Abstract
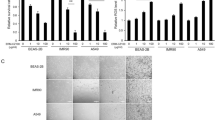
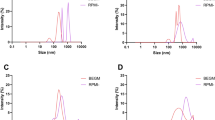
Indoor air pollution occurs due to hazardous pollutants, such as tobacco smoke, pesticides and carbon oxides, sulphur oxides and nitrogen oxides arising from combustion of biomass fuels. Exposure to these pollutants results in respiratory conditions like asthma, chronic obstructive pulmonary disease, lung cancer, pneumonia and other lower respiratory infections. Several of these infections are a result of inflammation and oxidative stress. Here we demonstrate the ability of N, N’-diacetylchitobiose in preventing oxidative DNA damage in peripheral blood mononuclear cells exposed to biomass smoke extracts and cigarette smoke extract. The cytotoxic effect of these pollutants was determined by trypan blue exclusion assay in peripheral blood mononuclear cells, where cytotoxicity in decreasing order was cigarette > wood > sawdust > cowdung. Cytotoxicity could be due to single- and double-strand breaks in the DNA as a result of oxidative stress. Comet assay measures the extent of DNA damage in the cells exposed to toxic agents. When mononuclear cells were treated with N, N’-diacetylchitobiose and later exposed to smoke extracts, the extent of DNA damage decreased by 44.5% and 57.5% as compared to untreated cells. The protection offered by N, N’-diacetylchitobiose towards oxidative DNA damage was at par with quercetin, a popular herbal medicine. Glutathione-S-transferase activity was determined in mononuclear cells exposed to smoke extracts, where oxidative stress in cells exposed to cigarette smoke extract was maximum. The present study demonstrates for the first time the ability of N, N’-diacetylchitobiose to alleviate the harmful effects of indoor air pollutants.
Similar content being viewed by others
Abbreviations
- BSEs:
-
biomass smoke extracts
- COPD:
-
chronic obstructive pulmonary disease
- COS:
-
N-acetylated chitooligosaccharides
- CSE:
-
cigarette smoke extract
- DMSO:
-
dimethyl sulfoxide
- (GlcNAc)2:
-
N, N’-diacetylchitobiose
- GST:
-
glutathione-S-transferase
- IAP:
-
indoor air pollution
- LPG:
-
liquefied petroleum gas
- OS:
-
oxidative stress
- PBMCs:
-
peripheral blood mononuclear cells
- PBS:
-
phosphate buffered saline
- ppm:
-
parts per million
- ROS:
-
reactive oxygen species
- SCGE:
-
single cell gel electrophoresis
- WHO:
-
World Health Organization
References
Azam M.S., Kim E.J., Yang H.S. & Kim J.K. 2014. High antioxidant and DNA protection activities of N-acetylglucosamine (GlcNAc) and chitobiose produced by exolytic chitinase from Bacillus cereus EW5. Springerplus 3: 354.
Balakrishnan K., Sambandam S., Ramaswamy P., Mehta S. & Smith K.R. 2004. Exposure assessment for respirable particulates associated with household fuel use in rural districts of Andhra Pradesh, India. J. Expo. Anal. Environ. Epidemiol. 14: S14–S25.
Basak S.S., Silig Y. & Dogan O.T. 2010. Effects of the exposed to biomass and cigarette smoke on antioxidant defense system, GST activity and GSH levels in rat liver. The 3rd International Congress on Cell Membranes and Oxidative Stress: Focus on Calcium Signaling and TRP Channels, 22–27 June 2010, Isparta, Turkey.
Bazzini C., Rossettia V., Antonio D., Civello D.A., Sassone F., Vezzoli V., Persani L., Tiberio L., Lanata L., Bagnasco M., Paulmichl M. & Meyer G. 2013. Short- and long- term effects of cigarette smoke exposure on glutathione homeostasis in human bronchial epithelial cells. Cell. Physiol. Biochem. 32: 129–145
Bolling A.K., Totlandsdal A.I., Sallsten G., Braun A., Westerholm R., Bergvall C., Boman J., Dahlman H.J., Sehlstedt M., Cassee F., Sandstrom T., Schwarze P.E. & Herseth J.I. 2012. Wood smoke particles from different combustion phases induce similar pro-inflammatory effects in a co-culture of monocyte and pneumocyte cell lines. Part. Fibre Toxicol. 9: 45.
