Abstract
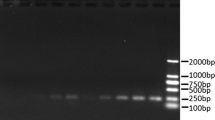
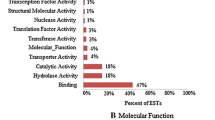
The molecular mechanisms underlying cold-resistance in Prunus campanulata Maxim. (P. campanulata) are not fully understood. This study aimed to establish a full-length library and analyze expressed sequence tags (ESTs) to provide tools to investigate the mechanisms of P. campanulata growth at low temperatures. Based on the switching mechanism at 5’end of RNA transcript technology, a full-length cDNA library was generated from young leaves of P. campanulata after 72 h treatment at 1 °C, and a preliminary EST analysis was carried out. Quantitative reverse transcription polymerase chain reaction was used to assess the expression of selected cold-related genes. The cDNA library titer was 1.2 × 106 cfu/mL−1, with a recombinant rate of 96%. The average size of inserted cDNA fragments was 1.3 Kb. EST data revealed the existence of 834 clones representing a total of 667 unigenes, including 574 singletons and 93 contigs. Blast analysis identified 475 unigenes with known and putative functions. Based on similarity search and GO annotation, 84 unigenes were associated with “response to stimuli”, suggesting that cold stress induced significant alterations in gene expression in P. campanulata cultivated at 1°C for 72 h. Interestingly, DRP, MYB, HSP, GPX and GA20-ox gene expression was significantly up-regulated in plants cultivated at low temperature, while transcript levels of TIL, CDPK were decreased. P. campanulata cultivating at low temperature express genes associated with “response to stimuli”, and in particular DRP, MYB, HSP, GPX and GA20-ox gene are up-regulated while TIL, CDPK are downregulated in response to low temperature-stress.
Similar content being viewed by others
References
Ainsworth C., Tarvis M. & Clark J. 1993. Isolation and analysis of a cDNA cloně encoding the small subunit of ADPglucose pyrophosphorylase from wheat. Plant Mol. Biol. 23: 23–33.
Chen B.-J., Wang Y., Hu Y.-L., Wu Q. & Lin Z.-P. 2005. Cloning and characterization of a droughtinducible MYBgene fromBoea crassifolia. Plant Sci. 168: 493–500.
Chen Z., Lu Y.L. & Wu W.X. 2011. Research advance,prospect and breeding strategy of Cerasus campanulata Maxim. J. of Nanjing Forestry University (Natural Sciences Edition), 36: 1–5.
Chen Z.H., Xiao M. & Chen X. 2008. Change in fiowering dates of japanese cherry blossoms (Prunus campanulata Maxim.) in Wuhan University campus and its relationship with variability of winter temperature. Acta Ecol. Sinica 28: 5209–5217.
Cheong E.J. 2012. Biotechnological approaches for improvement and conservation of Ispecies. Plant Biotech. Rep. 6: 17–28.
Da Silva C., Campos T., Scaloppi E., Maluf M., Goncalves P. & Souza A. 2011. Construction and analysis of a leaf cDNA library from cold stressed rubber tree clones. BMC Proč. 5: 24.
De los Reyes B., Morsy M., Gibbons J., Varma T., Antoine W., McGrath J., Halgren R. & Redus M. 2003. A snapshot of the low temperature stress transcriptome of developing rice seedlings (Oryza sativa L.) via ESTs from subtracted cDNA library. Theor. Appl. Genet. 107: 1071–1082.
Editorial Board for fiora (the Chinese Academy of Sciences) 1986. Fiora of China. Science Press, Beijing, China.
Frenette Charron J.B., Breton G., Badawi M. & Sarhan F. 2002. Molecular and structural analyses of a novel temperature stressinduced lipocalin from wheat and Arabidopsis. FEBS Lett. 517: 129–132.
Futamura N., Totoki Y., Toyoda A., Igasaki T., Nanjo T., Seki M., Sakaki Y., Mari A., Shinozaki K. & Shinohara K. 2008. Characterization of expressed sequence tags from a fulllength enriched cDNA library of Cryptomeria japonica male strobili. BMC Genomics 9: 383.
Imanishi J., Nakayama A., Suzuki Y., Imanishi A., Ueda N., Morimoto Y. & Yoneda M. 2010. Nondestructive determination of leaf chlorophyll content in two fiowering cherries using re- fiectance and absorptance spectra. Landsc. Ecol. Eng. 6: 219–234.
Jung H.A., Kim A.R., Chung H.Y. & Choi J.S. 2002. In vitro antioxidant activity of some selected Prunus species in Korea. Arch. Pharm. Res. 25: 865–872.
