412.
Nowicki M., Serke H., Kosacka J., Muller K. & Spanel-Borowski K. 2010b. Oxidized low-density lipoprotein (oxLDL) induces cell death in neuroblastoma and survival autophagy in schwannoma cells. Exp. Mol. Pathol. 89: 276–283.
Pfrieger F.W. & Ungerer N. 2011. Cholesterol metabolism in neurons and astrocytes. Prog. Lipid Res. 50: 357–371.
Poli G., Biasi F. & Leonarduzzi G. 2013. Oxysterols in the pathogenesis of major chronic diseases. Redox Biol. 1: 125–130.
Reitz C. 2013. Dyslipidemia and the risk of Alzheimer’s disease. Curr. Atheroscler. Rep. 15: 307.
Xiong H., Callaghan D., Jones A., Walker D.G., Lue L.F., Beach T.G., Sue L.I., Woulfe J., Xu H., Stanimirovic D.B. & Zhang W. 2008. Cholesterol retention in Alzheimer’s brain is responsible for high β- and γ-secretase activities and Aβ production. Neurobiol. Dis. 29: 422–437.
Yamchuen P., Aimjongjun S. & Limpeanchob N. 2014. Oxidized low density lipoprotein increases acetylcholinesterase activity correlating with reactive oxygen species production. Neurochem. Int. 78: 1–6.
Zhao Z., Zhou H., Peng Y., Qiu C.H., Sun Q.Y., Wang F. & Xie H.N. 2014. Expression and significance of plasma 3-NT and ox-LDL in patients with Alzheimer’s disease. Genet. Mol. Res. 13: 8428–8435.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamchuen, P., Jeenapongsa, R., Nudmamud-Thanoi, S. et al. Low density lipoprotein increases amyloid precursor protein processing to amyloidogenic pathway in differentiated SH-SY5Y cells. Biologia 72, 238–244 (2017). https://doi.org/10.1515/biolog-2017-0024
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2017-0024