Boot R.G., Blommaart E.F., Swart E., van der Ghauharali V.K., Bijl N., Moe C., Place A. & Aerts J.M.F.G. 2001. Identification of a novel acidic mammalian chitinase distinct from chitotriosidase. J. Biol. Chem. 276: 6770–6778
Bruce N., Neufeld L., Erick B.O. & West C. 1998. Indoor biofuel air pollution and respiratory health: the role of confounding factors among women in highland Guatemala. Int. J. Epidemiol. 27: 454–458
Campbell M.A., Golub M.S., Iyer P., Kaufman F.L., Li L.H., Messan M.F., Morgan J.E. & Donald J.M. 2009. Reduced water intake: implications for rodent developmental and reproductive toxicity studies. Birth Defects Res. B Dev. Reprod. Toxicol. 86:157–157.
Castillo S.S., Levy M., Thaikoottathil J.V. & Goldkorn T. 2007. Reactive nitrogen and oxygen species activate different sphingomyelinases to induce apoptosis in airway epithelial cells. Exp. Cell Res. 313: 2680–2686
Ceylan E., Kocyigit A., Gencer M., Aksoy N. & Selek S. 2006. Increased DNA damage in patients with chronic obstructive pulmonary disease who had once smoked or been exposed to biomass. Respir. Med. 100: 1270–1276
Chen A.S., Taguchi T., Sakai K., Kikuchi K., Wang M.W. & Miwa I. 2003. Antioxidant activities of chitobiose and chitotriose. Biol. Pharm. Bull. 26: 1326–1330
Downs C.A., Montgomery D.W. & Merkle C.J. 2011. Age-related differences in cigarette smoke extract-induced H2O2 production by lung endothelial cells. Microvasc. Res. 82: 311–317
Forchhammer L., Moller P., Riddervold I.S., Bonlokke J., Massling A., Sigsgaard T. & Loft S. 2012. Controlled human wood smoke exposure: oxidative stress, inflammation and microvascular function. Part. Fibre Toxicol. 9: 7.
Fullerton D.G., Bruce N. & Gordon S.B. 2008. Indoor air pollution from biomass fuel smoke is a major health concern in the developing world. Trans. R. Soc. Trop. Med. Hyg. 102: 843–851
Gyori B.M., Venkatachalam G., Thiagarajan P.S., Hsu D. & Clement M.V. 2014. OpenComet: an automated tool for comet assay image analysis. Redox Biol. 2: 457–465
Holz O., Jorres R., Kastner A. & Magnussen H. 1995. Differences in basal and induced DNA single-strand breaks between human peripheral monocytes and lymphocytes. Mutat. Res. 332: 55–62
Hopkins J.M. & Evans H.J. 1980. Cigarette smoke-induced DNA damage and lung cancer risks. Nature 283: 388–390
Létuvé S., Kozhich A., Humbles A., Brewah Y., Dombret M.C. Grandsaigne M. & Pretolani M. 2010. Lung chitinolytic activity and chitotriosidase are elevated in chronic obstructive pulmonary disease and contribute to lung inflammation. Am. J. Pathol. 176: 638–649
Liang T.W., Chen Y.J., Yen Y.H. & Wang S.L. 2007. The an-titumor activity of the hydrolysates of chitinous materials hydrolyzed by crude enzyme from Bacillus amyloliquefaciens V656. Process Biochem. 42: 527–534
Mates J.M., Segura J.A., Alonso F.J. & Marquez J. 2012. Oxidative stress in apoptosis and cancer: an update. Arch. Toxicol. 86: 1649–1665
Musonda C.A. & Chipman J.K. 1998. Quercetin inhibits hydrogen peroxide (H2O2)-induced NF-κB DNA binding activity and DNA damage in HepG2 cells. Carcinogenesis 19: 1583–1589
Nawani N.N., Prakash D. & Kapadnis B.P. 2010. Extraction, purification and characterization of an antioxidant from marine waste using protease and chitinase cocktail. World J. Microbiol. Biotechnol. 26: 1509–1517
Ngo D.N., Kim M.M., Qian Z.J., Jung W.K., Lee S.H. & Kim S.K. 2010. Free radical scavenging activities of low molecular weight chitin oligosaccharides lead to antioxidant effect in live cells. J. Food Biochem. 34: 161–177
Ozturk S., Vatansever S., Cefle K., Palanduz S., Guler K., Erten N., Erk O., Karan M.A. & Tascioglu C. 2002. Acute wood or coal exposure with carbon monoxide intoxication induces sister chromatid exchange. J. Toxicol. Clin. Toxicol. 40: 115–120
Prakash M., Bodas M., Prakash D., Nawani N., Khetmalas M., Mandal A. & Eriksson C. 2013. Diverse pathological implications of YKL-40: answers may lie in ‘outside-in’ signaling. Cell. Signal. 25:1567–1573.