Kang S.-G., Jeong H.K. & Suh H.S. 2004. Characterization of a new member of the glutathione peroxidase gene family in Oryza sativa. Mol. Cells 17: 23–28.
Kruse E., Mock H.P. & Grimm B. 1997. Isolation and characterisation of tobacco (Nicotiana tabacum,) cDNA clones encoding proteins involved in magnesium chelation into protoporphyrin IX. Plant Mol. Biol. 35: 1053–1056.
Li H.C., Guo X.L., Wang D.M. & Li G.L. 2010. Responses of HSP70 gene expression to temperature stresses in maize (Zea mays L.). Agric. Univ. of Hebei. 33: 12–15.
Liu M., Shi J. & Lu C. 2013. Identification of stressresponsive genes in Ammopiptanthus mongolicus using ESTs generated from coldand droughtstressed seedlings. BMC Plant. Biol. 13: 88.
Lu Y.L., Chen Z. & Shi J.S. 2006. Research advance,prospect and breeding strategy of Cerasus campanulata Maxim. J. of Nanjing Forestry University Natural (Sciences Edition). 30: 115–119.
Phukon M., Namdev R., Deka D., Módi M.K. & Sen P. 2012. Construction of cDNA library and preliminary analysis of expressed sequence tags from tea plant [Camellia sinensis (L.) O. Kuntze]. Gene 506: 202–206.
Short J.M. & Sorge J.A. 1992. In vivo excision properties of bacteriophage lambda ZAP expression vectors. Methods Enzymol. 216: 495–508.
Singh R.K., Singh S., Pandey P., Anandhan S., Goyary D., Pande V. & Ahmed Z. 2013. Construction of cold induced subtracted cDNA library from Cicer microphyllum and transcript characterization of identi fied novel wound induced gene. Protoplasma 250: 459–469.
Tong Z., Qu S., Zhang J., Wang F., Tao J., Gao Z. & Zhang Z. 2012. A modi fied protocol for RNA extraction from different peach tissues suitable for gene isolation and realtime PCR analysis. Mol. Biotechnol. 50: 229–236.
Tsai T.M., Chen Y.R., Kao T.W., Tsay W.S., Wu C.P., Huang D.D., Chen W.H., Chang C.C. & Huang H.J. 2007. PaCDPKl, a gene encoding calciumdependent protein kinase from orchid, Phalaenopsis amabilis, is induced by cold, wounding, and pathogen challenge. Plant Cell. Rep. 26: 1899–1908.
Wang H., Zou Z., Wang S. & Gong M. 2013. Global analysis of transcriptome responses and gene expression pro files to cold stress of Jatropha curcas L. PLoS One. 8: e82817.
Wang K., Wang Y., Bao Y.L., Meng X.Y., Wu G., Yi J.W. & Li Y.X. 2006. Construction and sequence analysis of a fulllength cDNA library of panax ginseng leaf tissues. J. Mol. Sci. 22: 58–62.
Wei H., Dhanaraj A.L., Rowland L.J., Fu Y., Krebs S.L. & Arora R. 2005. Comparative analysis of expressed sequence tags from coldacclimated and nonacclimated leaves of Rhododendron catawbiense Michx. Planta 221: 406–416.
Wilkie S.E., Roper J.M., Smith A.G. & Warren M.J. 1995. Isolation, characterisation and expression of a cDNA clone encoding plastid aspartate aminotransferase from Arabidopsis thaliana. Plant Mol. Biol. 27: 1227–1233.
Xu Y. 2009. A simplified method for quick screening positive clones and foreign DNA orientation by grouping primers PCR. J. of Chongqing University of Science and Technology (Natural Sciences Edition). 11: 52–54.
Zhang Y.F.J. & Gu R. 2009. Isolation and analysis of cold stress inducible genes in Zea mays by suppression subtractive hybridization and cDNA microarray. Plant Mol. Biol. Report. 27: 38–49.
Zhang Y., Rong J., Chen L., Li S., He T., Chen L. & Zheng Y. 2013. Response of chlorophyll fiuorescence parameters to different temperature stresses in Prunus campanulata Maxim, and Prunus yedoensis Matsum. seedlings. J. Food, Agricult. Environ. 11: 2797–2802.
Acknowledgements
This work was financially supported by Youth Fund of College of Forestry, Fujian Agriculture and Forestry University (6112C039T) and the Major Science and Technology Special Project of Fujian Province (2007SZ0001-8).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, YH., Rong, JD., Chen, LG. et al. Construction of cDNA library from Prunus campanulata leaves and preliminary expressed sequence tag (EST) analysis during cold stress. Biologia 70, 1070–1077 (2015). https://doi.org/10.1515/biolog-2015-0118
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1515/biolog-2015-0118