Rekhadevi P.V., Mahboob M., Rahman M.F. & Grover P. 2009. Genetic damage in wood dust-exposed workers. Mutagenesis 24: 59–65
Salgaonkar N., Prakash D., Nawani N.N. & Kapadnis B. P. 2015. Comparative studies on ability of N-acetylated chitooligosaccharides to scavenge reactive oxygen species and protect DNA from oxidative damage. Indian J. Biotechnol. 14: 186–192
Singh N.P., McCoy M.T., Tice R.R. & Schneider E.L. 1988. A simple technique for quantification of low levels of DNA damage in individual cells. Exp. Cell Res. 175: 184–191
Slezak M., Skalniak A., Groszek B., Piatkowski J. & Pach D. 2014. The effect of acute carbon monoxide poisoning on micronuclei frequency and proliferation in human peripheral blood lymphocytes (case-control study). Przegl. Lek. 71: 463–468
Smith K.R. 1993. Fuel combustion, air-pollution exposure and health: the situation in developing countries. Annu. Rev. Energy Environ. 18: 529–566
Smith K.R. 2003. Indoor air pollution implicated in alarming health problems. In: Indoor Air Pollution — Energy and Health for Poor. News Letter 1.
Smith K.R. & Liu Y. 1994. Indoor air pollution in developing countries, pp.151–184. In: Samet J.M. (ed.), Epidemiology of Lung Cancer. Lung Biology in Health and Disease, Marcel Dekker, New York.
Smith K.R., Mehta S. & Maeusezahl-Feuz M. 2004. Indoor air pollution from household use of solid fuels: comparative quantification of health risks. In: Ezzati M., Rodgers A.D., Lopez A.D. & Murray C.J.L. (eds), Comparative Quantification of Health Risks: Global and Regional Burden of Disease due to Selected Major Risk Factors, World Health Organization, Geneva, Switzerland.
Smith K.R., Samet J.M., Romieu I. & Bruce N. 2000. Indoor air pollution in developing countries and acute lower respiratory infections in children. Thorax 55: 518–532
Wang S.L., Hsu W.H. & Liang T.W. 2010. Conversion of squid pen by Pseudomonas aeruginosa K-187 fermentation for the production of N-acetyl chitooligosaccharides and biofertilizers. Carbohydr. Res. 345: 880–885
Wang S.L., Yang C.W., Liang T.W. Peng J.H. & Wang C.L. 2009. Degradation of chitin and production of bioactive materials by bioconversion of squid pens. Carbohydr. Polym. 78: 205–212
WHO. 2000. Air quality guidelines for Europe. WHO regional publications, 91. Copenhagen, Denmark.
WHO. 2009. Global health risks: mortality and burden of disease attributable to selected major risks. WHO publications, Geneva, Switzerland.
WHO. 2011. Indoor air pollution and health: fact sheet No. 292. WHO publications, Geneva, Switzerland.
WHO. 2014. WHO indoor air quality guidelines: household fuel combustion. WHO publications, Geneva, Switzerland.
Acknowledgements
This study was supported by Department of Science and Technology (DST), New Delhi, Government of India, through the research grant (Grant No. SR/S0/HS/0022/2011). NS is thankful to DST for Junior Research Fellowship. This study was partly funded by Swedish International Development Cooperation Agency (SIDA), Sweden, under the grant number AKT-2012-005. The authors are also thankful to Dr. D.Y. Patil Vidyapeeth, Pune, for the necessary infrastructure and support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salgaonkar, N.A., Thakare, P.M., Junnarkar, M.V. et al. Use of N,N’-diacetylchitobiose in decreasing toxic effects of indoor air pollution by preventing oxidative DNA damage. Biologia 71, 508–515 (2016). https://doi.org/10.1515/biolog-2016-0075
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2016-0075